1Research Institute for Pituitary Disease, Southern Tohoku General Hospital, Koriyama, Japan, 2Division of Nephrology, Endocrinology, and Vascular Medicine, Tohoku University Graduate School of Medicine, Aoba-ku, Sendai, Japan
Objective: We evaluated the long-term effects of growth hormone (GH) on
markers of quality of life, glucose metabolism, and lipid metabolism to
validate the adequacy of long-term GH replacement therapy for adult GH
deficiency (AGHD).
Design:
Eighty-three of 100 sequentially followed patients who received GH therapy were
selected for this study. Forty-nine were men aged 26 to 78 years (mean, 52
years) and 34 were women aged 20 to 78 years (mean, 56 years). The GH-releasing
peptide-2 stimulation test and arginine stimulation test were used to diagnose
AGHD. The adult hypopituitarism questionnaire (AHQ) and biochemical parameters
such as cholesterol (TC), triglyceride (TG), high-density lipoprotein (HDL)
cholesterol and low-density lipoprotein (LDL) cholesterol, and gyrated
hemoglobin (HbA1c) were determined before treatment, at 6 months of treatment,
and at 1, 2, 3, 4, 5, 6, 7, and 8 years of treatment. Considering age and sex
as factors potentially influencing the effect of GH therapy, the patients were
divided into age groups of <60 and ≥60 years and sex groups of men and women.
Repeated measured analysis of variance (ANOVA) was employed.
Results: ANOVA demonstrated significant
changes in mean AHQ scores during follow-up. Comparison of individual AHQ
scores with baseline values revealed sequential improvements, stabilization,
and decline in QOL. A significant elevation in HbA1c level was demonstrated.
LDL-C and HDL-C levels changed significantly upon GH treatment regardless of
sex or age. Levels of glucose, TC or TG did not change significantly.
Conclusion: The effect of GH therapy on
QOL showed sequential improvements and stabilization until 6-year follow-up.
Growth hormone deficiency, Growth hormone therapy, Long-term follow-up, Quality of life, Pituitary
INTRODUCTION
Many of the functions of growth hormone (GH) in humans are not known. It is involved in the regulation of body fluids, metabolism of sugars and fats, and possibly heart function. GH promotes lipid use by promoting triglyceride (TG) breakdown and oxidation in cells. It is involved in the regulation of blood glucose. GH suppresses the ability of insulin to promote glucose uptake in peripheral tissues. Benefits of GH supplementation for the treatment of GH deficiency include reduced fat mass, increased lean mass, increased bone density, improved lipid profile, and reduction in the risk of cardiovascular disease.1 These risk factors are favorably influenced by GH replacement therapy.
Adults with GH deficiency complain frequently of low energy levels, emotional lability, and mental fatigue that result in a low perceived quality of life (QOL). Adults with GH deficiency often have a higher prevalence of depression than adults not suffering from GH deficiency.2 GH replacement has been proposed as a treatment for depression as a result of GH deficiency, but the long-term effects of such therapy are not known.2
Hypomyelination in a mouse model of GH deficiency has been found to be due to arrested proliferation of glial cells and to be reversed by GH replacement.3 Thus, the GH-insulin-like growth factor 1 (IGF-1) axis is involved in regulation of the growth, development, and myelination of the brain.4 In addition, GH and IGF-1 affect cognition and biochemistry in the adult brain.5 Accordingly, accumulating evidence suggests that GH is closely associated with neuroprotection, regeneration, and functional plasticity in the adult brain.
However, concerns over the effectiveness of GH treatment in adults have been raised in the context of short-term follow-up.6
Only a few reports have addressed the beneficial effects of long-term GH treatment for adult GH deficiency (AGHD), particularly with regard to patient age and sex.7,8 Claessen et al8 reported the metabolic profiles after a minimum of 10 years of GH treatment in 98 patients with AGHD. They found that the prevalence of metabolic syndrome increased significantly during GH treatment, although an improvement in several cardiovascular risk factors was observed. Bunderen et al7 studied how GH treatment influences mortality and found that the mortality rate of patients with AGHD treated with GH supplementation did not differ from the control population regardless of sex. On the other hand, Gaillard et al1 reported a modest increase in mortality in patients with hypopituitarism who received GH replacement.
We wished to evaluate the long-term QOL for GH treatment of AGHD in addition to carrying out measurement of biochemical markers because these factors may influence mortality and development of the metabolic syndrome.
MATERIALS AND METHODS
The effectiveness of GH treatment is recognized in Japan and GH therapy was approved by the National Health Insurance Policy in 2006. In October 2009, hypopituitarism was included in the Research Project on Overcoming Intractable Diseases, which allows affected patients to receive financial assistance from the Department of Health and Welfare. Since then, this therapy has become much more readily available. Under these circumstances, 100 of 2000 patients who underwent pituitary surgery by one of the authors (H.I.) received GH therapy for 8 years.
Data were missing for 6 patients due to aggravation of diabetes mellitus, 6 patients who were victims of the tsunami in northern Japan in 2011, 2 patients with recurrence of brain tumors, and 2 patients who had moved to facilities in which self-injection was difficult. One patient was lost to follow-up.
Except for these 17 patients, the remaining 83 patients (94%) continued GH treatment irrespective of its efficacy. However, data for 12 out of the 83 patients were not measured at some time points, consequently statistical analyses were not carried out in these patients. Therefore, physical-examination and complete follow-up data were available for 71 patients. Five patients were followed up for 8 years, 6 patients for 7 years, 8 patients for 6 years, 23 patients for 5 years, 42 patients for 4 years, 45 patients for 3 years, 51 patients for 2 years, 64 patients for 1 year, and 71 patients for 6 months. We continued GH replacement therapy irrespective of whether GH administration was efficacious or not. As a result, data were missing for only 6% of participants and there was thus little risk of selection bias. Forty-nine patients were men aged 26 to 78 years (mean, 52 years) and 34 were women aged 20 to 78 years (mean, 56 years). Fifty-five patients had pituitary adenomas, 13 had craniopharyngiomas, 13 had Rathke’s cleft cysts, 1 had a meningioma, and 1 had hypophysitis. The backgrounds of the patients are summarized in Table 1.
The research protocol was approved by our institutional review board. The study was approved by the appropriate ethics committee and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All patients provided written informed consent.
DEFINITION OF GROWTH HORMONE DEFICIENCY
Prior to treatment of pituitary diseases, evaluation for the presence of growth hormone deficiency (GHD) must be performed in all patients in whom surgery is indicated. A low reserve of GH secretion is detected using GH secretory stimulation tests, namely, the insulin tolerance test, arginine tolerance test, L-dopa tolerance test, glucagon tolerance test, and GH-releasing peptide-2 (GHRP-2) tolerance test. In this study, the GHRP-2 tolerance test was performed first, followed by the arginine tolerance test if a second test was deemed necessary. For the arginine tolerance test, 30 g of arginine hydrochloride in 300 mL of normal saline were infused intravenously over 30 minutes and blood samples were collected before and at 30, 60, 90, and 120 minutes after infusion for determination of serum GH levels. For the GHRP-2 stimulation test, blood samples were collected before and 15, 30, 45, and 60 minutes after intravenous injection of 100 µg of pralmorelin hydrochloride. AGHD was defined as a peak GH level of <1.8 ng/mL for the arginine test and <9 ng/mL for the GHRP-2 stimulation test.
QOL measurement
Symptoms of GHD were considered to be fatigue, a low energy level, low concentration levels, decreased vigor, depression, and low sexual desire. Signs associated with AGHD were considered to be dry and thinning skin, softened body hair, increased body fat (fat on the internal organs), an increased waist/hip ratio, a decrease in lean body weight, and decreased bone mass and muscular strength. Generic or disease-specific QOL scales can be used to measure QOL in patients with hypopituitarism: Nottingham Health Profile; Psychological General Well Being Index; MOS 36 Item Short Form Health Survey (SF-36); Quality of Life-Assessment of Growth Hormone Deficiency in Adults (QoL-AGHDA); Questions on Life Satisfaction (QLS).
The adult hypopituitarism questionnaire (AHQ) was developed based on two domains: psychosocial and physical. The psychosocial domain was divided into six subdomains. The physical domain was divided into seven subdomains. Cronbach’s alpha coefficient was 0.72-0.93 for the psychosocial subdomain and 0.73-0.89 for the physical subdomain, denoting acceptable internal consistency. With regard to reproducibility, the intraclass correlation coefficient was 0.77-0.90 for psychosocial subdomains and 0.86-0.94 for physical subdomains, and reproducibility was considered sufficient. AHQ subdomains correlated moderately with all of SF-36v2 subdomains (0.13-0.68).9 Since the AHQ addresses all of these signs and symptoms of AGHD and has been shown to be suitable for Japanese patients, this questionnaire was used for evaluation.9 The same questionnaire was repeatedly used before treatment, at 6 months of treatment, and at 1, 2, 3, 4, 5, 6, 7, and 8 years of treatment.
Biochemical parameters
To clarify the effect of long-term GH replacement on the metabolism of lipids and sugar and cardiovascular risk factors, we selected biochemical markers: fasting serum total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL)-cholesterol, low-density lipoprotein (LDL)-cholesterol, glucose, and glycated hemoglobin (HbA1c). These biochemical parameters were measured before treatment, at 6 months of treatment, and at 1, 2, 3, 4, 5, 6, and 7 years of treatment; the levels of these values were assessed to determine whether GH therapy had significant effects on lipid and glucose metabolism. The sequential observation period for each parameter lasted from 6 months to 7 years (mean, 5 years).
Method of GH administration
GH was administered daily as an intradermal injection before sleep at an initial dose of 3 µg/kg body weight. Unless there were clear adverse effects, the dose was increased every 4 weeks with the goal of maintaining insulin-like growth factor 1 (IGF-1) levels within the expected ranges for age and sex.
Comparison of groups by age and sex
Most patients were in their fifties, followed by patients in their sixties; the mean age of men was 52 years and that of women was 56 years. In light of these findings, it seemed appropriate to divide the patients into two age groups with a cutoff of 60 years. We compared the effect of GH treatment between the following groups of patients: men and women aged <60 years and men and women aged >60 years.
Statistical analysis
For comparisons within groups, repeated measures analyses (ANOVA) using StatView v4.0 was used for continuous data to compare differences in parameters before and after GH treatment. If significant differences were detected, Wilcoxon signed rank analyses were carried out. The difference in QOL score from baseline to each post-baseline period (6 months to 8 years) at each time point was tested using Wilcoxon signed rank analyses. The Kruskal-Wallis test was used to evaluate the differences in background factors (Table 1) among the four groups divided by age and sex. Welch’s t-test was used to compare the differences of two independent groups. A p value of <0.05 was taken to be statistically significant.
RESULTS
Table 1 shows a summary of the demographic and clinical characteristics of the patients. The Kruskal-Wallis test revealed no significant difference between the two groups in background factors such as body mass index (BMI) and period of time between the diagnosis of GHD and introduction of GH therapy. Age at diagnosis of GDH was not significantly different between men and women (p=0.18).
No significant differences in either weight or height were observed between the two male age groups or between the two female age groups. Considering the serum IGF-1 SD scores, the dose of GH administered was ideal until 6 months after GH supplementation. Sequential changes in the hormone supplementation dose were observed only for thyroxin supplementation. The daily dose of thyroxin had to be increased after 6 months of GH supplementation therapy in 8 of the 18 men aged <60 years (44%), 6 of the11 men aged <60 years (55%), 5 of the 17 women aged <60 years (29%), and 8 of the 13 women aged <60 years (62%).
Sequential changes in the IGF-1 concentration and IGF-1 SD score among the four groups are shown in Table 2. Both the IGF-1 concentration and IGF-1 SD score were significantly higher than the pretreatment values and remained high in each group (Table 2).
Evaluation of the AHQ
Sequential changes in QOL and biochemical parameters upon GH supplementation varied widely among patients. Psychosocial AHQ scores changed significantly upon GH treatment at 6 months as well as 1-, 2-, 3-, 4-, 5-, 6-, 7-, and 8-year time points by ANOVA (Figure 1). A significant difference in the psychosocial domain between males and females at 6- and 7-year time points was noted (Figure 2). ANOVA also demonstrated significant changes in mean values of physical AHQ scores at 6 months as well as 1-, 2-, 3-, 4-, 5-, 6-, and 7-year time points (Figure 3). However, there were no significant differences in the physical AHQ domain with respect to age and sex.
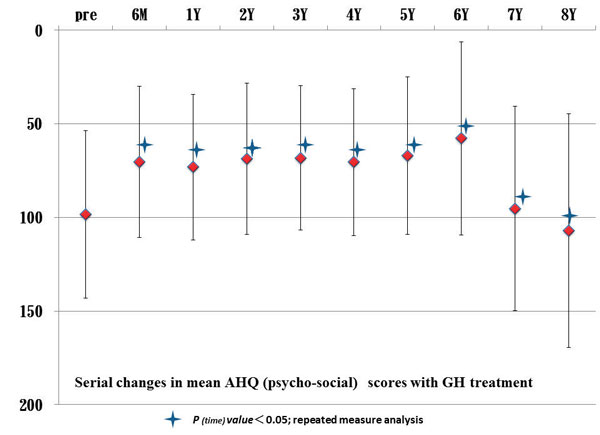
Figure 1. Sequential changes in mean AHQ (psychosocial) scores upon GH treatment.

Figure 2. Sequential changes in mean AHQ (psychosocial) scores upon GH treatment in males and females.
Comparison of individual AHQ values with baseline values revealed sequential improvements, stabilization, and decline in QOL. Sequential changes in mean psychosocial AHQ values revealed a decline at 7-year follow-up (Figure 1). Physical AHQ values also revealed a decline at 6-year follow-up (Figure 3). In this context, mean values of psychosocial and physical scores tended to become lower than baseline values at 7-8-year follow-up.

Figure 3. Sequential changes in mean AHQ (physical) scores upon GH treatment.
Evaluation of metabolic markers
ANOVA demonstrated significant changes in mean values of HbA1C at 1, 2, 3, 4, and 5 years (Figure 4). Comparison of baseline values of HbA1C with sequentially measured mean values of HbA1C showed that HbA1c levels increased significantly from 6-month to 6-year follow-up (Table 3). ANOVA demonstrated significant changes in mean values of HDL-C at 1, 2, 3, 4, and 5 years (Figure 5). Comparison of baseline values of HDL-C with sequentially measured mean values of HDL-C revealed that HDL-C levels increased significantly from 4-year to 6-year follow-up (Table 3).
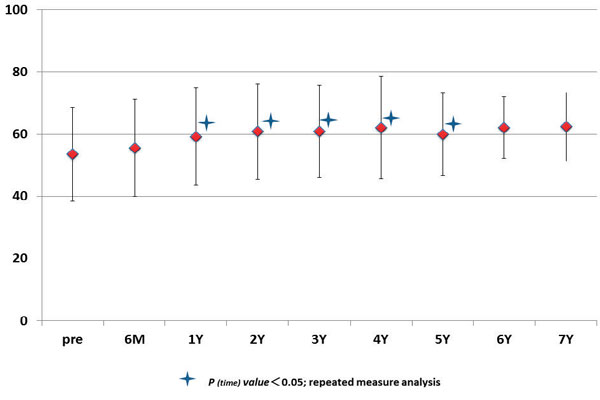
Figure 4. Sequential changes in mean levels of HbA1C in serum upon GH treatment.
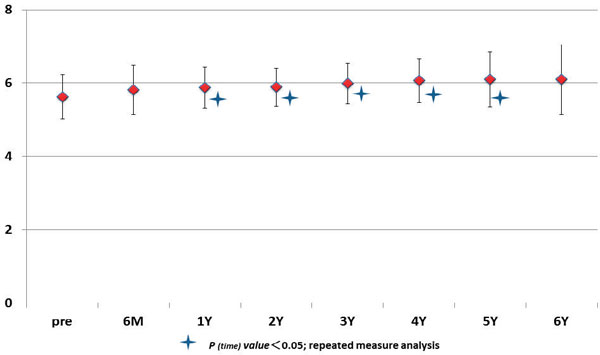
Figure 5. Sequential changes in mean levels of HDL-cholesterol in serum upon GH treatment.
Significant changes in mean values of LDL-C were observed at 3, 4, 5, and 6 years (Figure 6). Comparison of baseline values of LDL-C with sequentially measured mean values of LDL-C revealed that LDL-C levels decreased significantly from 1-year to 4-year follow-up (Table 3). There were no significant changes in mean values of TG, TC or glucose during follow-up. Changes in serum glucose were not related to changes in the duration of GH treatment at any time point (3 years, p=0.29; 4 years, p=0.43; 5 years, p=0.79; 6 years, p=0.69). Changes in serum TG were not related to changes in the duration of GH treatment at any time point (3 years, p=0.36; 4 years, p=0.50; 5 years, p=0.62; 6 years, p=0.32). Changes in TC were not related to changes in the duration of GH treatment except at 4 years (1 year, p=0.88; 2 years, p=0.58; 3 years, p=0.21; 4 years, p=0.04; 5 years, p=0.42; 6 years, p=0.36). The age and sex of patients had no significant effect on their levels of metabolic markers.
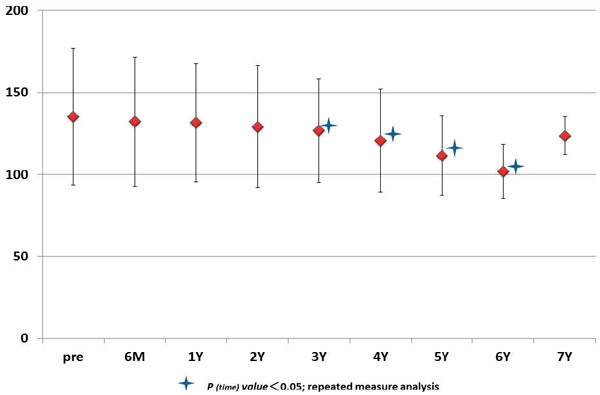
Figure 6. Sequential changes in mean levels of LDL-cholesterol in serum upon GH treatment.
DISCUSSION
AGHD adversely affects patients’ cardiovascular risk profile, bone density, and QOL. GH replacement, when dose-titrated, is well tolerated and has been shown to have beneficial effects on patients’ body composition, lipid profile, bone mineral density, and psychological well-being.10 Patients with AGHD on 5-year GH replacement appear to demonstrate mortality rates similar to those of country-specific background populations, in contrast to a previous report.11
Patients with AGHD have also been demonstrated to have lower QOL than control individuals. The Nottingham Health Profile, Psychological General Well-Being Index, MOS 36-Item SF-36, QoL-AGHDA, and QLS-hypopituitarism (QLS-H) have been used in European countries and the United States to evaluate QOL. Because the NHP and PGWB Index are not disease-specific but are instead used for many types of diseases without focusing on specific characteristic problems occurring in patients with GHD, these questionnaires lack sensitivity in evaluation of such patients. Although previous reports have indicated that QOL improvement by GH administration can be evaluated by the QoL-AGHDA or OLS-H in patients from both Europe and the United States, these tools are not suitable for QOL evaluation in Japanese patients with AGHD because of the absence of a large difference between GH-treated and placebo-treated control groups. Thus, the Japanese AHQ was developed to assess QOL parameters for Japanese patients with hypopituitarism.9
When the effects of GH administration data were studied, QOL appeared to improve during long-term GH replacement, although the majority of the beneficial effects on QOL were typically seen within the first year of treatment.12 Svenson et al13 and Holdaway et al,14 using a follow-up of 3 years, reported improvement in QOL. Wiren et al,15 using a follow-up of 20-50 months, reported that the QOL score improved significantly compared with the baseline score. Jorgensen et al16 reported that QOL improvement was significant in the early years for most subgroups but that some of the observed improvement 6-10 years later did not reach significance (which may have been because of missing data). The reason why some results did not reach significance is because of the small sample size in later years and because in that observational study data were gathered from several countries.17 A limitation of this type of observational study is that patients unresponsive to GH therapy are lost to the study spontaneously.17
In our study, ANOVA demonstrated significant changes in mean values of AHQ scores (psychosocial domain) at 6 months as well as at 1, 2, 3, 4, 5, 6, 7, and 8 years. A significant difference in the psychosocial domain was noted between males and females at 6 and 7 years. ANOVA also demonstrated significant changes in mean values of AHQ scores (physical domain) at 6 months as well as at 1, 2, 3, 4, 5, 6, and 7 years. Sequential changes in mean AHQ (psychosocial) values declined at 7-year follow-up and the AHQ (physical) value also declined at 6-year follow up. Mean values of psychosocial and physical scores became lower than baseline values at 7-8-year follow-up, a finding that is consistent with the report of Jorgensen et al.16
Comparison of individual AHQ scores with baseline values showed sequential improvements, stabilization, and decline in QOL. Ten-year follow-up data gathered from Europe and the USA by Mo et al17 revealed that QOL improvements remained significant throughout that decade. In that study17 various physicians attended to patients, which may have led to biases in study results (e.g., selection bias, unresponsive patients lost to the study). Owing to these selection biases, the trend of decline in QOL may have disappeared from their data. Conversely, our study was carried out by only one physician, which resulted in identical treatment and follow-up for all patients. In addition, we continued GH replacement irrespective of whether GH administration was efficacious or not. Data were missing for only 6% of our study cohort, thus there was little scope for selection bias. Hence, our study design was ideal for evaluation of the true efficacy of GH replacement compared with the large cooperative study by Mo et al.17 The score range of the psychosocial domain was 7 to 167 (range: 0-204) and that of the physical domain was 9 to 181 score (range: 0-252), so there were no peaks or troughs in values.
During long-term follow-up, some patients suffered from other disorders (e.g., femoral fracture, esophageal cancer, hemorrhagic colon, multiple diverticula), which led to a reduction in QOL. Moreover, patients who continued GH replacement for >5 years had a decline in QOL because of aging. These two effects could also have been responsible for the decline in QOL. QOL is reportedly strongly influenced by BMI; however, the BMI in our four groups of patients did not differ significantly according to the Kruskal-Wallis test.
The purpose of GH therapy is not only to improve QOL after it has been impaired by GH deficiency, but also to correct metabolic abnormalities such as increased body fat, abnormal body composition including a decrease in lean body weight, and abnormal blood lipid levels. Low-dose GH supplements therapy for 6 months in patients with AGHD significantly improved both the total and LDL-cholesterol levels compared with the pretreatment levels. Such improvements reportedly continue for 2 years on GH treatment.6 Therefore, we measured the fasting total cholesterol, triglyceride, HDL-cholesterol, and LDL-cholesterol levels before and sequentially during treatment.
There are controversial publications concerning the effect of GH supplementation on various factors such as QOL, mortality, lipid metabolism, and glucose metabolism. There are conflicting reports from Europe and the United States regarding diabetic patients. One study recommended not to treat diabetic patients with GH and another concluded that GH treatment posed no risk of increasing either the glucose or HbA1c level.18,19 In our study, significant changes in mean values of HbA1C were demonstrated at 1, 2, 3, 4, and 5 years (ANOVA) and there were no significant changes in glucose levels during long-term GH treatment.
Holdaway et al,14 using a follow-up of 3 years, reported no significant changes in levels of TC, LDL-C, TG, fasting blood sugar or HbA1C. Jorgensen et al,16 using a follow-up of 42 months, reported significant changes in levels of LDL-C and HDL-C, but not in levels of TG, HbA1C, TC or glucose. Our study demonstrated significant changes in mean levels of HDL-C at 1, 2, 3, 4, and 5 years (ANOVA), a result that is in accordance with that of Jorgensen et al.16 Significant changes in mean levels of LDL-C were noted at 3, 4, 5, and 6 years (ANOVA), a result that is in accordance with that of Jorgensen et al.16 There were no significant changes during follow-up in levels of TG or TC, a result that is in accordance with that of Holdaway et al.14
For appropriate evaluation of GH therapy, further studies involving more subjects who are evaluated for an extended period of time are necessary.
CONCLUSION
In patients with AGHD, long-term GH administration showed sequential improvements, stabilization, and decline in QOL. GH therapy elicited significant changes in the psychosocial domain of AHQ until 8-year follow-up. GH therapy elicited significant changes in the physical domain of AHQ until 7-year follow-up. Levels of biochemical markers such as HbA1C, HDL-C, and LDL-C showed significant changes during long-term follow-up.
REFERENCES
1. Gaillard RC, Mattsson AF, Akerblad AC, et al, 2012 Overall and cause-specific mortality in GH-deficient adults on GH replacement. Eur J Endocrinol 166: 1069-1077.
2. Prodam F, Caputo M, Belcastro S, et al, 2012 Quality of life, mood disturbances and psychological parameters in adult patients with GH deficiency. Panminerva Med 54: 323-331.
3. Lobie PE, Zhu T, Graichen R, Goh LK, 2000 Growth hormone, IGF-1 and the CNS: localization, function and mechanism of action. Growth Horm IGF Res 10: uppl B: 51-56.
4. Isgaard J, Aberg D, Nilsson M, 2007 Protective and regenerative effects of the GH/IGF-1 axis on the brain. Minerva Endocrinol 32: 103-113.
5. Aberg, ND, Brywe KG, Isgaard J, 2006 Aspects of GH and insulin-like growth factor-1 related to neuroprotection, regeneration, and functional plasticity in the adult brain. SientificWorldJournal 6: 53-80.
6. Florakis D, Hung V, Kaltsas G, et al, 2000 Sustained reduction in circulating cholesterol in adult hypopituitary patients given low dose titrated growth hormone replacement therapy; a two year study. Clin Endocrinol 53: 453-459.
7. Bunderen CC, Nieuwpoort C, Arwert LI, et al, 2011 Dose growth hormone replacement therapy reduce mortality in adults with GHD? Data from the Dutch national registry of GH treatment in Adults. JCEM 96: 3151-3159.
8. Claessen KM, Appelman-Dijkstra, Adoptie DMM, et al, 2013 Metabolic profile in GHD adults after long-term recombinant Human GD therapy. JCEM 91: 352-361.
9. Hitoshi Ishii, Akira Shimatsu, Yasuhiko Okimura, et al, 2012Development and validation of a new questionnaire assessing quality of life in adults with hypopituitarism: adult hypopituitarism questionnaire (AHQ) PLoS One. 7: e44304.
10. Jorgensen JOL, Christiansen JS 2005 Growth Hormone Deficiency in Adults. In Grossman AB, (ed) Frontiers of Hormone Research, Baser, Karger; pp, 209-221.
11. Wilton P, Koppeschaar HPF 2000 Safety of growth hormone replacement in adukts. In Monson JP, Bengtsson BA (eds), GH replacement in Adults-The First 5 years of KIMS. Oxford, OxfordPhrma Genesis.
12. Appelman-Dijkstra N, Claessen KM, Roelfsema F, Pereira A, Biermasz NR, 2013 Therapy of endocrine disease; Long-term effects of recombinant human GH replacement in adults with GH deficiency; a systematic review. Eur J endocrinol 169: R1-R14.
13. Svensson J, Mattsson A, Rosen T, et al, 2004 Three-years of growth hormone replacement therapy in GH-deficient adults: effects on quality of life, patient-reported outcomes and healthcare consumption. Growth Horm IGF Res 14: 207-215.
14. Holdaway IM, Manning HP, Cutfields W, et al, 2015 Three-year experience with access to nationally funded growth hormone (GH) replacement for GH-deficient adults. Clin Endocrinol 83: 85-90.
15. Wiren L, Bengtsson BA, Johannsson G, 1988 Beneficial effects of long-term GH replacement therapy on quality of life in adults with GH deficiency. Clin Endocrinol 48: 613-620.
16. Jorgensen AP, Fougner KJ, Ueland T, et al, 2011 Favorable long-term effect of growth hormone replacement therapy on quality of life, bone metabolism, body composition and lipid lebels in patients with adult-onset GH deficiency. Growth Horm IGF Res 21: 69-75.
17. Mo D, Blum WF, Rosilio M, Webb SM, Qi R, Strasburger CJ, 2014 Ten-year change in quality of life in adults on growth hormone replacement for GH deficiency: an analysis of the hypopituitary control and complication study. J Clin Endocrinol Metab 99: 4581-4588.
18. Cutfield WS, Wilton P, Bennmarker H, et al, 2000 Incidence of diabetes mellitus and impared glucose tolerance in children and adolescents receiving GH treatment. Lancet 355: 610-613.
19. Svensson J, Fowelin J, Landin K, Bengtsson BA, Johansson J, 2002 Effects of seven years of GH-replacement therapy on insulin sensitivity in GH-deficient adults. JCEM 7: 2121-2127.
Address for correspondence:
Hidetoshi Ikeda, Masataka Kudo;
Research Institute for Pituitary Disease, Southern Tohoku General Hospital, 7-115 Yatsuyamada, Koriyama 963-8563, Japan; Tel.: +81-24-934-5322, Fax:
+81-24-922-5320, E-mail: ikeda@nsg.med.tohoku.ac.jp
Received: 06-08-2015, Accepted: 30-10-2015