1Endocrinology and Metabolism Center of Chania, 22nd Surgical Department Athens Naval and Veterans’ Hospital, Athens, 3Department of Pathology University Hospital of Heraklion, 4Department of General Surgery University Hospital of Heraklion, Crete, Greece
OBJECTIVE: To evaluate the application of color flow Doppler (CFD) sonography in the preoperative management of benign and malignant cold thyroid nodules. DESIGN: Eighty-five patients with a cold thyroid nodule larger than 1cm were examined with CFD sonography prior to thyroidectomy. The histological examination revealed that 18 (21%) patients had malignant nodules and 67 (79%) had benign ones. The sonographic characteristics of the nodules used for evaluation were: a) absence of vascularization, b) presence of peripheral vascularization, c) presence of central vascularization and d) size of the nodule. The correlations between the pre-operative sonographic characteristics, as defined above, and the histological findings of the nodules were determined. results: The results showed that the vascular signals were absent in 4/18 (22%) of malignant nodules and 16/67 (23%) of benign ones. The smaller nodules (<2.5cm.) were more frequently avascular (15/37=40%) compared to the larger ones (5/48=10%) (p<0.05). Moreover, for the nodules with vascularization, the peripheral distribution of vascularization was more frequently encountered in benign nodules (p<0.01 specificity=0.77, sensitivity=0.46), while central vascularization was more frequent in malignant ones (p<0.01, specificity=0.70, sensitivity=0.66). Thus, absence of vascularization in a nodule does not exclude the probability of malignancy, since vascularization depends on the size rather than the histological features of the nodule. Furthermore, for the vascularized nodules, peripheral distribution of vascularization is a characteristic of benignancy with high specificity, while central distribution is a characteristic of malignancy with medium specificity. CONCLUSIONS: CFD sonography contributes to the differential diagnosis of the large vascularized nodules, but it is less helpful in the smaller non-vascularized ones.
Color Doppler sonography, Power Doppler sonography, Thyroid cancer, Thyroid nodules
INTRODUCTION
Thyroid nodules are very frequent in the general (Crete) Greece, population. The prevalence of palpable nodules in subjects over the age of 50 is reported to vary from 4% to 21%, while for sonographically diagnosed nodules it varies from 21% to 67%.1-5 On the other hand, thyroid carcinoma is relatively rare since it constitutes only 1% of all carcinomas encountered in humans. Therefore the possibility that a thyroid nodule may harbor a carcinoma is relatively small (5%).6 Moreover, most thyroid carcinomas are of a papillary type, which usually has a good prognosis.7 The pre-operative management of thyroid nodules is a relatively strenuous process because there is no method with 100% sensitivity and specificity for the identification of thyroid cancer.8 The examinations usually performed are biochemical tests, such as the measurement of TSH, calcitonin and thyroid autoantibodies, which offer little, however, in the diagnosis of the common types of thyroid cancer. Furthermore, isotopic scintiscan differentiates cold from hot nodules with a high specificity but low sensitivity, since nearly all the malignant nodules are cold but only 10-15% of cold nodules are malignant.9,30 Fine Needle Aspiration (FNA) biopsy has the highest sensitivity (85-95%) and specificity (85-95%); however, it is interventional and not always accepted by patients.10,11 Ultrasonography provides information on the size and the location of the nodules and additionally helps in performing ultrasound-guided aspiration biopsy in small non-palpable ones. Moreover, it has been reported, and we have also confirmed this in a previous study,12 that, with the new generation high-resolution ultrasound machines, it is possible to obtain useful information for the differential diagnosis between benign and malignant thyroid nodules.12-14
Today the use of color flow Doppler sonography (CFDS) is more widely applied for the assessment of suspected malignant tumors.25-29 However, there is significant disagreement among specialists concerning the validity of the method in the differential diagnosis of benign and malignant thyroid nodules. Some researchers claim that it is of great value while others do not agree with this notion.15-18 The aim of our study was to evaluate the role of CFDS in the preoperative management of cold thyroid nodules.
Patients and Methods
Between 1997 and 2004 we examined more than 1000 patients with thyroid nodules of different sizes using CFDS. In this retrospective study we included 85 patients with solitary and multiple thyroid nodules larger than 10mm, who were submitted to thyroidectomy after the sonographic examination. All the nodules included in this evaluation were cold on the isotopic scintigram. They underwent surgical resec-tion owing to risk of malignancy (82 patients), compressive symptoms (2 patients) and cosmetic reasons (1 patient). We considered a nodule as being high risk for malignancy if it had clinical or sonographic characteristics predictive for malignancy (firm nodule,irregularshape,rapidgrowth,abnormalcontour, hypoechogenicity, microcalcifications), as well as if the FNA biopsy was suspicious for malignancy. Certainly, the patient selection for surgery was not based on the preoperative CFDS findings. The histological examination revealed that 18 patients (15 women and 3 men between the ages of 20 and 86) had malignant nodules (11 papillary, 4 follicular, 2 Hurthle cell, 1 undifferentiated carcinoma). The remaining 67 (53 women and 14 men aged 27 to 75 years) had benign nodules (60 hyperplastic nodules and 7 follicular ad-enomas) (Table 1 ). Twenty-six out of 67 benign and 5 out of 18 malignant nodules were solid with cystic degeneration and the remaining were pure solid. We did not include any pure cyst in our material. More-over, in one previous study we did not find any correlation between cystic degeneration and the malignant feature of a nodule.12 The sonographic examinations were performed by the same operator (E.V.) using the same machine (ESAOTE BIOSOUND USA MODEL AU4 IDEA). The scanner was equipped with a linear transducer operating at 7.5-10 MHz for morphological study and 4.5-7 MHz for color flow Doppler evaluation. The CFD examinations were performed with biplanar scanning. The amplifier gain was adjusted in each case at a level to block the appearance of random color noise. Besides CFD, which estimates the relative velocity and direction of the blood cells in the vessels, the instrument also provided the power Doppler system. This is a color flow mapping technique used to map the strength of the Doppler signal coming from the flow rather than the frequency shift of the signal. This technique enables the system to estimate the blood flow based on the number of blood cells regardless of their velocity. It is considered by certain authors to be more sensitive than conventional color Doppler in the imaging of the small vessels of the tumors.29 The color and power Doppler settings were standardized in all cases. The sonographic characteristics studied were: a) the total absence of vascularization of the nodules (Figure 1) and the correlation of this feature with their size and their histology, b) the presence of central vascularization (Figures 2, 3) and correlation with their size and histology, and c) the presence of peripheral vascularization of the nodules (Figure 4) and correlation of this finding with their size and histological features. We considered a nodule as being without vascularization if no flow signals were detected were inside or in the periphery of the nod-ule. A nodule was considered as having peripheral vascularization if the flow signals existed only or mainly in the periphery of the nodule while a nodule wasdesignatedashavingcentralvascularizationifthe flow signals presented only or mainly at the central part of the nodule. There were no cases in which the ultrasound could not determine which type of vascularization was predominant.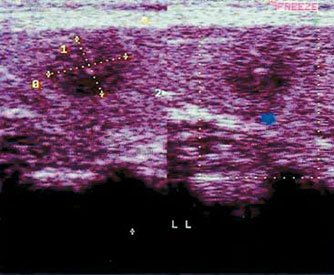
Figure 1. Small (10 mm.) papillary carcinoma without vascularization.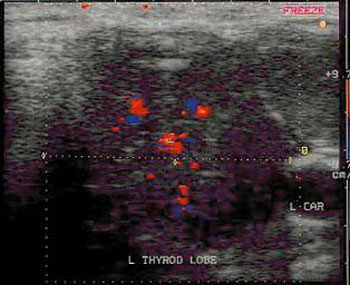
Figure 2. Papillary carcinoma 32 mm in size with central vascularization.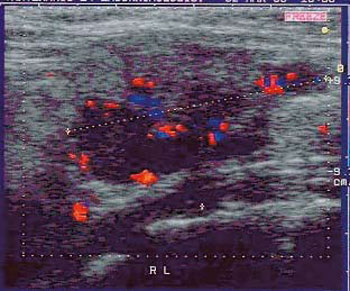
Figure 3. Follicular carcinoma 34 mm in size with central vascularization.
Figure 4. Benign cold nodule 38 mm in size with peripheral vascularization.
STATISTICS
Statistical analysis of the findings was carried out using the x2 test with Yates correction. A p-value of less than 0.05 was considered to indicate statistical significance.
RESULTS
Thirty-seven out of 85 nodules (43.5%) were smaller than 2.5cm, while the remaining (56.5%) were larger than 2.5cm. Fifteen out of 37 nodules smaller than 2.5cm (40%), and 5 out of 48 nodules larger than 2.5 cm (10%) did not present any vascularization.Thedifferencewasstatisticallysignificant: x2= 8.92, p<0.01. Twelve out of 37 nodules smaller than 2.5 cm (32.4%) and 21 out of 48 nodules larger than 2.5cm (43.7%) had peripheral vascularization. The difference was not statistically significant: p>0.1. Furthermore, 10 out of 37 nodules smaller than 2.5cm (27%) and 22 out of 48 nodules larger than 2.5cm (45.8%) had central vascularization. The dif-ference was not statistically significant: p>0.1. We thus found that the small nodules were more often avascular than the larger ones and that the pattern of vascularization (central or peripheral) did not correlate with the size of the nodules (Tables 2 & 3 , Figure 5).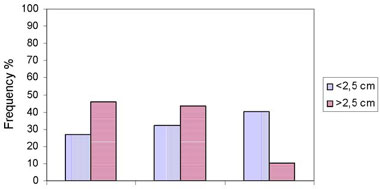
Figure 5. Frequency (%) of the various vascular patterns of the nodules according to their size.
Sixteen out of 67 benign nodules (23.9%) and 4 out of 18 malignant nodules (22.2%) did not have vascularization. The difference was not statistically significant: p>0.1. We therefore found that the ab-sence of vascularization did not correlate with the benign or malignant feature of the nodules.
Thirty-one out of 67 benign nodules (46.3%) and 2 out of 18 malignant (11.1%) had peripheral vascularization. The difference was statistically significant:X2=5.9,p<0.01.Wethusfoundthatperipheral vascularization is a feature of a benign nodule with high specificity (16/18=0.88) but low sensitivity (31/67=0.46).
Twenty out of 67 benign (29.8%) nodules and 12 out of 18 malignant ones (66.6%) had central vascularity. The difference was statistically significant: x2=6.6 p<0.01. We thus found that central vascularization of a nodule is a feature of malignancy with medium specificity (47/67=0.70) and sensitivity (12/18=0.66) (Tables 4 & 5 , Figure 6).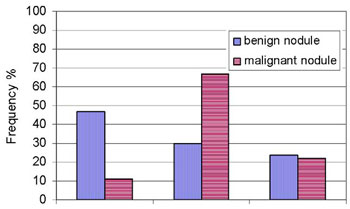
Figure 6. Frequency (%) of the various vascular patterns in benign and malignant nodules.
DISCUSSION
During the past decade the application of CFD sonography has greatly increased in the differential diagnosis of benign and malignant tumors. This is be-cause malignant tumors usually present with higher vascularization than benign ones. CFDS, based on the Doppler phenomenon, calculates the difference in the frequency of emission and reception of an ultrasound beam reflecting the moving blood cells inside the vessels and providing color images of the vessels on a gray scale image. It also provides flow information including the maximum blood velocity and the resistance index. Certain authors therefore propose the use of the power Doppler, a color flow mapping technique that usually comes with CFD and is used to measure the strength of the Doppler signal emitted from the tissue rather than the frequency shift of the signal. Using this technique, the ultrasound system plots color flow based on the number of reflectors that are moving, regardless of their velocity.29 Some reports in the literature under-line the usefulness of color flow Doppler and power Dopplersonography inthediagnosisofcertainforms of malignant tumors in various organs, such as liver, breast, parotid gland, ovary, kidney, prostate tumors, as well as in the differential diagnosis of lymph nodes enlargement.25-29 However, the role of CFDS in the context of preoperative management of thyroid nod-ules remains controversial. Several authors have claimed that color Doppler sonography helps considerably,16,20,22,24 while others consider this method of limited diagnostic value.17,19
In our study the vascular patterns and the size of the nodules were correlated with their malignant or benign histologic features. We found that nodules smaller than 2.5cm were more frequently avascular than the larger ones, although the pattern of vascurarization (central or peripheral) did not correlate with the size of the nodules. Furthermore, the entire absence of vascularization was found at nearly the same frequency in benign (23%) and malignant (22%) cold nodules. We therefore conclude that the absence of vascularization correlates with the size of the nodules, but not with their benign or malignant feature.Consequently,theabsenceofvascularization does not exclude the probability of malignancy of a nodule. On the other hand, in our material, 4 of the 11 papillary carcinomas did not show vascularization, probably because some papillary carcinomas tend to be fibrotic and consequently non-vascularised. By contrast, all the follicular, Hurthle cell and undifferentiated carcinomas (n=7) had increased vascu-larization.Fratesetalalsoreportedthatthepresence of vascularization in compact nodules increases the probability of malignancy by nearly 45%. However, they stated that the absence of vascularitzation cannot be cited to exclude malignancy, since 14% of the nodules without vascularization in their study were malignant.19 Moreover, Shiamamoto et al found that the detection of color signals inside the thyroid nodule depends on its size but not on its histologic features.17
We selected cold nodules for our study because we observed that hot nodules, which are always benign, nearly always had peripheral and central flow signals, whereas cold nodules, of which 15% were malignant, were only 40-50% vascularized. Clark et al also reported that when examined by color Doppler sonography, cold nodules are presented mainly with peripheral vascularization and hot ones with central vascularization. They therefore concluded that color Doppler sonography cannot reliably distinguish benign from malignant thyroid nodules.18
Among the vascularized nodules we found that the presence of peripheral vascularization was statistically more frequent in benign nodules. In contrast, central vascularization was statistically more frequent in malignant nodules than in benign ones. We thus consider that peripheral vascularization is indicative of benignancy with high specificity while central vascularization is indicative of malignancy with medium specificity. Mesina et al reported that a large majority of malignant nodules (90%) have in-ternal vascularization. They also suggested the use of color Doppler sonography as a routine examination in conjunction with FNA biopsy in the diagnosis of thyroid nodules.20 Holden also reported that all neoplastic nodules (adenomas, carcinomas) contained intranodular flow signals, while the majority of colloid nodules were either avascular or had halo flow signals only.16 In our study we found that only 66% of malignant nodules had internal vascularization.
Furthermore, Solbiati et al reported that the presence of intranodular flow signals is a characteristic suggestive of malignancy, particularly if it is combined with microcalcifications.24 Microcalcifications are shown by high-resolution conventional ultrasonography as hyperechoic dots inside the nodule smaller than 2mm, occasionally with an acoustic shadow behind them. In a previous study we found that microcalcifications were characteristic of malignancy with low sensitivity (56%) but very high specificity (89%).12 Papini et al studied non-palpable nodules 8-15mm in size and found that 74% of ma-lignant nodules had internal flow signs, while 19% of the benign ones had internal vascularity. They consider that internal vascularization is an independent risk factor of malignancy, just as are irregular marginsandmicrocalcifications.21 Wealsofoundthat central vascularization is an independent risk fac-tor of malignancy; however, in our material 40% of nodules smaller than 2.5 cm were a vascular regardless of their histological features. Rago et al stated that the presence of intranodular flow signals does not alone constitute a feature of malignancy; however, the coexistence of increased internal vascularization and the absence of a peripheral halo is an important characteristic of malignancy.22 These findings seem toconfirmourown,suggestingthatperipheralvascu-larization in CFDS, which represents the peripheral halo of conventional sonography, is a characteristic of benignancy, while central vascularization constitutes a feature of malignancy.
Argalia et al observed that vascular patterns alone are not particularly helpful in distinguishing among thyroid nodules; however, they pointed out that the measurement of Peak Velocity and Resistance Index may be very useful in the characterization of solid nodules and the selection of patients to undertake FNA.23 Other researchers such as Spiezia et al propose the use of power Doppler sonography in combination with Doppler velocity measurement and the assessment of the vascularres is tance indexto improve the diagnostic protocol and management of thyroid nodules.31 We simultaneously used color flow Doppler and power Doppler sonography to examine our patients, but we observed no difference between the two methods in depicting the vascular characteristics of both benign and malignant nodules. However, we did not measure any vascular parameters, such as resistance index or maximum velocity.
In conclusion, color Doppler sonography contributes to the differential diagnosis of vascularised large nodules, although it is less helpful in non-vascularized small ones. Peripheral vascularization is a feature of benignancy with high specificity, although it can also be revealed by high resolution conventional sonography. Central vascularization is a feature of malignancy with medium specificity. CFD sonography helps only in the differential diagnosis of cold nodules; therefore, it should be combined with an isotopic scintiscan. Accordingly, we consider CFDS to be a useful diagnostic modality in the preopera-tive management of thyroid nodules, although the primary means should be FNA Biopsy.
REFERENCES
1. Mazzaferi EL, De Los Santos ET, Rofagna-Keyhani S, 1988Solitarythyroidnodule:Diagnosisandmanagement. Med Clin North America 72: 1177-1211.
2. Gharib H, 1997 Changing concepts in the diagnosis and management of thyroid nodules: Endocrinol Metab Clin North Am 26: 777-800.
3. Ezzat S, Sarti DA, Cain DR, Braunsein GD, 1999 Thyroid incidentalomas. Prevalence by palpation and ultrasonography: Arch Int Med 22: 1838-1840.
4. McCaffrey TV, 2000 Evaluation of the thyroid nodule: Cancer Control 7: 223-228.
5. Miki H, Oshimo K, Inoue H, et al, 1993 Incidence of ultrasonographically detected thyroid nodules in healthy adults: Tokushima J Exp Med 40: 43-46.
6. Castro MR, Gharib H, 2000 Thyroid nodules and cancer. When to wait and watch, when to refer. Postgrad Med 107:113-116, 119-120, 123-124.
7. Burrow NG, The thyroid nodules and neoplasia. In: Felig P, Baxter J, Broadus A, Frohman L (eds) Endocrinology and Metabolism 2nd edition: McGraw Hill Book Company: Inc, pp, 473-504.
8. Garber J, 2006 Thyroid Nodules 2006: Managing what has been known for over 50 years. Hormones (Athens) 5: 179-186.
9. Danese D, Sciachitano S, 1993 The thyroid nodule. Diagnostic considerations: Minerva Endocrinol 18: 129-137.
10. Amrikachi M, Ramzy I, Rubenfeld S, Wheeler TM, 2001 Accuracy of fine needle aspiration of thyroid:ArchPathol Lab med 125: 484-488.
11. Tabaqchali MA, Hanson JM, Johnson SJ, Wadehra V, Lennard TW, Proud G, 2000 Thyroid aspiration cytology in Newcastle: a six-year cytology/histology correlation study: Ann R Coll Surg Engl 82: 149-155.
12. Varverakis E, Neonakis E, 2002 Contribution of high-resolution ultrasonography in the differential diagnosis of benign from malignant thyroid nodules. Hormones (Athens) 1: 51-56.
13. Haber RS, 2000 Role of ultrasonography in the diagnosis and management of thyroid cancer: Endocr Pract 6: 396-400.
14. Leenhard L, 1993 Comments on the ultrasonographic evaluation of thyroid nodules: Ann Endocrinol Paris 54: 237-240.
15. Giammanco M, Digesu G, Massenti MF, Di Trapani B, Vetri G, 2002 Role of color flow Doppler sonography in preoperative diagnostics of the thyroid pathology. Minerva Endocrinol 27: 1-10.
16. Holden A, 1995 The role of colour and duplex Doppler ultrasound in the assessment of thyroidnodules. Australas Radiol 39: 343-349.
17. Shiamamoto K, Endo T, Ishigaki T, Sakuma S, Makino N, 1993 Thyroid nodules: evaluation with color Doppler ultrasonography. J Ultrasound Med 12: 673-678.
18. Clark KJ, Cronan JJ, Scola FH, 1995 Color Doppler sonography: anatomic and physiologic assessment of the thyroid. J Clin Ultrasound 23: 215-223.
19. Frates MC, Benson CB, Doubilet PM, Cibas ES, Marqusee E, 2003 Can color Doppler sonography aid in the prediction of Malignancy of Thyroid Nodules. J Ultrasound Med 22: 127-131.
20. Mesina G, Viceconti N, Trinti B, 1996 Echotomography and color Doppler in the diagnosis of thyroid carcinoma. Ann Ital Med Int 11: 263-267.
21. Papini E, Guglielmi R, Biancini A, et al, 2002 Risk of malignancy in non-palpable thyroid nodules: predictive value of ultrasound and color-Doppler features. J Clin Endocrinol Metab 87: 1941-1946.
22. Rago T, Vitti P, Chiovato L, et al, 1998 Role of conventional ultrasonography and colorflow-Doppler sonography in predicting malignancy in cold thyroid nodules. Eur J Endocrinol 138: 41-46.
23. Argalia G, D’ Ambrosio F, Lucarelli F, et al, 1995 Echo Doppler in characterization of thyroid nodular disease. Radiol Med (Torino) 89: 651-657.
24. Solbiati L, Cioffi V, Ballarati E, 1992 Ultrasonography of the neck. Radiol Clin North Am 30: 941-954.
25. Tanaka S, Kitamra T, Fujita M, Yoshioka F, 1998 Value of contrast-enhanced color Doppler sonography in diagnosing hepatocellular carcinoma with special attention to the color-filled patern. J ClinUltrasound 26: 207-212.
26. Sohn C, Beldemann F, Bastert G, 1997 Sonographic blood flow measurements in malignant breast tumors. A potential new prognostic factor. Surg Endosc 11: 957-960.
27. Schick S, Steiner E, Gahleitner A, et al, 1998 Differentiation of benign and malignant tumors of the parotidgland: value of pulsed Doppler and color Doppler sonography. Eur Radiol 8: 1462-1467.
28. Kidron D, Berneim J, Aviram R, et al, 1999 Resistance to blood flow in ovarian tumors: correlation between resistance index and histological patern of vascularization. Ultrasound Obstet Gynecol 13: 425-430.
29. Martinoli C, Pretolesi F, Crespi G, et al, 1998 Power Doppler sonography: clinical applications. Eur J Radiol 27: Suppl 2: 133-140.
30. Erdogan MF, Anil C, Ozer D, Kamel N, Erdogan G, 2003 Is it useful to routinely biopsy hot nodules in iodine deficient areas? J Endocrinol Invest 26: 128-131.
31. Spiezia S, Cerbone G, Colao A, Assanti AP, Picone GM, Lombardi G, 1997 Usefulness of power Doppler in the diagnostic management of hypoechoic thyroid nodules. Eur J Ultrasound 6: 165-170.
Address for correspondence:
Varverakis Emmanuel, 21-25 K. Palama Str., 73100 Chania
(Crete) Greece, Tel.+30 2821052052 Fax +30 2821052752,
e-mail: manvar@cha.forthnet.gr
Received 24-07-06, Revised 10-11-06, Accepted 25-11-06