Department of Endocrinology, Institute of Medicine, Sahlgrenska Academy, University of Gothenburg and Sahlgrenska University Hospital, Gothenburg, Sweden
OBJECTIVE: Diabetes insipidus (DI)
and primary polydipsia (PP) are characterised by polyuria and polydipsia. It is
crucial to differentiate between these two disorders since the treatment is
different. The aim of this study was to evaluate the diagnostic value of the
short and an extended variant of the water deprivation test (WDT) and of
measuring urinary vasopressin (AVP) in patients with polyuria and polydipsia.
DESIGN: A retrospective, single-centre study based on WDTs performed between
2004 and 2014 including 104 consecutive patients with the polyuria-polydipsia
syndrome. During a strict water deprivation, weight, urinary osmolality,
urinary vasopressin and specific gravity were collected until one of the
following was reached: i) >3% weight reduction, ii) Urinary specific gravity
>1.020 or, urinary osmolality >800 mOsm/L, iii) Intolerable adverse
symptoms such as excessive thirst.
RESULTS: Out of 104 patients (67 women, 37
men), 21 (20%) were diagnosed with DI and 83 (80%) with PP. The median
(interquartile range; range) test duration was 14 hours (10-16; 3-36) in
patients with DI and 18 hours (14-24; 7-48) in patients with PP (P=0.011).
Of those diagnosed with PP, 22 (26%) did not reach urinary specific gravity
>1.020 nor urine osmolality >800 mOsm/L. Urine AVP did not overlap
between patients with PP and patients with central DI.
CONCLUSIONS: The short
WDT is of limited value in the diagnostic work-up of polydipsia and polyuria
and a partial DI may have been missed in every fourth patient diagnosed with
PP. Urinary AVP has excellent potential in discriminating PP from central DI.
Diabetes insipidus, Polydipsia, Polyuria, Primary polydipsia, Urinary vasopressin, Water deprivation test
INTRODUCTION
The polyuria-polydipsia syndrome is defined by chronic diuresis of abnormally large volumes (>30ml/kg/day) of non-concentrated urine accompanied by increased thirst and fluid intake.1 There are three types of the polyuria-polydipsia syndrome. Central, or neurogenic, diabetes insipidus (DI) is caused by a failure to produce and secrete the antidiuretic hormone agrinine-vasopressin (AVP). Nephrogenic DI is caused by AVP insensitivity in the kidneys. In both central and nephrogenic DI thirst, and subsequently water intake, are increased in order to reduce the rising plasma osmolality. The third cause of the polydipsia-polyuria syndrome is called primary polydipsia (PP), to distinguish it from the secondary polydipsia that follows the water loss in DI. AVP secretion in PP tends to be reduced due to lower plasma osmolality, while urine output is increased in order to prevent overhydration.2-4
It is crucial to accurately determine the aetiology of the polyuria-polydipsia syndrome as each underlying entity requires different treatment. The cornerstone of the diagnostic algorithm is a water deprivation test (WDT), often followed by administration of synthetic AVP (desmopressin). This protocol, originally developed by Miller et al,5 has been used with various modifications in order to assess urinary concentrating ability as a function of AVP activity. Often, a diagnostic attempt is made on the basis of the clinical setting in which DI occurred, as well as based on urinary concentrating ability after an overnight (short) water deprivation. Only a few clinical studies, on a small series of patients, have been published to validate its utility, as recently reviewed by Fenske and Allolio.6
Direct evaluation of AVP activity by measuring plasma AVP has not been completely successful in exploring the polyuria-polydipsia syndrome because of the well-known pre-analytical instability of plasma AVP, as well as problems with the AVP assay.6,7 Nevertheless, measurement of urinary AVP seemed to be a promising approach when studied by Dunger et al in children,8 and later by Diederich9 et al in adults, with the polyuria-polydipsia syndrome.
The aim of this study was to evaluate the clinical value of the short WDT, and an extended variant of the WDT, as well as to assess the diagnostic potential of urinary AVP in evaluating patients with the polyuria-polydipsia syndrome.
MATERIAL AND METHODS
Study design and participants
This was a retrospective, single-centre study based on results from 104 consecutive WDTs performed between 2004 and 2014. All patients had been referred to our department for evaluation of polyuria and/or polydipsia. Other causes of polyuria and polydipsia such as diabetes mellitus, hypercalcaemia and hypokalaemia had been ruled out prior to the WDT. Patients with the polyuria-polydipsia syndrome, referred for WDT, with urine osmolality >800 mOsm/L at baseline were excluded from the analysis (n=3).
All patients were followed for up to 12 years after the WDT with regard to treatment, i.e., whether they were on desmopressin treatment or not, and to any residual symptomatology related to the polyuria-polydipsia syndrome.
Water deprivation protocol
The same protocol was used during the entire study period and conducted by the same nurse. The patients were admitted to our ward the day before the test started. Patients with a reported urine volume <7 litres per 24 hours started the test at midnight, while those with a reported urine volume >7 litres started the test at 08:00 AM. Consumption of any liquids during the test was strictly prohibited and patients were not allowed to leave the ward. The test continued, without any duration limits, until one of the following criteria was met: i) More than 3% weight reduction, ii) Urinary specific gravity above 1.020 or, urine osmolality above 800 mOsm/L (considered to be diagnostic for PP), and iii) Intolerable adverse symptoms such as excessive thirst. Body weight, urine osmolality and urinary specific gravity were measured on every occasion the patient urinated throughout the test and not at predefined time points except for additional testing 8 hours after initiation of the WDT. Serum sodium and osmolality were measured at baseline, at 8 hours and at the end of the test. In the case of low urine osmolality <300 mOsm/Kg and a weight reduction of at least 3%, 4 µg of desmopressin (Minirin®; Ferring SA Holding Lausanne, CH. Switz.), was administered intravenously in order to differentiate between central and nephrogenic DI. In case of desmopressin administration, additional measurements of urine osmolality and urinary specific gravity were performed after one and three hours. At the end of the test, the diagnosis of PP, DI or partial DI was made with regard to the overall clinical setting of the patient.
In addition, urinary AVP was measured at the start and at the end of the WDT between 2008 and 2013 (n=54).
Biochemical analyses
Serum and urine osmolality were measured by depression of the freezing point method (2400 Osmometer, Fiske, Norwood, MA, USA) during the whole study period. The analytical range was 0-2000 mOsm/kg. The repeatability of the method was as follows: 0-400 mOsm/kg H2O: ±3 mOsm/kg and 400-2000 mOsm/kg H2O: ±0.75%, respectively.
Urinary specific gravity was assessed with Hydrometer ALLA360.100PMG, 1000-1060, 0.001g/ml, standardised at 20°C (ALLA France, Chemillé en Anjou - France). If the hydrometer reading was taken at a temperature other than the standard temperature for the hydrometer, the reading would be in error due to the change in volume of the hydrometer between the two temperatures. In that case, appropriate corrections were made according to the manufacturer’s instructions.
Urinary AVP was measured as follows. Samples of 200 µL and calibrators of 100 µL were analysed in duplicates. After addition of 100 µL AVP antiserum (Baylis), final dilution 1/72 000 (until June 4, 2010), or a final dilution 1/80 000 (from June 15, 2010) in assay buffer (0.35% bovine serum albumin in 0.05 M sodium phosphate buffer, pH 7.4), samples were incubated at 6°C for 24 hours. A second incubation in the same manner was performed after addition of 100 µL 125I-AVP [Radioactive ligand was synthesised using a modified chloramine-T method,10 purified with RP-HPLC using a µ-Bondapak C18 column (3.9×300 mm, 125 Å, 10 µm, Waters Code no. 27324), diluted 1/10 in CH3CN and stored at -20°C until use] (diluted to 5000 cpm 10% in assay buffer). Free and bound tracer were separated using 100 µL anti-rabbit IgG (AA-Sac1 from IDS), incubation at room temperature for 30 min, addition of 1 mL deionised water and centrifugation (2500×g, 21°C, 5 min). The supernatant was discarded and the precipitate measured in a gamma counter [automatic gamma counter, Wizard 1470, counting efficiency 75%, connected with an immunoassay software programme (MultiCalc Advanced, Wallac Oy, Finland)]. Mean coefficients of variation were 16% (range 13–18%) for internal control samples with a concentration of 0.4 pmol/L, 17% (range 15–20%) for 0.8 pmol/L and 17% (range 15–19%) for 2.2 pmol/L.
Ethical considerations
The study was approved by the Ethics Committee at the University of Gothenburg. The study was conducted according to the Declaration of Helsinki.
Statistical methods
All statistical analyses were performed with SPSS, version 22.0 for Windows. Data are presented as median [interquartile range (IQR)] for continuous variables and percentages for categorical variables. Regarding urinary AVP concentrations and duration of the WDT in the PP and DI group, respectively, range is also given. For comparison between groups, DI versus PP, the Mann-Whitney U-test was used for continuous variables and the χ2 test was used for proportions. The significance level was set at 0.05 (two-sided p-value). Receiver operating characteristic (ROC) analysis of urine osmolality was performed in order to identify the cut-offs that give the best sensitivity in relation to specificity.
RESULTS
Subjects characteristics
The cohort consisted of 104 consecutive patients, of whom 64 (62%) were women. The median (IQR) age was 43 (30-58) years. The median reported fluid intake upon referral was 5 (4-6) litres per day (Table 1).
There was no difference in the reported fluid intake prior to the WDT between patients later diagnosed with DI as compared to patients later diagnosed with PP (Table 1; Figure 1). In contrast, serum osmolality and serum sodium levels were higher, whereas urine osmolality and urinary specific gravity were lower in patients with DI compared to those with PP.

Figure 1. Box plots (median, interquartile range, 5th and 95th percentile) showing: A) Reported fluid intake prior to the WDT, B) Serum osmolality, C) Urine osmolality and, D) Urinary specific gravity, at baseline in patients diagnosed with diabetes insipidus and primary polydipsia.
Water deprivation test, DI versus PP
Of 104 patients, 21 (20%) were diagnosed with DI and 83 (80%) with PP. Seventeen patients had central DI in whom the most common cause was iatrogenic DI following transsphenoidal pituitary surgery (n=5), or other pituitary lesions [hypophysitis (n=2), Langerhans histiocytosis (n=2), Wegener’s granulomatosis (n=1), posttraumatic (n=1), unknown (n=1) and idiopathic DI (n=5). Four patients had nephrogenic DI, one due to treatment with lithium.
The median
(IQR; range) time to termination of the test was 14 hours (10-16; 3-36) in
patients with DI and 18 hours (14-24; 7-48) in patients with PP (P=0.010;
Table 2 and Figure 2). Of those diagnosed with PP, 22 (26%) did not reach
either urinary specific gravity above 1.020 or urine osmolality above 800
mOsm/L. Only 9 patients met the criteria for termination of the test within 8
hours, of whom 4 (24%) were diagnosed with DI and 5 (6%) with PP (Table 2).
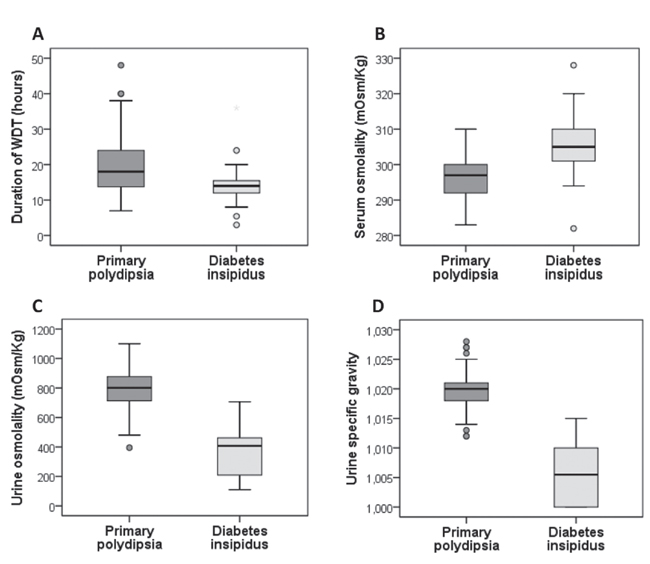
Figure 2. Box plots (median, interquartile range, 5th and 95th percentile) showing: A) Duration of the water deprivation test (WDT), B) Serum osmolality, C) Urine osmolality and, D) Urinary specific gravity, at the end of WDT in patients diagnosed with diabetes insipidus and primary polydipsia.
The median weight loss was 3.0% (2.1-3.4) in patients with DI and 2.1% (1.3-3.0) in patients with PP (Table 2). At the end of the WDT, serum osmolality, serum sodium and urine output volume were higher in patients with DI than in those with PP, whereas urine osmolality and urinary specific gravity were lower.
ROC analysis was performed for calculation of the cut-off values of urine osmolality that best differentiate DI from PP. A cut-off level of 600 mOsm/kg gave the best predictive value for DI diagnosis [area 0.96, (95% CI 0.93-1.0); P <0.001] with sensitivity and specificity of 90%, respectively.
All patients with PP had urine osmolality >400 mOsm/kg, corresponding to 100% sensitivity and 47% specificity (i.e., every other patient with urine osmolality >400 mOsm/kg had partial DI). None of the patients with DI had urine osmolality >710 mOsm/kg. The cut-off of 710 mOsm/kg corresponded to 100% sensitivity and 76% specificity for DI (i.e., every fourth patient with urine osmolality <710 mOsm/kg had PP).
When the cut-off value was set even higher at 800 mOsm/kg the specificity was 100% for excluding DI (i.e., only patients with PP had urinary concentrating capacity greater than 800 mOsm/kg), but the sensitivity in the ROC analysis was only 51% (i.e., up to every other patient with urinary concentrating capacity of less than 800 mOsm/kg could have had PP).
Urinary AVP at the start and the end of WDT
Urinary AVP was analysed in 54 patients: 41 with PP, 10 with central DI and 3 with nephrogenic DI. Patients with central DI had lower urinary AVP than patients with PP, both at the start and at the end of the WDT (Figure 3). In fact, the urinary AVP at the end of the WDT could discriminate central DI [median 2 (0.7-3.4); range 1-5 pmol/L] from PP [median 22 (13-40); 6-122 pmol/L] in 100% of the cases (Figure 3). The three patients with nephrogenic DI had urinary AVP concentrations comparable with patients with PP (data not shown).

Figure 3. Box plots (median, interquartile range, 5th and 95th percentile) of urinary arginine-vasopressin (AVP) concentration at the start (left) and at the end (right) of the water deprivation test (WDT) in patients with primary polydipsia and central diabetes insipidus, respectively.
Follow-up of the clinical diagnosis
The median follow-up time was 3 (5-7) years. All patients diagnosed with DI (n=21) were still on desmopressin treatment by the time of follow-up. Of 83 patients with PP, 3 (4%) reported residual symptoms and/or had started treatment with desmopressin.
DISCUSSION
This study, the largest series on WDTs performed at a single study centre, demonstrates the limited value of the short overnight test. Specifically, only 20% of the patients with DI met the diagnostic criteria within eight hours. For the vast majority it took, roughly, at least double the time. Similarly, the diagnosis of PP was made within the same time frame in only 6% of the patients. Furthermore, 26% of the patients who received the diagnosis of PP did not reach the cut-off of 800 mOsm/kg in urine osmolality, despite an unlimited duration of the WDT, for up to 48 hours in one case. In other words, every fourth patient in the cohort, with possible partial DI, may have been misdiagnosed with PP.
The polyuria-polydipsia syndrome still remains a diagnostic challenge. Even though considerable efforts have been made to develop new methods to improve the accuracy of the diagnostic work-up, for instance measurements of direct AVP or copeptin, the WDT remains the gold standard.6 Although the test has been used for decades, convincing cut-off levels that would add to the test’s ability to differentiate between different aetiologies of the polyuria-polydipsia syndrome, in particular milder forms of DI and PP, are still lacking. The majority of the few studies available to date have evaluated the urine concentrating capacity after the administration of hypertonic solutions and not the most widely used WDT.11-13
Very few studies have validated the clinical utility of the WDT. In a study by Fenske et al, similar criteria were used as in the current study (with the exception that the duration of the test was limited to 16 hours), aiming at diagnosing DI or PP in 50 patients with the polyuria-polydipsia syndrome.14 Without taking AVP measurements into account, the authors reported an overall correct diagnosis in 70% of the participants and in only 41% of patients with PP. The reference diagnosis was based on clinical information and treatment response. In a recently published study by de Fost et al, the diagnostic performance of WDT was evaluated prospectively in 40 patients with polyuria, followed for up to 18 years after the WDT had been performed.15 The authors achieved a sensitivity of 100% in diagnosing DI when lowering the urine osmolality cut-off to 680 mOsm/kg compared to 96% using the widely accepted limit of 800 mOsm/kg. Similarly, in our study a reduced cut-off in urine osmolality at 710 mOsm/kg guaranteed 100% sensitivity in diagnosing DI.
It is obvious that the real challenge in clinical praxis is to discriminate PP from partial DI and the present study confirms the pitfalls in the currently recommended work-up algorithms. In fact, none of the variables analysed with the WDT can be used to perfectly discriminate between the two disorders. In agreement with Fost et al,15 lowering the threshold of urine osmolality to 710 mOsm/kg safely rules out DI. However, it must be kept in mind that one fourth of patients with PP may still have urine osmolality below the cut-off of 710 mOsm/kg and can therefore not be distinguished from patients with DI.
On the other hand, in our cohort urinary AVP showed a remarkable diagnostic capacity to discriminate PP from central DI, including patients with partial DI. Despite the fact that earlier studies showed a promising performance of urinary AVP in both polyuric disorders,16 as in normal subjects,17 by testing it in both children8,18 and adults,9 this variable has not been integrated into clinical practice. As previously demonstrated, contrary to plasma AVP, urinary AVP is more stable19 and exists in much higher concentrations than in plasma8,17,16 so that even less-sensitive RIAs are equally adequate to detect it.9 Thus, our results are in full agreement with those of Diederich et al9 and the authors of the current report suggest taking the measurement of urinary AVP into account when assessing the various polyuric states.
The underlying mechanisms of the various disorders behind the polyuria-polydipsia syndrome contribute in the overlapping phenotypes in terms of symptoms and biochemical response to the WDT, thus making the interpretation of the results demanding. Patients with partial DI have still a considerable capacity to produce AVP under severe dehydration, whereas patients with acquired nephrogenic DI can have incomplete resistance to AVP.1 Furthermore, patients with partial DI may present with normal urine concentrations, especially those with decreased glomerular filtration rate.3,20 On the other hand, the chronic water diuresis in patients with PP may result in a “washout” of the renal medullar concentrating gradient and a down-regulation of the expressed aquaporin 2 channels and can, in this way, decrease the urinary concentration capacity.21-23
The major strength of the present report is the large number of WDTs included, all performed by the same experienced nurse at a single centre, using the same protocol throughout the study period. Moreover, the spectrum of the different underlying diagnoses was fairly wide, providing a clear patient-oriented picture of the efficacy of the WDT. The limitations of the study are the retrospective design and that the follow-up of the clinical diagnosis was limited to reviewing the medical journals available in our region. Unfortunately, not many cases with central DI were included in the present cohort since the vast majority of central DI diagnoses in our clinic were made in the clinical setting of postoperative pituitary surgery or known hypothalamic-pituitary lesion. In addition, AVP or copeptin concentrations in serum were not taken into account. Nevertheless, the main aim of the study was to evaluate the diagnostic value of the WDT in terms of urine concentrating capacity and urine AVP, and in that regard no additional biochemical tests were warranted.
In conclusion, the short WDT is of limited value in the diagnostic work-up of patients with the polyuria-polydipsia syndrome when the evaluation is based solely on urinary osmolality. Despite the unlimited test duration for up to 48 hours, a partial DI may have been missed in every fourth patient. Urinary AVP had excellent diagnostic power in fully discriminating PP from all forms of central DI and could be useful in the diagnostic work-up of the polyuria-polydipsia syndrome. Larger studies are however warranted in order to further evaluate the clinical relevance of urinary AVP.
DECLARATION OF INTEREST
None of the authors has any conflict of interest.
FUNDING
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
AUTHOR CONTRIBUTIONS
PT and OR performed the patient selection process and data collection. All authors contributed to the design of the study as well as to data interpretation and analysis, and in writing and revising the report. All authors are responsible for the integrity of the data and accuracy of the analysis, and all approved the final report.
REFERENCES
1. Robertson GL,1995 Diabetes insipidus. Endocrinol Metab Clin North Am 24: 549-572.
2. Robertson GL,1987 Dipsogenic diabetes insipidus: a newly recognized syndrome caused by a selective defect in the osmoregulation of thirst. Trans Assoc Am Physicians 100: 241-249.
3. Barlow ED, De Wardener HE, 1959 Compulsive water drinking. Q J Med 28: 235-258.
4. Goldman MB, Luchins DJ, Robertson GL, 1988 Mechanisms of altered water metabolism in psychotic patients with polydipsia and hyponatremia. N Engl J Med 318: 397-403.
5. Miller M, Dalakos T, Moses AM, Fellerman H, Streeten DH, 1970 Recognition of partial defects in antidiuretic hormone secretion. Ann Intern Med 73: 721-729.
6. Fenske W, Allolio B, 2012 Clinical review: Current state and future perspectives in the diagnosis of diabetes insipidus: a clinical review. J Clin Endocrinol Metab 97: 3426-3437.
7. Robertson GL, 2016 Diabetes insipidus: Differential diagnosis and management. Best Pract Res Clin Endocrinol Metab 30: 205-218.
8. Dunger DB, Seckl JR, Grant DB, Yeoman L, Lightman SL, 1988 A short water deprivation test incorporating urinary arginine vasopressin estimations for the investigation of posterior pituitary function in children. Acta Endocrinol (Copenh) 117: 13-18.
9. Diederich S, Eckmanns T, Exner P, Al-Saadi N, Bahr V, Oelkers W, 2001 Differential diagnosis of polyuric/polydipsic syndromes with the aid of urinary vasopressin measurement in adults. Clin Endocrinol (Oxf) 54: 665-671.
10. Hunter WM, Greenwood FC, 1962 Preparation of iodine-131 labelled human growth hormone of high specific activity. Nature 194: 495-496.
11. Zerbe RL, Robertson GL 1981 A comparison of plasma vasopressin measurements with a standard indirect test in the differential diagnosis of polyuria. N Engl J Med 305: 1539-1546.
12. Milles JJ, Spruce B, Baylis PH, 1983 A comparison of diagnostic methods to differentiate diabetes insipidus from primary polyuria: a review of 21 patients. Acta Endocrinol (Copenh) 104: 410-416.
13. Moses AM, Streeten DH, 1967 Differentiation of polyuric states by measurement of responses to changes in plasma osmolality induced by hypertonic saline infusions. Am J Med 42: 368-377.
14. Fenske W, Quinkler M, Lorenz D, et al, 2011 Copeptin in the differential diagnosis of the polydipsia-polyuria syndrome--revisiting the direct and indirect water deprivation tests. J Clin Endocrinol Metab 96: 1506-1515.
15. de Fost M, Oussaada SM, Endert E, et al, 2015 The water deprivation test and a potential role for the arginine vasopressin precursor copeptin to differentiate diabetes insipidus from primary polydipsia. Endocr Connect 4: 86-91.
16. Miller M, Moses AM, 1972 Urinary antidiuretic hormone in polyuric disorders and in inappropriate ADH syndrome. Ann Intern Med 77: 715-721.
17. Miller M, Moses AM, 1972 Radioimmunoassay of urinary antidiuretic hormone in man: response to water load and dehydration in normal subjects. J Clin Endocrinol Metab 34: 537-545.
18. Dunger DB, Broadbent V, Yeoman E, et al, The frequency and natural history of diabetes insipidus in children with Langerhans-cell histiocytosis. N Engl J Med 321: 1157-1162.
19. Tausch A, Stegner H, Leake RD, Artman HG, Fisher DA, 1983 Radioimmunoassay of arginine vasopressin in urine: development and application. J Clin Endocrinol Metab 57: 777-781.
20. Berliner RW, Davidson DG, 1957 Production of hypertonic urine in the absence of pituitary antidiuretic hormone. J Clin Invest 36: 1416-1427.
21. De Wardener HE, Herxheimer A, 1957 The effect of a high water intake on the kidney’s ability to concentrate the urine in man. J Physiol 139: 42-52.
22. Epstein FH, Kleeman CR, Hendrikx A, 1957 The influence of bodily hydration on the renal concentrating process. J Clin Invest 36: 629-634.
23. Harrington AR, Valtin H, 1968 Impaired urinary concentration after vasopressin and its gradual correction in hypothalamic diabetes insipidus. J Clin Invest 47: 502-510.
Address for correspondence:
Oskar Ragnarsson; Section for Endocrinology, Sahlgrenska University Hospital, Gröna Stråket 8, SE-413 45, Gothenburg, Sweden; Tel.: +46 31 3421000; Fax: +46 31 821524; E-mail: oskar.ragnarsson@medic.gu.se
Received: 25-08-2017, Accepted: 20-09-2017