1Department of Internal Medicine and Endocrinology, 2Laboratory of Endocrinology, University Medical Center Utrecht, Utrecht, 3Department of Endocrinology, Radboud University Nijmegne Medical Center, Nijmegne, The Netherlands
OBJECTIVE Patients with Addison’s disease require lifelong treatment with glucocorticoids. At present, no glucocorticoid replacement therapy (GRT) can exactly mimic normal physiology. As a consequence, under- and especially overtreatment can occur. Suboptimal GRT may lead to various side effects. The aim of this study was to investigate the use of salivary cortisol day curves (SCDC) in the individual adjustment of GRT in order to approach normal cortisol levels as closely as possible, reduce over- and underreplacement and study the short-term effects on quality of life (QoL). DESIGN AND METHODS Twenty patients with Addison’s disease were included in this prospective study. A SCDC was obtained and compared to normal controls; general and disease specific QoL-questionnaires were completed. Based on SCDC assessment of over- and undertreatment (calculated as duration (h) × magnitude (nmol/L) at different time points, glucocorticoid dose and regime were adjusted. After 4 weeks SCDC and QoL assessment were repeated and the effect of adjusting GRT was analysed. RESULTS At baseline, underreplacement was present in 3 and overreplacement in 18 patients; total calculated overreplacement was 32.8 h.nmol/L. Overreplacement decreased significantly to 13.3 h. nmol/L (p =0.005) after adjustment of GRT. Overreplacement was found particularly in the afternoon and evening. After reducing overreplacement in the evening, complaints about sleep disturbances significantly decreased. CONCLUSIONS Individual adjustment of GRT based on SCDC to approach normal cortisol concentrations during the day can reduce overreplacement, especially in the evening. This can lead to a reduction of sleep disturbances and fatigue in patients with Addison’s disease. A SCDC is a simple and patient-friendly tool for adjusting GRT and can be useful in the follow-up of patients with Addison’s disease.
Addison’s disease, Glucocorticoid replacement therapy, Quality of Life, Salivary cortisol day curve
Introduction
Since synthetic corticosteroids have become available, the life expectancy of patients with Addison’s disease treated with lifelong replacement therapy with glucocorticoids and mineralocorticoids has increased dramatically. However, current glucocorticoid replacement therapy (GRT) cannot exactly mimic normal physiology and often leads to overtreatment.1 Chronic overtreatment can lead to serious side effects in the long term, such as osteoporosis, weight gain or increased cardiometabolic risk and even increased mortality.2-5 In addition, suboptimal GRT can also be the cause of reduced quality of life (QoL).6 Optimal GRT can be achieved by mimicking the physiological circadian cortisol secretion pattern as closely as possible. In daily practice, GRT is most often assessed by clinical judgment only. Random serum cortisol or ACTH measurements and 24-hour urinary excretion of free cortisol do not render a precise picture of the fluctuations of cortisol concentrations throughout the day. One means of assessing and adjusting GRT is by performing a serum cortisol day curve, but this is time-consuming and expensive because hospitalization is required.7 On the other hand, assessment of cortisol in saliva has been reported to be a good alternative for plasma cortisol measurements.8,9 A salivary cortisol day curve (SCDC) is inexpensive, easy to perform and non-invasive and can be performed at home.
The first aim of this study was to explore cortisol levels throughout the day in patients with Addison’s disease by using SCDC in order to detect under- and overreplacement. The second was to investigate whether adjustments in glucocorticoid therapy based on SCDC were able to create a more physiological cortisol day curve and whether this had short-term effects on QoL.
Patients and methods
Patients
We prospectively studied 20 adults with established primary adrenal insufficiency attending the Department of Endocrinology of the University Medical Center, Utrecht. All subjects had been on stable GRT via either hydrocortisone or cortisone acetate, with a twice or thrice daily regime. Subjects were excluded if they had acute intercurrent disease or unplanned alteration of GRT dose or regime, if they were pregnant or breastfeeding or if they had gingivitis. Any other additional medical treatment was not changed during the study, including mineralocorticoid replacement and DHEA. None of the 12 women used contraceptives.
QoL assessment
Before and 4 weeks after adjustment of GRT, general and disease specific QoL was assessed using 3 questionnaires. To measure subjective symptoms of fatigue, a 14-item visual analogue scale (VAS) was used.10 This consists of a 100 mm horizontal line anchored by word descriptors at each end. Participants were asked to mark the line on the point that they felt to represent their perception of their current state. A high score represented a low QoL. The second QoL questionnaire was the Addison questionnaire (ADD), used for measuring QoL in Addison patients.11 It is a self-rating scale containing 11 items of common complaints of Addison patients. It has a total score of 44. Each item consists of a question asking about the intensity of a specific complaint. The possible answers range from “not present” (0) to “severe” (4). A low score indicates absence of complaints and good well-being. Finally, the general health questionnaire (GHQ) was employed which measures psychological well-being and focuses on 3 factors: anxiety and depression, social dysfunction and loss of confidence.12 It contains 12 items and every item incorporates 4 possible answers: not at all, no more than usual, more than usual, a lot more than usual. For scoring, the Likert scale (0, 1, 2, 3 from left to right) was used. A score of 11-12 is typical. A score of >15 means evidence of distress and >20 suggests severe problems and psychological distress.
Salivary cortisol day curve
The salivary day curve was performed before (on current GRT) and 4 weeks after adjustment of glucocorticoid dosage and frequency of daily dosage. Saliva was collected by absorption into a cotton roll (Salivettes R, Sarstedt, Numbrecht, Germany). Samples were taken at fixed moments. The first sample was taken shortly after wakening, before the first dosage of glucocorticoids. After 60-120 minutes the second sample was taken. This was also performed before and after the second and (if applicable) third ingestion of glucocorticoids and just before going to sleep. Before a sample was taken and after every dose of glucocorticoid participants had to rinse their mouth with water. They were not allowed to brush their teeth or drink or eat 15 minutes before taking the sample. The salivettes were sent to the laboratory by mail.
Adjustment of salivary cortisol day curve
Glucocorticoid dose and regime were adjusted based on SCDC assessment. We considered overreplacement or underreplacement to be present if cortisol concentrations were respectively above or under the reference range for normal controls (n=59, 20-60 years), i.e. 9-30 nmol/L in the morning (6.00-12.00 a.m.), 4-12 nmol/L in the afternoon (12.00-18.00 p.m.) and 1-6 nmol/L in the evening (18.00-24.00 p.m.). Over- (or under-) replacement was calculated as duration (h) × magnitude (nmol/L) at different time points. The time at which the cortisol concentration was above or below the reference value at the different time points was multiplied by the magnitude of deviation. Optimal replacement over 24 hours was defined as under- or overreplacement of 0 h.nmol/L. All patients on a twice daily regime were given a thrice daily regime in order to facilitate optimal adjustment of SCDC. Based upon the SCDC, the dose of hydrocortisone or cortisone acetate was reduced or increased by 2.5-5 mg at 1 or more time points. After 4 week, SCDC assessment was repeated and the effect of adjusting GRT was analysed.
Saliva cortisol assessment
Cortisol in saliva was assessed using an in-house competitive radioimmunoassay. The lower limit of quantitation was 0.5 nmol/L. The day-to-day variation ranged from 6.5 to 11.5%. Samples were run in duplicate.
Statistical analysis
All values are presented as mean ± SD unless stated otherwise. All statistical analyses were done with IBM SPSS version 20.0. To compare the results of QoL questionnaires, GRT factors and overreplacement before and after adjustment of GRT repeated measures analysis was performed. In the event of violation of the assumption of sphericity, Greenhouse-Geisser values were used. A P <0.05 was accepted as significant for differences between variables and mean scores in all tests.
Results
Baseline characteristics
The baseline characteristics of the studied patients are displayed in Table 1. The majority of the patients used hydrocortisone and had a trice daily regime at baseline. All participants used fludrocortisone and 55% used DHEA, mostly women (83%). The cause of Addison’s disease was autoimmune adrenalitis in 17 (85%) patients. Clinical and laboratory characteristics related to possible under- or overtreatment are shown in Table 2.
SCDC assessment
At baseline, underreplacement at 1 or more of the measured time points was present only in 3 patients. Overreplacement, on the other hand, was seen in 18 patients, mainly occurring in the afternoon and evening (Figure 1). Total calculated overreplacement was 32.8 h.nmol/L. After adjustment of GRT, overreplacement decreased significantly (Figure 2) to 13.3 h nmol/L (p=0.005). Overreplacement decreased more in patients already on a thrice daily regime at baseline (from 36.5 to 14.8 h nmol/L, p=0.017) and in patients using hydrocortisone (from 41 to 15.1 h nmol/L, p=0.008).
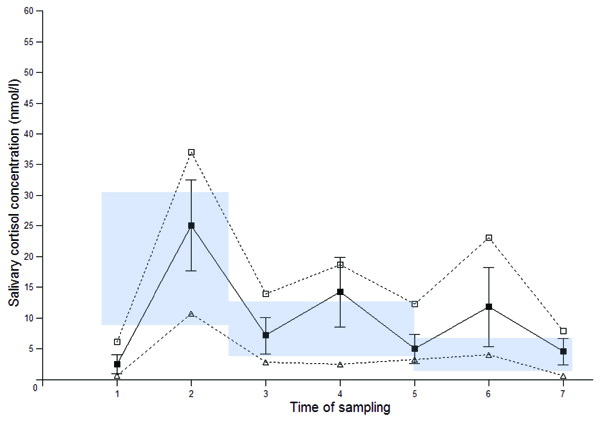
Figure 1. SCDC before adjustment of GRT: solid line mean with SD, dotted lines: range with maximal (□) and minimal (Δ) values. Shaded area: reference range.
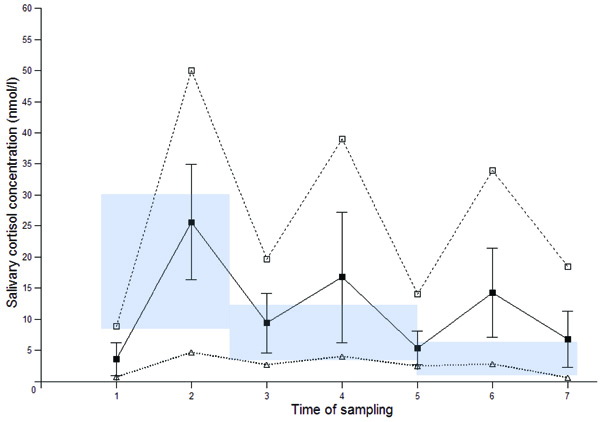
Figure 2. SCDC after adjustment of GRT: solid line mean with SD, dotted lines: range with maximal (□) and minimal (Δ) values.
Shaded area: reference range.
Glucocorticoid dosage
Mean total daily glucocorticoid dosage did not decrease significantly (26 ± 6.1 mg before and 25.4 ± 6.4 mg after adjustment, p= 0.398). After adjustment of GRT there was a significant decrease of the evening glucocorticoid dosage for participants who were on a thrice daily regimen at the start of the study from 7 ± 2.6 to 4.8 ±1.8 mg (p= 0.005); for patients on a twice daily regimen the evening dose also decreased from 7.9 ± 3.3 to 4.7 ± 1.1 mg (p= 0.017). Tables 3 and 4 show the changes in dose at different time points after adjustment of GRT.
QoL scores
After adjustment of GRT, the ADD changed from 14.4 ± 8.6 to 12.9 ± 7.9 (p=0.131), VAS fatigue improved from 44.4 ±26 to 41.8 ± 22.5 (p=0.292) and GHQ scores improved from 12.1±5.3 to 11 ± 3.7 (p=0.349). At the start of the study 70% of patients had a thrice daily regimen. After the first SCDC assessment all other patients were given a thrice daily regimen. All QoL scores improved in patients already on a thrice daily regime at baseline, but only the decrease in VAS fatigue score was significant (p=0.026). We found no significant changes in QoL in patients who switched from a twice to a thrice daily regime. There was no significant correlation between general QoL and total daily glucocorticoid dosage, type of GRT and overreplacement. A large number of patients complained of sleep (65%), concentration (80%) and mood (65%) disturbances before adjustment of GRT. These complaints decreased after adjustment of GRT to 45% (p=0.042), 50% (p=0.010) and 40% (p=0.021) respectively (Table 5 and 6).
Discussion
Patients with Addison’s disease are treated with glucocorticoid replacement therapy (GRT) for life. Currently, no oral glucocorticoid preparation can precisely mimic normal physiological diurnal variation in cortisol levels. As a consequence, under- and overreplacement occur. We found overreplacement in almost all of our patients, particularly during the afternoon and evening. Underreplacement, on the other hand, was only found in a minority. Inadequate GRT can lead to complications of mild overreplacement with glucocorticoids in the long term, such as hypertension, impaired glucose tolerance, immune dysfunction and osteoporosis.13 These complications are in turn responsible for the almost twofold increased risk of premature death from cardiovascular and infectious diseases in patients with Addison’s disease.4,5
Thus, optimizing GRT is of great importance, but minor over- or underreplacement is clinically difficult to detect.14 At present, GRT is assessed mainly by clinical judgment. Various other methods to assess adequacy of GRT have been used, such as random serum cortisol as well as urinary 24-h free cortisol excretion. These are, however, of little value because they do not depict cortisol picture concentrations at various moments during the day. Another method of detecting under- or overreplacement is a serum cortisol day curve. Arlt et al assessed quality of GRT with timed serum cortisol concentrations.15 A newly developed quality of glucocorticoid replacement score based on adding and subtracting points for signs and symptoms of under- or overreplacement was compared to results of three cortisol measurements after morning glucocorticoid dose. Arlt et al found that the mean z score of serum cortisol differed significantly between under- and overreplaced patients but neither group differed significantly from well-replaced patients. Afternoon and evening cortisol measurements were not assessed in their study. A disadvantage of obtaining a serum cortisol day curve is that it is time-consuming and that hospitalization is required, which makes it expensive. In our explorative study we used salivary cortisol day curves (SCDC) to monitor GRT. None of the participants experienced any difficulty in performing a SCDC at home. By using only a single salivary cortisol day curve we were able to reduce overreplacement just by small changes in dosage and regime. The changes did not lead to a significant change of total daily glucocorticoid dose. A SCDC, unlike serum cortisol measurements, can easily be used to detect overreplacement at various moments during the day. In addition, the SCDC is non-invasive and inexpensive and it gives a valid picture of the free cortisol concentrations throughout the day.16-18
An important finding in our study was that overreplacement was found particularly in the afternoon and evening. After reducing overreplacement in the evening, complaints about sleep disturbances significantly decreased. In our patients who switched from a twice to a thrice dosage regime, evening dose significantly decreased. Before adjustment of GRT half of these patients had sleep disturbances, but after dividing and lowering the evening dose sleep disturbances in this group disappeared completely. Sleep disturbances are not unusual in patients with Addison’s disease. Lovas reported sleep disturbances in one third of patients with Addison’s disease, characterized by difficulty falling asleep, repeated awakenings and early morning awakenings.19 A high percentage of patients who took their last glucocorticoid dose after 1800 h reported awakenings as well as tiredness during daily activities. In addition, we found that small adjustments of GRT resulted in a decrease in VAS fatigue scores in patients on a thrice daily regime but had little effect on other general QoL scores.
The main limitation of this study is the relatively small number of subjects studied. This could be responsible for the absence of significant changes in some of the QoL scores. In addition, we did not assess other clinical endpoints such as cardiometabolic risk factors or adrenal crisis as a result of the short study period. Another limitation is that we did not perform a blinded study to investigate the effect of adjustment of GRT on QoL. However, it is reassuring that the reduction in cortisol dose did not seem to be associated with a decrease in QoL. New pharmaceutical formulations have been developed to provide glucocorticoid replacement that replicates physiology more closely. The reported cortisol profiles of these modified and delayed release glucocorticoids and continuous subcutaneous glucocorticoid infusions seem promising.20-23 The long-term beneficial effects of physiological glucocorticoid replacement on QoL will have to be determined in future studies.
In conclusion, individual adjustment of GRT based on SCDC to approach normal cortisol concentrations during the day can reduce overreplacement, especially in the evening. This can lead to a reduction of sleep disturbances and fatigue in patients with Addison’s disease. A SCDC is a simple and patient-friendly tool for assessing GRT and can be useful in the follow-up of patients with Addison’s disease.
Declaration of interest
The authors declare that there is no conflict of interest that would prejudice the impartiality of the research reported.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
References
1. Peacy SR, Guo CY, Robinson AM, et al, 1997Glucocorticoid replacement therapy: are patients over treated and does it matter? Clin Endocrinol 46: 255-261.
2. Zelissen PMJ, Croughs RJ, van Rijk PP, Raymakers JA, 1994 Effect of glucocorticoid replacement therapy on bone mineral density in patients with Addison’s disease. Ann Intern Med 120: 207-210.
3. Giordano R, Marzotti S, Balbo M, et al, 2009 Metabolic and cardiovascular profile in patients with Addison’s disease under conventional glucocorticoid replacement. J Endocrinol Invest 32: 917-923.
4. Bergthorsdottir R, Leonsson-Zachrisson M, Oden A, Johannsson G, 2006 Premature mortality in patients with Addison’s disease: a population-based study. J Clin Endocrinol Metab 91: 4849-4853.
5. Bensing S, Brandt L, Tabaroi F, et al, 2008 Increased death risk and altered cancer incidence pattern in patients withisolated or combined autoimmune primary adrenocortical insufficiency. Clin Endocrinol (Oxf) 69: 697-704.
6. Lovas K, Havard Loge J, Eystein Husebye S, 2002 Subjective health status in Norwegian patients with Addison’s disease. Clin Endocrinol 56: 581-588.
7. Howlett TA, 1997 An assessment of optimal hydrocortisone replacement therapy. ClinEndocrinol 46: 263-286.
8. Lewis JG, 2006 Steroid Analysis in saliva: An overview. Clin Biochem Rev 27: 139-146.
9. Gatti R, Antonelli G, Prearo M, Spinella P, Cappellin E, de Palo EF, 2009 Cortisol assays and diagnostic laboratory procedures in human biological fluids. Clin Biochem 42: 1205-1217.
10. Chalder T, Berelowitz G, Pawlikowska T, et al, 1993 Development of a fatigue scale. J Psychosomatic res 37: 147-153.
11. Bleicken B, Hahner S, Loeffler M, Ventz M, Allolio B, Quinkler M, 2008 Impaired subjective health status in chronic adrenal insufficiency: impact of different glucocorticoid replacement regimens. Eur J Endocrinol 159: 811-817.
12. Goldberg DP, Hillier VF, 1979 A scaled version of the General Health Questionnaire.Psychol Med 9: 139-145.
13. Lovas K, Giesdal CG, Christensen M, et al, 2009 Glucocorticoid replacement therapy and pharmacogenetics in Addison’s disease: effects on bone. Eur J Endocrinol 160: 993-1002.
14. Crown A, Lightman S, 2005 Why is management of glucocorticoid deficiency still controversial: a review of the literature. Clinical Endocrinol 62: 1-10.
15. Arlt W, Rosenthal C, Hahner S, Allolio B, 2006 Quality of glucocorticoid replacement in adrenal insufficiency: clinical assessment vs. timed serum cortisol measurements. Clin Endocrinol 64: 384-389.
16. Aardal E, Holm A, 1995 Cortisol in saliva-Reference ranges and relation to cortisol inserum. Eur J Clin Chem Biochem 33: 927-932.
17. Wong V, Yan T, Donald A, Mclean M, 2004 Saliva and bloodspot cortisol: novel sampling methods to assess hydrocortisone replacement therapy in hypoadrenal patients. Clin Endocrinol 61: 131-137.
18. Lovas K, Thorsen TE, Husebye ES, 2006 Saliva cortisol measurement: simple and reliable assessment of the glucocorticoid replacement therapy in Addison’s disease. J Endocrinol Invest 29: 727-731.
19. Lovas K, Husebye ES, Holstein F, Bjorvatn B, 2003 Sleep disturbances in patients withAddison’s disease. Eur J Endocrinol 148: 449-456.
20. Johannsson G, Bergthorsdottir R, Nilsson AG, Lennernas H, Hedner T, Skrtic S, 2009 Improving glucocorticoid replacement therapy using a novel modified-release hydrocortisone tablet: a pharmacokinetic study. Eur J Endocrinol 161: 119-130.
21. Lovas K, Husebye ES, 2007 Continuous subcutaneous hydrocortisone infusion in Addison’s disease. Eur J Endocrinol 157: 109-112.
22. Debono M, Ghobadi C, Rostami-Hodjegan A, et al, 2009 Modified-release hydrocortisone to provide circadian cortisol profiles. J Clin Endocrinol Metab 94: 1548-1554.
23. Johannsson G, Nilsson AG, Bergthorsdottir R, 2012 Improved cortisol exposure-time profile and outcome in patients with adrenal insufficiency: a prospective randomized trial of a novel hydrocortisone dual-release formulation. J Clin Endocrinol Metab 97: 473-481.
Address for correspondence:
Lisanne C.C.J. Smans, University Medical Center Utrecht,
P.O. Box 85500, 3508 GA Utrecht, The Netherlands,
Tel.: +31 88 7556304, Fax: +31 88 7555514,
e-mail: L.C.C.J.Smans@umcutrcht.nl
Received 08-08-12, Accepted 11-12-12