1Department of 2nd General Surgery, Sisli Etfal Training and Research Hospital, Sisli, 2Golden Horn University, Aksaray, 3Department of Pathology, Sisli Etfal Training and Research Hospital, Sisli, Istanbul, Turkey
The malignancy risk is low in hot thyroid nodules verified by scintigraphy. We present a rare case of papillary carcinoma, initially treated as an autonomous hot nodule. Case report. A 36-year old male patient with a hot thyroid nodule and subclinical hyperthyroidism was treated with 10mCi 131I. On admission, both 99mTc and 131I thyroid scintigraphic imaging revealed a hot nodule at the right lobe accompanied by lower uptake in the remaining thyroid tissue. After treatment, there was a progressive increase in the nodule size; a fine needle aspiration biopsy was thus performed which showed findings compatible with papillary thyroid cancer. The patient was referred to our department for further management. Total thyroidectomy with right central neck dissection was performed. The pathologic examination showed that the whole nodule (1.5 cm diameter) was a columnary type papillary thyroid cancer. Conclusion: In the case of a small-sized toxic thyroid nodule, the possiblility of malignancy cannot be totally ruled out. Suspicious hot nodules should be cytologically evaluated before radioactive iodine treatment to determine the existing malignancy risk. Fine needle aspiration biopsy should be performed in all hot thyroid nodules that increase in size after radioactive iodine treatment.
Autonomous thyroid nodule, Hot nodule, Papillary thyroid cancer, Radioactive iodine theraphy, Solitary thyroid nodule
INTRODUCTION
Thyroid nodules are the most frequent thyroid disorder. They may be a feature of many thyroid diseases. The vast majority of thyroid nodules are benign. The main clinical problem is to distinguish the malignant lesions requiring surgical treatment from the benign ones.1,2 Usually, malignant thyroid nodules do not take up radionuclides and appear as “cold” areas in thyroid scintigraphy. However, on rare occasions malignancy may be found in functional nodules.1-3
We describe the unexpected diagnosis of malignancy in a patient with a hyperfunctioning solitary thyroid nodule, which had been treated with radioiodine.
PATIENT’S DESCRIPTION
A 36-year old male patient living in an endemic, iodine deficient area currently under iodized salt supplementation, was admitted to a medical center with the complaint of excessive sweating, even over little effort, of 6 months duration. The laboratory investigation showed: TSH<0.05 uIU/ml (0.34-5.60), FT3 3.8 pg/ml (normal limits 1.9-4.9), FT4 1.75 ng/dl (normal limits 1-1.8). 99mTc thyroid scintigraphy revealed a hyperactive nodule at the superior-medial side of the right lobe accompanied by reduced activity in the remaining thyroid gland (Figure 1). 131I scintigraphy confirmed the hyperactivity of the nodule at the right lobe and the uptake of 131I at 24 hours was 25.5% (normal range: 15-35%) (Figure 2). The thyroid ultrasonography (US) showed a hypoechoic nodule at the right lobe, 14x12 mm in size, containing internal echos and normal parenchymal echogenicity in the remaining thyroid gland.
Figure 1. Hyperactive nodule at the right lobe and minimal activity in the remaining areas shown in 99m Tc scintigraphy.
The patient was prescribed a treatment dose of 10 mCi of radioidine 131I (RAIT), which he received as an outpatient at a Nuclear Medicine Center.
At follow-up after the radioactive iodine treatment, TSH returned to the normal range and the sweating problem improved. At the US investigation after 5 months, the nodule at the right lobe was 16x12mm in diameter and hypoechoic. Six months later the thyroid hormone levels were normal. However, a slight further increase of the nodule size was detected in the ultrasound, which now measured 18x14mm (Figure 3). A fine needle apiration biopsy (FNAB) of the nodule was performed and found to be positive for papillary thyroid carcinoma. No pathological lymphadenopathy could be detected by cervical US. Chest radiography and abdominal US findings were normal. At this point, the patient was referred to our department for further evaluation.
Total thyroidectomy with right central neck dissection was performed. Multiple lymph nodes—the largest being 0.5 cm—were palpated at the right central part of the neck. No palpable lymphadenopathy was evident at the left cervical area. Right central neck dissection was performed with protection of the recurrent laryngeal nerve and of the superior and inferior parathyroid glands. Pathology disclosed columnary type papillary thyroid cancer, occupying almost all of the 1.5 cm nodule, which contained some colloid and was covered by cells forming solid groups and resembling high columnary epithelium in the lumen (Figure 4). The patient was discharged on the first postoperative day. The postoperative follow-up did not show any residual or recurrent disease.
DISCUSSION
The incidence of nodular thyroid disease as well as that of functioning thyroid nodules is increased in iodine deficient areas. Malignancy, however, is rare in hot nodules.1-3
Some thyroid nodules may give different images with different radioactive tracers. Thus, well-differentiated thyroid cancer nodules may seem warm or hot with 99m Tc and cold with 131I.4 If the nodule appears hot with both types of scintigraphies, the probability of malignancy is very low.5 In our case, the nodule was imaged as hot with both Tc99m and 131I scintigraphy.

Figure 2. The 24th hour appearance of the hyperactive nodule at the right lobe in 131I scintigraphy.
However, it is recognised that there is a possibility of cancer in autonomous nodules.1-3,6 The case herein described presented with subclinical hyperthyroidism before RAIT. In the paper by Mizukami et al7 thyroid cancer was detected in 12% of cases with autonomous nodules. Niedziela et al8 found an increase in the incidence of hot nodules in children in a region of iodine deficiency after salt iodination; cancer incidence in hot nodules as high as 29% was reported in this study. Based on 31 cases, this study also disclosed an association between the incidence of malignancy and the uptake of the radiopharmaceutical in the remainder of the gland. Only one out of the 17 cases, where the radioactivity uptake was completely suppressed in the extranodular thyroid, proved malignant, yielding an incidence of 5.9%; on the other hand, in the remaining 14 cases where there was evident (although reduced) uptake of the radionuclide by the gland parenchyma outside the nodule, the cancer incidence was considerably higher (57.1%). Our case thus shows marked similarity with this second high risk group of Niedziela’s patients; our patient lived in an area with iodine deficiency, recently under iodized salt supplementation, and the scintigraphy showed minimal uptake in the extranodular thyroid areas.

Figure 3. Hypoechoic nodule 18x14mm in size at the right lobe after one year of 131I treatment.
Due to different definitions and criteria, it is difficult to determine the real incidence of cancer in hyperfunctioning thyroid nodules.9 ln the literature, autonomous differentiated thyroid cancers are very rare and primarly reported as sporadic cases.4,9,10 Majima et al11 found 25 such cases in their study published in 2005. In addition, thyrotoxicosis due to thyroid carcinoma has been described in association with extensive functioning metastases.12
The points that need to be addressed are the expected change in nodule size after radioiodine treatment and the possibility that histology may have been affected by the previous radioiodine. Although some studies showed that in 70-90% of patients the toxic thyroid nodule remains palpable after RAIT, O’Brien et al13 noted complete nodular regression in 56.3% of patients in the first year after treatment. On the other hand, Erdogan et al14 consider that complete nodular regression after RAIT does not occur, and observed a 50% reduction in the size of toxic nodules over a period of 12 months. In our case the increase size of the nodule was the observation that lead to further evaluation and diagnosis. Some nonspecific histologic changes may appear in the thyroid after the treatment of hyperthyroidism with radioactive iodine.15 At the FNABs carried out after RAIT, nuclear changes suggestive of either papillary or anaplastic carcinoma may be seen.16 Furthermore, thyroid cancers that appeared after RAIT for hyperthyroidism have been reported. In these cases, however, the latent period was generally longer than 3 years with a mean of 11.4 years.17 In another study, no malignant change was observed, either clinically or cytologically, during a follow-up period of up to 20 years following RAlT.15 Thus, the oncogenic effect of RAIT on the adult thyroid gland has yet to be proven.18
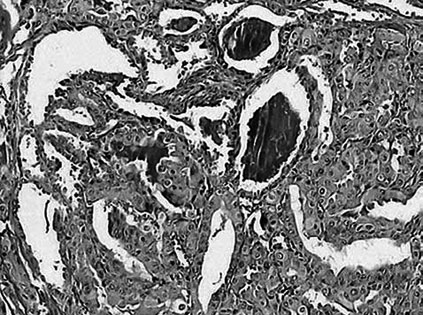
Figure 4. Papillary thyroid cancer that covers almost all of the nodule, containing colloid in some parts only, covered by cells forming solid groups and resembling high columnary epithelium (Hematoxylin-Eosin stain X 100).
In our case, since only one year had passed after the 131I treatment, we assume that the autonomous nodule itself was malignant already at presentation. The increasing instead of decreasing size after apparently successful RAIT may be considered as a sign of malignancy. Furthermore, the nodule was found to harbor papillary carcinoma. Although the majority of such cases are follicular cancers, papillary thyroid carcinoma has also been described.7 Before the wide application of FNAB, the traditional work-up in routine studies usually began with thyroid scintigraphy.2 Although sintigraphy has considerable value in the identification of autonomously functioning thyroid tissue, it presents major limitations with respect to its ability to distinguish benign from malignant lesions. Thus, because of its diagnostic accuracy, FNAB should be the initial procedure used in nodule evaluation.19 On the other hand, the serum TSH levels are measured as a first step; hyperfunctioning nodules are rarely malignant, so if there’s no suspicious clinical feature, observation alone or treatment with RAl can be a reasonable alternative to surgery.3,20 Routine biopsy of a hot nodule may prevent unnecessary surgery.3
We conclude that a functioning/toxic thyroid nodule, especially when of small diameter, does not totally rule out malignancy. ln thyroid scintigraphy, minimal activity with incomplete suppression of radio¬pharmaceutical uptake in the extranodular thyroid areas may be a risk factor of malignancy. Fine needle aspiration biopsy should be performed in the case of a hot thyroid nodule accompanied by suspicious clinical features. If RAIT is given to a patient with toxic nodules, the structural (anatomic) progress should be followed, as increase of the nodule size may be a sign of malignancy. In such cases, FNAB should be performed in order to exclude the possibility of malignancy.
REFERENCES
1. Wong CK, Wheeler MH, 2000 Thyroid nodules: Rational management. World J Surg 24: 934-941.
2. Gharib H, 1997 Changing concepts in the diagnosis and management of thyroid nodules. Endocrinol Metab Clin North Am 26: 777-799.
3. Erdogan MF, Anil C, Ozer D, Kamel N, Erdogan G, 2003 Is it useful to routinely biopsy hot nodules in iodine deficient areas? J Endocrinol Invest 26: 128-131.
4. Turner JW, Spencer RP, 1976 Thyroid carcinoma presenting as a pertechnetate hot nodule, but without 131I uptake: case report. J Nucl Med 17: 22-23.
5. Appetecchia M, Ducci M, 1998 Hyperfunctioning differentiated thyroid carcinoma. J Endocrinol Invest 21: 189-192.
6. Falk SA 1997 Surgical treatment of hyperthyroidism. In: Falk SA (ed) Thyroid Disease, Lippincott-Raven, New York; pp, 319-340.
7. Mizukami Y, Michigishi T, Nonomura A, et al, 1994 Autonomously functioning (hot) nodule of the thyroid gland. A clinical and histopathologic study of 17 cases. Am J Clin Pathol 101: 29-35.
8. Niedziela M, Breborowicz D, Trejster E, Korman E, 2002 Hot nodules in children and adolescents in western Poland from 1996 to 2000: clinical analysis of 31 patients. J Pediatr Endocrinol Metab 15: 823-830.
9. Lupi A, Orsolon P, Cerisara D, Deantoni-Migliorati G, Vianello-Dri A, 2002 “Hot” carcinoma of the thyroid. Case reports and comments on the literature. Minerva Endocrinol 27: 53-57.
10. Ducci M, Appetecchia M, Marzetti A, 1996 Differentiated carcinoma in autonomously functioning thyroid nodule: case report. Acta Otorhinolaryngol Ital 16: 281-285.
11. Majima T, Doi K, Komatsu Y, et al, 2005 Papillary thyroid carcinoma without metastases manifesting as an autonomously functioning thyroid nodule. Endocr J 52: 309-316.
12. Paul SJ, Sisson JC, 1990 Thyrotoxicosis caused by thyroid cancer. Endocrinol Metab Clin North Am 19: 593-612.
13. O’Brien T, Gharib H, Suman VJ, van Heerden JA, 1992 Treatment of toxic solitary thyroid nodules: surgery versus radioactive iodine. Surgery 112: 1166-1170.
14. Erdogan MF, Kucuk NO, Anil C, et al, 2004 Effect of radioiodine therapy on thyroid nodule size and function in patients with toxic adenomas. Nucl Med Commun 25: 1083-1087.
15. Oz F, Urgancioglu I, Uslu I, Dervisoglu S, Oz B, Kanmaz B, 1994 Cytologic changes induced by 131I in the thyroid glands of patients with hyperthyroidism; results of fine needle aspiration cytology. Cytopathology 5: 154-163.
16. Granter SR, Cibas ES, 1997 Cytologic findings in thyroid nodules after 131I treatment of hyperthyroidism. Am J Clin Pathol 107: 20-25.
17. Tezelman S, Grossman RF, Siperstein AE, Clark OH, 1994 Radioiodine-associated thyroid cancers. World J Surg 18: 522-528.
18. Schlumberger M, De Vathaire F, 1996 131 iodine: medical use. Carcinogenic and genetic effects. Ann Endocrinol (Paris) 57: 166-176.
19. Gharib H, 2004 Changing trends in thyroid practice: understanding nodular thyroid disease. Endocr Pract 10: 31-39.
20. Castro MR, Gharib H, 2000 Thyroid nodules and cancer. When to wait and watch, when to refer. Postgrad Med 107: 113-116.
Address for correspondence:
M. Uludag, General Surgeon, Atakent Mah. 3. Etap, Blok:
D21/1, D:1, Kucukcekmece, Istanbul, Turkey (34303),
Tel.: 90 212 231 22 09/ext. 1580, 90 212 470 06 88,
Fax: 90 212 283 26 70, e-mail: drmehmetuludag@yahoo.com.tr,
drmuludag@hotmail.com
Received 03-05-07, Revised 10-09-07, Accepted 20-11-07