1Department of Endocrinology, Athens Naval & VA Hospital, Athens; 2Unit of Reproductive Endocrinology, First Department of Obstetrics and Gynaecology, Medical School, Aristotle University of Thessaloniki, Thessaloniki; Greece
Despite the variety of available female contraceptive methods, many pregnancies (~50%) are still undesired. Many men (>60%) want to participate equally with their partner in family planning; however, male contraceptive methods (MCMs) account for only 14% of those used worldwide and no pharmaceutical MCM is available so far. The only two MCMs currently available are condoms, which despite protecting against sexually transmitted diseases have high failure rates (~19%), and vasectomy, which though very efficient (99%) is poorly reversible (<50%). Among MCMs under investigation, male hormonal contraceptives (MHCs) are those that have come closest to commercialization. The action of MHCs relies on the disruption of spermatogenesis that exogenous androgen administration evokes by suppressing the hypophyseal-gonadal axis. Various regimens of androgens as monotherapy or in combination with progestins have been tested in clinical trials achieving a Pearl Index <1.0 (equal to that of the female oral contraceptive pill); however, concerns regarding the variable response rates observed (non-responders: 5-20%), the impracticality of parenteral administration and long-term prostate-associated or cardiovascular morbidity have deflected the interest of the pharmaceutical industry from further research. Non-hormonal contraception methods may be, at least theoretically, more specific by selectively disrupting spermatogenesis and sperm transport or fertilizing ability. Nevertheless, only a few have been tested in clinical trials (Reversible Inhibition of Sperm Under Guidance, RISUG, and Intra Vas Plugs); most of them are still in pre-clinical development or have been abandoned due to toxicity (gossypol). Consequently, until a reliable, safe and practical MCM is developed, women will continue to bear most of the contraception burden.
Male contraception, RISUG, Testosterone, Vasectomy
INTRODUCTION
It has been shown historically that uncontrolled population growth results in inadequate distribution of wealth and a failure to improve living standards in societies.1 However, even in our “post-Oral Contraceptive Pill” era, a large percentage of births remain undesirable. According to US statistics, of 6 million pregnancies recorded annually, only 50% are desirable; of the latter, about 50% result in abortion.2 Modern women can choose among a spectrum of 11 different contraceptive methods, whereas for men essentially only two methods have been developed: the male condom and the dissection of the vas deferens (vasectomy).3 Despite the paucity of available options, male contraceptive methods account for about 14% of those used worldwide, while this percentage increases to 30% in developed societies (Figure 1).4

Figure 1. Use of existing male contraceptives in developed compared to developing regions. (Data from the United Nations Population Division World Contraceptive Use 2003).
Although female contraceptive methods are very effective, they are contraindicated in certain groups of women or have adverse effects that often lead to discontinuation. Notably, a recent study has shown that among minority couples in the US, adolescent mothers do not have an accurate perception of their partners’ pregnancy intentions and use contraceptive methods that are not within their control.5 On the other hand, many men want to participate equally with their partner in family planning. According to a recent study, more than 60% of men in Germany, Spain, Brazil and Mexico were willing to use a male contraceptive method. This option was more popular among men with higher income and higher education6 and was supported by 98% of women in long-term relationships.7
Unfortunately, and despite the favorable social conditions, the pharmaceutical industry has deflected its interest from research in male contraception methods (MCMs). The one and only phase II study funded by pharmaceutical companies was abandoned 7 years ago, despite its promising results.8 Currently, the funding of research programs for MCMs comes only from public programs, international organizations (WHO) and charities (Population Council, New York).
EXISTING METHODS OF CONTRACEPTION
The male contraceptive methods are classified into three main categories, based on their target of action:
1. Methods that hinder the transport of sperm in the female reproductive system
2. Methods that suppress spermatogenesis
3. Methods that disrupt the maturation or fertilizing ability of spermatozoa.
Both of the two existing male contraceptive methods (condom and vasectomy) belong to the first category; however, they fulfil only a minority of the characteristics that a male contraceptive method should ideally possess (Tables 1 & 2).
Condoms
The condom is the oldest method of contraception; however, it was not until 2004 that the WHO established the manufacturing specifications for condom production.9 Currently, 5.7% of couples worldwide use condoms as a contraceptive method, with the percentage being higher in developed countries: 20% in the US, 25% in Germany and 80% in Japan.10 A key advantage of the condom is the protection it offers against sexually transmitted diseases,11 whereas its main disadvantage is high failure rates, which in “real life” conditions concern up to 19% of couples during the first year of use.3 In general, the older the condom, the higher the risk of rupture.12
Vasectomy
Vasectomy became popular as a MCM in the 1960s, particularly in the US.13 Nowadays, vasectomy is selected by 2.7% of couples seeking contraception (10% in the U.S.), totalling over 40 million men worldwide.14 It is more popular among advanced societies and more highly educated men due mainly to cultural and religious restrictions.15 The operation procedure includes the dissection of the vas deferens followed by ligation and electrocoagulation of the two ends, a minimally invasive technique of high efficiency with failure rates lower than 1% and low complication rates.16 The successful outcome of the operation should be confirmed by obtaining a sperm analysis at least 3 months postoperatively. The main disadvantage is its poor reversibility, a fact that should be emphasized preoperatively to the patient and which renders this option suitable for men who do not intend to father children in the future.
Short-term complications include bleeding and hematomas, wound infection and epididymitis, at rates of 1-6%.14 A major long-term complication is procedure failure due to recanalization of the vas deferens (0-3% of cases).17 Moreover, the rise of intra-epididymal pressure as a result of the ligation of the distal end of the vas deferens may produce discomfort in the scrotum and can occasionally cause rupture of the epididymis and semen leakage, which in turn is associated with the formation of scrotal granulomas and the development of anti-sperm antibodies in the serum. Recently, concerns have been raised regarding an association of vasectomy with a modestly increased incidence of high-grade prostate cancer; however, a causal relation could not be established.18
The rate of operated men wishing to regain their fertility after vasectomy is at around 3.5% and is showing an increasing trend as a result of the changes taking place in modern societies (higher divorce rates, family planning variability).19 Modern microsurgical techniques of anastomosis achieve patency restoration rates of around 90%;20 however, pregnancy rates fluctuate significantly below these values at approximately 50%.21 This discrepancy may be attributed to the re-occlusion of the vas deferens (~3%) or the presence of anti-sperm antibodies (70%).22 The determining factor for the recovery of fertility is the time elapsed after the vasectomy, as chances of success are reduced after two years and practically eliminated after 10 years.23
HORMONAL CONTRACEPTION
The physiology of male hormonal contraception (MHC)
According to current knowledge, the pulsatile release of gonadotropin-releasing hormone (GnRH) from the hypothalamus stimulates the release of luteinizing hormone (LH) and follicle stimulating hormone (FSH) from the pituitary, which, in turn, stimulate Leydig cells to produce testosterone (T) and Sertoli cells to promote spermatogenesis, respectively. Conversely, circulating T regulates gonadotropin production by exerting an inhibitory effect at the level of the hypothalamus and pituitary gland in the setting of a feedback circuit.9 Consequently, the exogenous administration of T is expected to suppress the gonadal axis, thereby inhibiting endogenous T production and testicular spermatogenesis. These effects are completely reversible after the withdrawal of exogenous T.24 Because the human spermatogenesis cycle lasts approximately 72 days, there is a delay of 2-3 months after the initiation of hormonal contraceptives until they achieve their full contraceptive effect.
The ultimate goal of MHC is to reduce the number of sperm in the ejaculation so drastically that it is impossible to achieve fertilization. The ideal goal, therefore, is to achieve azoospermia. However, pregnancy rates as low as those attained with the female contraceptive pill may be achieved with a concentration of up to 1 million sperm/ml, which may be considered acceptable.25
Androgen monotherapy
The use of exogenous testosterone as a MCM was introduced in the 1970s; by the 1990s, the WHO had funded a number of clinical trials.
Testosterone enanthate
T enanthate (TE) was administered in two of the studies supported by the WHO. In the former, a multicenter study including 271 men of Asian and Caucasian origin, 200 mg TE were administered by weekly injections for 6 months.26 Seventy percent of the men achieved azoospermia. Only azoospermic subjects were selected to enter the efficacy phase of the study, which lasted for one year, and the result was a single pregnancy (1/157 men). The second study27 involved 357 couples; 200 mg TE were administered by weekly injections. Apart from azoospermia, severe oligozoospermia (<3 million/ml) was also acceptable for entry into the efficacy phase. As expected, failure rates in the oligozoospermic subjects were higher than in the azoospermic men, but clearly lower compared to those using only condoms.
Testosterone undecanoate
The first phase III study with T undecanoate (TU) involved 305 men of Chinese nationality who received 500 mg dissolved in tea seed oil by monthly intramuscular injections after a loading dose of 1000 mg.28 In 296 men (97%), severe oligozoospermia (<3 million/ml) was achieved; these men were selected to enter the 12-month efficacy phase. Semen recurrence was observed in six men and one pregnancy occurred (96.7% effectiveness). The reappearance of sperm (“sperm rebound”) might be attributed to the high peaks of circulating T levels after each injection.29 This study was followed by a larger one, also in China, during which 500 mg TU were administered intramuscularly in 1000 men on a monthly basis.30 In this study, 855 men entered the effectiveness phase, during which nine pregnancies were recorded. In Europe, TU formulation is dissolved in castor oil, which augments its bioavailability and allows delivery at 12-week intervals. However, administration of TU in Caucasian men every 6 weeks achieved azoospermia in only 60% of them.31 The large intervals between administrations render the option of TU attractive, although the large amount of excipient needed (~4.0 ml) makes the administration more painful.
Alternative testosterone formulations
Τ buciclate (TB) was developed in the 1970s via the collaboration of the WHO with the National Health Institute (NIH) in an effort to develop a form of T with a prolonged time of action. The half-life of TB is 29.5 days compared to 4.5 days of TE, while the total duration of action is 3-4 months32. A single intramuscular injection of TB can cause suppression of spermatogenesis comparable to that of weekly injections of TE;33 however, this substance has not attracted any commercial interest as yet.
The administration of T in the form of subcutaneous implants can provide stable levels of circulating T for many months, avoiding the spikes observed with injectable testosterone formulations, a possible cause of the “sperm rebound” phenomenon.29 A single application of implants totalling a dose of 1200 mg has an efficacy equal to that of weekly administration of TE34 and can be combined with a progestin for higher security. Their placement can be applied on an outpatient basis through a small incision of the skin. A disadvantage is the possibility of an automatic rejection, a complication, which occurs at a rate of 3-7%.35
The oral administration of T has not proven to be effective as a method of male contraception, since the circulating T levels are neither constant nor sufficient to completely suppress the secretion of gonadotropins. Oral administration of 240 mg TU once daily for 12 weeks resulted in adequate contraception in only one out of seven volunteers. More frequent administration (QID)36 or the co-administration of T with 5α-reductase inhibitors in an oily solution37 achieved more stable concentrations of circulating T but did not improve the regimen’s efficacy. Regarding the transdermal route of administration, the efficacy of patches as MHC was poor whether as monotherapy or in combination with progestins, while local irritation upon application was a common drawback.38
Adverse effects
The fact that supernormal T levels are necessary to achieve the desirable suppression of spermatogenesis has raised concern over the long-term effects of contraceptive treatment on men’s health, especially as concerns the cardiovascular system and prostate-associated morbidity.
So far there are no data to directly correlate the administration of androgens with increased cardiovascular events. A recent study involving T administration in cardiovascular compromised patients has been strongly criticized for methodological problems.39 Exogenous administration of androgens in young healthy males correlates with weight gain, which mainly concerns fat free body mass,40 and decreases high-density (HDL) cholesterol. However, a parallel decrease in the concentration of total and low-density (LDL) cholesterol, which is mainly considered as atherogenic, is observed, ameliorating at least theoretically the cardiovascular risk.41
Data that correlate high levels of T to the development of prostate malignancy are conflicting.42 Moreover, existing clinical trials on male contraceptives generally concern young people and are too short in length to allow reliable conclusions. Nevertheless, given the large male population which will be affected and the long-term effects anticipated by the commercial use of a contraceptive method, the dose of administered androgens should be reduced to the minimum effective for achieving contraception.43 A less severe side effect is the development of acne, whereas sexual function and overall satisfaction seem to slightly improve during androgen administration.44
Additional concerns have been raised regarding the reversibility of MHC. However, no single case of permanent infertility as a result of MHC has been reported in any clinical study. In a recent systematic review, which included 30 studies with a total of 1549 men, recovery of spermatogenesis was observed in all participants within a median time of 3-4 months.45 The longer duration of androgen administration and the use of formulations with sustained action were associated with longer recovery time.
Combination of androgens with GnRH-analogues
The administration of GnRH-agonists, after an initial phase of stimulation (flare-up), eventually suppresses gonadotropin secretion by reducing GnRH-receptors on the pituitary gland (down-regulation). On the other hand, in studies in which GnRH-agonists were co-administered with T as a means of MHC, adequate suppression of spermatogenesis could not be achieved, probably because of the lack of full FSH suppression.46
Unlike agonists, GnRH-antagonists can cause complete suppression of both LH and FSH, which is achieved within a few hours, avoiding the phenomenon of flare-up.46 Thus, co-administration of the GnRH-antagonist Nal-Glu with TE for 12 weeks caused azoospermia in 88% of the men and this favorable effect was maintained for an additional 20 weeks of monotherapy with TE.47 Accordingly, high efficiency was observed by the addition of daily injections of cetrorelix or abarelix to androgen contraceptive regimens,48 while the antagonist acyclin can suppress gonadotropin secretion for 2 weeks after a single subcutaneous administration.49 Nevertheless, the requirement for frequent parenteral administration and the high cost of such agents make them impractical as a MCM. The development of non-peptidic antagonists suitable for oral administration50 or slow-release implants (histrelin) may reignite interest in testing these substances.51
Androgen - progestin combinations
Progestins, comprising an integral component of female contraception, have been studied extensively in women, A similar suppression of the reproductive axis by progestins has been observed in men and in combination with androgens they can achieve comparable suppression with androgen monotherapy, using lower doses of androgen. Consequently, numerous studies combining androgens with progestins as means of MHC have been conducted. Progestins other than progesterone are of synthetic origin and are classified into 19-norsteroids, derived from T and 21-progestins or pregnanes derived from progesterone.
Norethisterone is a 19-norsteroid that may be administered in the form of enanthate (NETE) as a long-acting injectable formulation and, in conjuction with TU, achieves rates of azoospermia up to 90%.52,53 A multicentre phase II study testing the contraceptive efficacy of 200 mg NETE/1000 mg TU every 8 weeks was funded by the Agency for Contraception Research and Development (CONRAD) in collaboration with the WHO and initially included 321 couples. However, the trial was prematurely interrupted due to the emergence of serious mood-related adverse events. Analysis of the interim results has not been published yet (http://conrad.org/contraception-trials.html).
Levonorgestrel (LNG) is a derivative of norethisterone, which when administered in the form of long-acting implants in conjunction with TU may result in azoospermia in approximately 90% of men.54 Desogestrel is a derivative of LNG, which when administered orally is converted to its active metabolite, etonogestrel (ENG). The combination of ENG implants with TU intramuscularly every 10 to 12 weeks versus placebo was studied in a randomized controlled trial (RCT) funded by the pharmaceutical industry and including 354 volunteers.8 In 90% of men, levels of oligozoospermia ≤1 million/ml were achieved; however, the study did not proceed to the contraceptive efficacy phase due to withdrawal of commercial interest.
Medroxyprogesterone acetate (MPA), a progesterone derivative, has been tested on MHC since the early 1970s.55 The injectable depot form (DMPA) has been tested in combination with T implants or TU and significant oligozoospermia (<1 million/ml) was achieved in >96% of men.56,57 The main disadvantage was the slow onset of the suppression of spermatogenesis. Cyproterone acetate (CPA) is also a progesterone derivative with potent anti-androgenic activity and co-administration of 100 mg CPA with 100 mg TE per week resulted in azoospermia in all 15 volunteers,58 while small doses of CPA (2 mg/day) were able to sustain the severe oligozoospermia caused by a loading dose of CPA 20 mg/day with 1000 mg TU every 6 weeks.59 The time required for these results is significantly less than that of TU monotherapy, probably as a result of a direct activity of CPA at the testicular level; however, the co-administration of androgen is mandatory to avoid androgonopenia.58
Recently, a transdermal gel-based MHC containing T and Nestorone® was tested in a RCT resulting in suppression of spermatogenesis to <1 million/ml in 88.5% of the subjects without serious adverse effects.60 Interestingly, more than 50% of men receiving such therapy were satisfied or extremely satisfied and would use this regimen if it were commercially available.61 Despite these promising results, a Cochrane systematic review published in 2012 evaluating the effectiveness of the various MHC combination regimens underscored their limitations. Thirty-three RCTs were analyzed and the endpoint was the achievement of azoospermia. The conclusion was that although the addition of progestin generally improved the contraceptive effectiveness of T, none of the studied combinations was considered suitable for clinical use as the included studies lacked the required power to detect significant differences and had serious methodological problems.62 The principal trials of T and progestin combinations are summarized in Table 3.
Adverse effects
The co-administration of progestin together with androgens appears to have no adverse effects on body weight and results in HDL-cholesterol concentrations similar to those of androgen monotherapy. The route of progestin administration seems to play an important role, as oral administration is associated with pro-atherogenic action, whereas transdermal administration (implants) is not, probably due to the bypassing of hepatic metabolism.63 In women, the administration of progestins appears to increase cardiovascular disease endpoints and inflammatory markers (IL-6, C-reactive protein); in men, however, the available evidence is inadequate to draw firm conclusions due to the much lower experience and the small size of clinical studies.43 The addition of progestin to the regimen does not seem to affect the time required for the recovery of spermatogenesis.45
Alternative hormonal regimens
An approach that at least theoretically may retain the contraceptive efficacy of T while neutralizing its effects on the prostate gland is the combination of T with 5α-reductase inhibitors. However, no improvement in the efficiency of spermatogenesis suppression was observed in clinical studies.64
Similarly, Selective Androgen Receptor Modulators (SARMs) are compounds that may reserve the desired androgenic effects without affecting metabolism and the prostate gland. The SARM 7a-methyl-19-nor-testosterone (MENT) is minimally converted to dihydrotestosterone (DHT) and thus has negligible adverse effects on the prostate.65 The administration of MENT resulted in azoospermia in 73% of men;66 unfortunately, these results were of short duration, probably due to the weak binding of MENT with sex-hormone binding globulin (SHBG) and its resultant short half-life in the circulation. Accordingly, the development of non-steroidal SARMs suitable for oral administration is under investigation.67
Unanswered questions
Why is MHC not effective in all men?
The fact that some men do not respond to MHC is an important obstacle to further commercialization of the various hormonal regimens. The percentage of non-responders in various clinical trials ranges from 5 to 20%. Up to now, there is no safe way to predict which men will not respond,68 as non-responders present the same pre-therapeutic levels of gonadotropins and T as well as sperm concentrations compared to responders.69 Furthermore, the higher levels of 5α-reductase observed in the non-responding males does not appear to be related to this resistance to treatment, since the administration of 5α-reductase inhibitors did not improve their response.
Recent studies indicate that non-responders presented higher levels of Insulin-Like Growth Factor-3 (IGF-3, a marker of Leydig cell function) during MHC, but a causal relationship could not be established.70 Additionally, it was shown that obese men with higher fat mass were more likely to be non-responders,71 a drawback that could possibly be overcome by adjusting the dose of androgens according to weight and body composition. Finally, genetic polymorphisms in the first exon of the androgen receptor, in particular the increased number of iterations of the trinucleotide CAG, are associated with reduced sensitivity to androgens and, therefore, reduced efficacy as MHC.72
Why do differences in the effectiveness of the contraceptive treatment exist among different races?
Notably, in all studies on MHC men of Asian origin show consistently higher response rates than Caucasians.68 Thus, monotherapy with T may cause azoospermia in more than 90% of Chinese volunteers, whereas the corresponding figures concern only two thirds of Caucasian men.9 So far, no genetic differences have been found that explain this variation. One possible explanation may be the lower body fat mass characterizing Asians, allowing the same doses of androgens to be more effective.
NON-HORMONAL METHODS OF CONTRACEPTION
The non-hormonal contraception methods comprise the most promising field of research on MCMs, given that they target more specific biological processes than the corresponding MHC and could possibly have fewer adverse effects. Nevertheless, with the exception of gossypol, all other methods are still in experimental stages, since both toxicity and reversibility data are discouraging. The non-hormonal methods are categorized into those aiming to a) inhibit spermatogenesis and b) disrupt the movement and maturation of sperm in the epididymis.
Inhibition of spermatogenesis
Chemical/pharmaceutical methods
Although many chemicals can effectively suppress spermatogenesis, most of them are systematically toxic and often produce irreversible sterility. They act either on somatic or germ cells and, in order to affect the meta-meiotic stages of spermatogenesis, it is necessary that they can pass through the blood-testis barrier.73
Gossypol - Triptolide
Gossypol and Triptolide are herbal extracts used principally in China. Gossypol is a phenolic compound derived from cottonseeds that acts as an inhibitor for several dehydrogenase enzymes and has proapoptotic properties, affecting both spermatogenesis and sperm motility. Its use as a MCM was suggested by Chinese researchers, who noted that men consuming cottonseed oil in their diet showed unusually high infertility rates. In studies in China in the 1980s, oral administration of gossypol in large scale trials resulted in severe oligozoospermia (<1 million/ml) at 90% of participants.74 However, in 20% of men this effect was irreversible, whereas in some cases severe hypokalemia occurred, effects that led the WHO to recommend the discontinuation of further investigations on gossypol. Triptolide is derived from the Chinese herb Trypterigium wilfordii and acts both by inhibiting spermatogenesis and impairing sperm mobility; however, studies have shown that triptolide has immunosuppressive effects, while administration in rodents leads to irreversible infertility.75 A systematic review of studies with gossypol and triptolide as MCMs concluded that neither of the two substances was either effective or safe.76
Adjudin - Indenopyridines
Adjudin is a derivative of lonidamine, a nephtoroxic substance that disrupts spermatogenesis without nephrotoxic effects. Adjudin interferes at the connections of germ cells with the Sertoli cells resulting in the release of immature spermatids into the semen, which lack fertilizing capacity. Oral administration of adjudin in rats resulted in reversible infertility; however, unexpected adverse effects were observed, such as hepatic malaise and muscle atrophy,77 while low doses led to low bioavailability of adjudin in the testicular milieu. This could be overcome by binding adjudin on a modified FSH molecule that acts as a transporter towards Sertoli cells. Administration of this preparation in rats with a single injection caused infertility 4-6 weeks after the injection, while fertility returned within 5 months, resulting in normal offspring.77 Indenopyridines are substances found in newer antihistaminics and their contraceptive properties were observed while testing the toxicity of these drugs. Their effect on spermatogenesis seems to involve both Sertoli and germ cells.78 Experimental administration of indenopyridines causes reversible, severe oligozoospermia in both rats and monkeys,79 without apparent toxicity. The exact mechanism of their action is unclear.
Inhibition of retinoic acid at the testicular level
It has been known since the 1920s that vitamin A is essential for normal sperm production in men.80 The use of bis-dichloro-acetyl-diamine (WIN 18446) was one of the first attempts at pharmaceutical contraception in US men in the 1960s and resulted in severe oligozoospermia (<1 million/ml), which was fully reversible.81 However, these trials did not result in a commercial product, since concomitant alcohol intake could cause severe spells of hot flashes, nausea and palpitations, a phenomenon known as “disulfiram reaction”. The mechanism of action of WIN 18446 was attributed several years later to the inhibition of vitamin A to retinoic acid conversion.82
In the 1980s, BMS 189453, a non-selective retinoic acid receptor (RAR) antagonist, which acts on all three known types of RARs (alpha, beta and gamma), was tested on animals as a treatment of inflammatory skin diseases and appeared to cause irreversible infertility and significant hepato- and myelotoxicity.83 However, when lower doses were used in specific fertility experiments, it was demonstrated that infertillity was reversible without significant toxicity.84 Better results could possibly be achieved with the deployment of specific inhibitors of the RAR alpha, which is selectively encountered in the testis.
Mechanical methods
Thermal method
Warm baths before the sexual act is a method of contraception that was used in China long before its scientific documentation. An increase of testicular temperature impairs spermatogenesis due to increased apoptosis of germ cells. Hence, the position of the testes in the scrotum can maintain a temperature of 3-4oC lower than that of the body core.85 The daily use of special underwear (suspensories) holding the testis within the inguinal canal for 42 weeks resulted in a very low sperm count (<1.6 million/ml) in all participants, the only pregnancy that occurred being due to poor compliance. Recovery of spermatogenesis to pre-treatment levels was observed within 12 to 18 months, without any case of irreversible infertility.
Other research groups focused on the use of external heat to achieve contraception, particularly in the form of water baths. The background for these studies was set by the anecdotal observations of Dr M. Voegeli in India who, in the 1950s, implemented various combinations of water temperature, duration of immersion and number of iterations to determine the most efficient regimen.86 A clinical study including immersion of the scrotum in water at 43°C for 30 min every day for six consecutive days resulted in reduction of the concentration and the motility of spermatozoa, though never below 3 million/ml.87 There have been concerns regarding the outcomes on future offspring, as heat may impair DNA synthesis during spermatogenesis. Few trials in animals have shown no increased incidence of birth defects, while Voegeli stated: “all progeny born to patients after discontinuation of thermal treatment were normal”.88
Ultrasound
Research on the use of ultrasound as a MCM dates back to the 1970s when the group of Fahim applied brief sessions of ultrasound waves on the testes of animals, as well as four volunteers, resulting in substantial reduction of the produced spermatozoa without affecting the levels of circulating T. The technique and the frequencies used were those applied in the treatment of musculoskeletal injuries,89 while the effects of ultrasound on spermatogenesis were associated with a provisional increase in the temperature of the testicles. These results were confirmed in subsequent studies where recovery of sperm counts to pre-treatment levels was observed in all cases and the recovery time was proportional to the intensity and duration of ultrasound sessions.90,91
Disruption of transport and maturation of spermatozoa in the epididymis
Pharmaceutical methods
The contraceptive methods applied at the level of the epididymis target already formed sperm with the aim of either preventing further transfer to the ejaculate or of rendering spermatozoa infertile. Therefore, these methods have the theoretical advantage of rapid onset of action and the equally rapid recovery of fertility after withdrawal, a feature that renders them suitable for use even immediately before intercourse.
Disruption of sperm transport in the epididymis
Various agents acting on the autonomous nervous system (e.g. sympathomimetics, sympatholytics, parasympatholytics) result in a rapid induction of azoospermia, which is irreversible at high doses.92,93 These agents seem to induce a discoordination among the muscle layers of the vas deferens and a subsequent inefficiency of sperm promotion.94 Use of phenoxybenzamine can cause a gradual decrease of semen volume up to complete aspermia; however, it is accompanied by unpleasant adverse effects, such as dizziness, tachycardia and nasal congestion.95 Moreover, the long-term use of such agents raised concerns about abnormalities that may be induced in the stored spermatozoa and the production of anti-sperm antibodies, this pointing to the need for intermittent use of these agents only during the fertile days and their interruption during menstruation.96
Conversely, accelerated passage of sperm through the epididymis by substances that increase the contractility of the epididymal walls is associated with poor fertilizing capacity, regardless of their number in the ejaculate (functional sterilization), as a result of reduced interaction with the beneficial epididymal micro-environment. Such agents as peptidergic agonists (e.g. vasopressin, oxytocin, endothelin, angiotensin) or P2a-purinergic receptor antagonists (α-β-methylene-ATP) tested in animals result rapidly in reversible infertility.97 Of concern is the fact that without a proportional reduction in the number of sperm in the ejaculate it is feasible that immature spermatozoa might approach and possibly fertilize the ovum with unknown consequences to the offspring.98
Disruption of the composition of the epididymal micro-environment
The lumen of the epididymis comprises a unique micro-environment characterized by a relatively high osmolarity and acidic pH compared to the serum due to its particular content in electrolytes and small molecule compounds. This micro-environment exerts a significant effect on sperm fertilizing capacity, as shown by the higher fertilization rates achieved in MESA-ICSI by spermatozoa retrieved by distal regions of the epididymis.99
Reduction of the concentration of small molecule compounds such as carnitine in the epididymal fluid (EF) or administration of substances that selectively interfere with the secretory function of the epididymal epithelium such as a-chlorohydrin and 6-chloro-6-deoxyglucose100 decreases the EF osmolarity, causing intraluminal swelling of the sperm. However, experimental lowering of the levels of carnitine in the blood and the epididymis of rodents by administering pivalic acid did not result in infertility.101 Similarly, the acidic pH of the EF appears to be important for the preservation of spermatozoa at rest within the epididymis102 and is maintained by the collaboration of an intracytoplasmic carbonic anhydrase and a (H+)-ATP-pump. However, administration of the carbonic anhydrase inhibitor acetazolamide to mammals does not seem to affect the intraluminal pH of the epididymis.103
Incapacitation of spermatozoa stored within the epididymis
One way to incapacitate sperm is the irreversible inhibition of enzymes involved in their metabolism, which directly impair the mobility of spermatozoa.104 Such an inhibitor is alpha-chlorohydrin; however, its actions are not limited to the sperm, resulting in systemic toxicity. Alternatively, inhibition of sperm activation (capacitation) that occurs in the epididymis can also impair sperm mobility. Recent studies have focused on a new group of voltage-sensitive calcium channels, called cation-channels of sperm (CatSpers), which allow the entry of calcium into the immature sperm, thereby increasing the frequency and amplitude of tail vibration.105 Blockage of CatSpers may prevent this hyperactivated motility, as shown by experiments in animals employing CatSper blockers, such as HC-056456106 or anti CatSper1 immunoglobulin G.107
Finally, a promising approach for a MCM is by interfering in the interaction between the sperm and the ovum. Sperm contain the enzyme N-acetyl-beta-D hexosaminidase type B or HEX-B, which cleaves the ZP-glycoproteins of the zona pellucida of the ovum. In vitro employment of ZP-glycoprotein analogues to block HEX-B of rodent and bovine semen had 98% efficacy regarding fertilization failure,108 while in vivo, the corresponding efficiency rates were approximately 90%,109 without overt adverse effects and restoration of fertility one week after discontinuation of treatment.
Immunological approaches
The detection of positive anti-sperm antibodies, and particularly of the IgA type, is associated with reduced probability of recovery of fertility after vasectomy.110 Similarly, the active immunization against sperm has already been widely used for the control of wildlife populations;111 however, unpredictable individual responses have been observed, and the fact that the reversibility of immunization is theoretically difficult because of the existence of immunological memory has not favored the implementation in humans as yet.112 Immunization experiments in mice against urokinase-type plasminogen activator, a protease expressed throughout the male reproductive system, and more recently in primates by administering recombinant human eppin (an epididymal protein) resulted in reversible contraception; however, in order to maintain a high antibody titer, booster doses were required every 3 weeks throughout the course of treatment.113,114 Recently, vaccines targeting gonadotropins or GnRH have been tested in women (phase I studies) and could be applied in men.115 Immunization of male monkeys against the FSH receptor reduced their fertility, while the serum hormone levels and testicular histology were not disturbed.116
Mechanical methods
These are methods alternative to vasectomy, which are also aimed at preventing the passage of sperm to the ejaculate with higher and faster reversibility.
Reversible Inhibition of Sperm Under Guidance (RISUG)
Perhaps this is the most promising of these techniques, as it has already been tested in phase I and II studies in the Indian male population and has been shown to provide effective contraception for up to 10 years after a single application.117,118 During RISUG, a solution of 60 mg of styrene maleic anhydride (SMA), a crystal clear polymer dissolved in 120 µl of dimethyl sulfoxide (DMSO) (1:2), is injected into the vas deferens and induces infertility within 10 days. SMA (Xiran®) partially occludes the vas deferens and at the same time develops morphological aberrations of the sperm that manage to pass through (Figure 2A).119 A single application is readily effective, has few adverse effects and is easily reversible by injecting 200-500 µl of DMSO or 5% NaHCO3 into the vas deferens, causing the extrusion of RISUG from the urethra. Adverse effects include slight testicular swelling without associated pain, which is self-limited within 15 days.118 In contrast to vasectomy, it does not cause granulomas or an autoimmune response. There are concerns with respect to the potential toxic effects and teratogenicity of the material, which is not included in the hazardous substances database (HSDB). Nevertheless, recent studies of genotoxicity in rodents have demonstrated that RISUG and its reversal are unlikely to produce any DNA damage.119 An extended phase III clinical trial has been ongoing since 2002 and is expected to elucidate these challenges.120
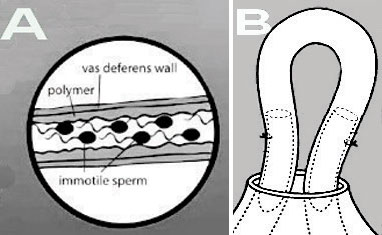
Figure 2. A. How RISUG works: styrene maleic anhydride polymer is injected into the vas deferens and acts by both inducing the partial occlusion of the vas deferens as well as by developing morphological aberrations of the sperm that manage to pass through. B. Intra-vas plugs.
Intra-vas plugs
Two different types of injectable intraluminal plugs have been tested: medical polyurethane (MPU) and medical silicone resin (MSR) plugs (Figure 2B). The MPU plugs have been tested mainly in China since the early 1980s in large-scale clinical trials, with the largest group including 12,000 men. Almost 98% of men achieved azoospermia;121 however, 18 to 24 months were required to achieve this level of efficiency. The results for the MSR plugs were not particularly promising as azoospermia rates did not exceed 80%;122 on the other hand, their placement is easier. The complication rate is low, since the placement of the plugs does not require a surgical incision, while plug removal is performed under local anesthesia on an outpatient basis and restores fertility in approximately 85% of men. The reversal of the contraceptive action is slow but steady and may be achieved in 2-4 years.123 Moreover, it has the theoretical advantage of repeatability, since removing and replacing can be performed without substantial damage. Concerns exist about the potential toxicity of aromatic amines contained in MPU. So far, the experience of many years of use in China has been free of incidents of toxicity.123
FUTURE APPROACHES
Glycosphingolipids are expressed in abundance in the sperm. Mice deficient in enzymes involved in the biosynthesis and metabolism of glycosphingolipids show significantly reduced fertility. However, administration of miglustat, a glycosphingolipid synthesis inhibitor, in healthy men had no effect on the fertilizing capacity of sperm.124 Recent experiments include the study of the administration of small molecule JQ1, an inhibitor for the specific-testis protein bromo-domain BRDT. BRDT is a protein involved in the conformation of histones and chromatin folding and occurs in meiotic spermatocytes and spermatids. Administration of JQ1 inhibitor in mice induces reversible infertility. Given the similarity of mouse BRDT to that of humans, this could be a promising MCM.125 Ultimately, the development of the science of proteomics is setting the stage for the discovery of new targets on spermatozoa, for a more eclectic approach to contraception. The fact that sperm are rich in membrane proteins in comparison to other cells and that protein synthesis is arrested beyond the stage of spermatids facilitates the study of their protein profile.126
CONCLUSIONS
Despite the social need and willingness of men to participate in family planning, no male pharmaceutical contraceptive is available for clinical use so far. Currently, only two male contraceptive methods are available: the male condom and vasectomy, which both present significant limitations. Among contraceptive methods under investigation, MHC are those that have come closest to commercialization. Unfortunately, concerns regarding the impracticality of their use, unpredictable response rates and long-term associated prostate and cardiovascular morbidity have deflected the interest of pharmaceutical industry from further research in this field. Hopefully, the favorable results and increased patient satisfaction reported recently regarding an all-gel testosterone-progestin combination may reignite interest in MHC. On the other hand, non-hormonal contraception methods comprise the most promising field of research into MCM, since at least theoretically, they may be very specific; however, most of them are still in experimental stages or have long been abandoned due to their toxicity. Consequently, until a reliable, safe and practical male contraceptive method is developed, women will still have to bear most of the contraception burden.
FUNDING
The authors state that there is no conflict of interest.
REFERENCES
1. Population Division of the Department of Economic and Social Affairs of the United Nations Secretariat 2007 World population prospects: the 2006 revision and world urbanization prospects: the 2005 revision.
2. Finer LB, Henshaw SK, 2003 Abortion incidence and services in the United States in 2000. Perspect Sex Reprod Health 35: 6-15.
3. Trussell J, 1998 Emergency contraception: WHO Task Force study. Lancet 352: 1222-1223.
4. Anderson RA, Van Der Spuy ZM, Dada OA, et al, 2002 Investigation of hormonal male contraception in African men: suppression of spermatogenesis by oral desogestrel with depot testosterone. Hum Reprod 17: 2869-2877.
5. Lewin A, Mitchell SJ, Hodgkinson S, Gilmore J, Beers LS, 2014 Pregnancy intentions among expectant adolescent couples. J Pediatr Adolesc Gynecol 27: 172-176.
6. Heinemann K, Saad F, Wiesemes M, Heinemann LA, 2005 Expectations toward a novel male fertility control method and potential user types: results of a multinational survey. J Androl 26: 155-162.
7. Glasier AF, Anakwe R, Everington D, et al, 2000 Would women trust their partners to use a male pill? Hum Reprod 15: 646-649.
8. Mommers E, Kersemaekers WM, Elliesen J, et al, 2008 Male hormonal contraception: a double-blind, placebo-controlled study. J Clin Endocrinol Metab 93: 2572-2580.
9. Page ST, Amory JK, Bremner WJ, 2008 Advances in male contraception. Endocr Rev 29: 465-493.
10. Mosher WD, Martinez GM, Chandra A, Abma JC, Willson SJ, 2004 Use of contraception and use of family planning services in the United States: 1982-2002. Adv Data 1-36.
11. April K, Köster R, Schreiner W, 1993 [How effectively do condoms prevent HIV transmission?]. Med Klin (Munich) 88: 304-311.
12. Steiner M, Foldesy R, Cole D, Carter E, 1992 Study to determine the correlation between condom breakage in human use and laboratory test results. Contraception 46: 279-288.
13. Dowbiggin I 2008 The Sterilization Movement and Global Fertility in the Twentieth Century. Oxford University Press New York;
14. Schwingl PJ, Guess HA, 2000 Safety and effectiveness of vasectomy. Fertil Steril 73: 923-936.
15. Rizvi SA, Naqvi SA, Hussain Z, 1995 Ethical issues in male sterilization in developing countries. Br J Urol 76: Suppl 2: 103-105.
16. Trussell J, Hatcher RA, Cates W, Stewart FH, Kost K, 1990 Contraceptive failure in the United States: an update. Stud Fam Plann 21: 51-54.
17. Goldstein M, 1983 Vasectomy failure using an open-ended technique. Fertil Steril 40: 699-700.
18. Siddiqui MM, Wilson KM, Epstein MM, et al, 2014 Vasectomy and risk of aggressive prostate cancer: a 24-year follow-up study. J Clin Oncol 32: 3033-3038.
19. Cos LR, Valvo JR, Davis RS, Cockett AT, 1983 Vasovasostomy: current state of the art. Urology 22: 567-575.
20. Goldstein M, Li PS, Matthews GJ, 1998 Microsurgical vasovasostomy: the microdot technique of precision suture placement. J Urol 159: 188-190.
21. Engelmann UH, Schramek P, Tomamichel G, Deindl F, Senge T, 1990 Vasectomy reversal in central Europe: results of questionnaire of urologists in Austria, Germany and Switzerland. J Urol 143: 64-67.
22. Heidenreich A, Bonfig R, Wilbert DM, Strohmaier WL, Engelmann UH, 1994 Risk factors for antisperm antibodies in infertile men. Am J Reprod Immunol 31: 69-76.
23. Belker AM, Thomas AJ, Fuchs EF, Konnak JW, Sharlip ID, 1991 Results of 1,469 microsurgical vasectomy reversals by the Vasovasostomy Study Group. J Urol 145: 505-511.
24. Roth MY, 2012 Male hormonal contraception. Virtual Mentor 14: 126-132.
25. Aaltonen P, Amory JK, Anderson RA, et al, 2007 10th Summit Meeting consensus: recommendations for regulatory approval for hormonal male contraception. J Androl 28: 362-363.
26. World Health Organization Task Force on Methods for the Regulation of Male Fertility (1990) Contraceptive efficacy of testosterone-induced azoospermia in normal men. Lancet 336: 955-959.
27. World Health Organization Task Force on Methods for the Regulation of Male Fertility 1996 Contraceptive efficacy of testosterone-induced azoospermia and oligozoospermia in normal men. Fertil Steril 65: 821-829.
28. Gu YQ, Wang XH, Xu D, et al, 2003 A multicenter contraceptive efficacy study of injectable testosterone undecanoate in healthy Chinese men. J Clin Endocrinol Metab 88: 562-568.
29. McLachlan RI, Robertson DM, Pruysers E, et al, 2004 Relationship between serum gonadotropins and spermatogenic suppression in men undergoing steroidal contraceptive treatment. J Clin Endocrinol Metab 89: 142-149.
30. Gu Y, Liang X, Wu W, et al, 2009 Multicenter contraceptive efficacy trial of injectable testosterone undecanoate in Chinese men. J Clin Endocrinol Metab 94: 1910-1915.
31. Kamischke A, Plöger D, Venherm S, von Eckardstein S, von Eckardstein A, Nieschlag E, 2000 Intramuscular testosterone undecanoate with or without oral levonorgestrel: a randomized placebo-controlled feasibility study for male contraception. Clin Endocrinol (Oxf) 53: 43-52.
32. Waites GM, 2003 Development of methods of male contraception: impact of the World Health Organization Task Force. Fertil Steril 80: 1-15.
33. Behre HM, Baus S, Kliesch S, Keck C, Simoni M, Nieschlag E, 1995 Potential of testosterone buciclate for male contraception: endocrine differences between responders and nonresponders. J Clin Endocrinol Metab 80: 2394-2403.
34. Handelsman DJ, Conway AJ, Boylan LM, 1992 Suppression of human spermatogenesis by testosterone implants. J Clin Endocrinol Metab 75: 1326-1332.
35. McLachlan RI, 2000 Male hormonal contraception: a safe, acceptable and reversible choice. Med J Aust 172: 254-255.
36. Nieschlag E, Hoogen H, Bölk M, Schuster H, Wickings EJ, 1978 Clinical trial with testosterone undecanoate for male fertility control. Contraception 18: 607-614.
37. Amory JK, Bremner WJ, 2005 Oral testosterone in oil plus dutasteride in men: a pharmacokinetic study. J Clin Endocrinol Metab 90: 2610-2617.
38. Gonzalo IT, Swerdloff RS, Nelson AL, et al, 2002 Levonorgestrel implants (Norplant II) for male contraception clinical trials: combination with transdermal and injectable testosterone. J Clin Endocrinol Metab 87: 3562-3572.
39. Vigen R, O’Donnell CI, Barón AE, et al, 2013 Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA 310: 1829-1836.
40. Bhasin S, Woodhouse L, Storer TW, 2001 Proof of the effect of testosterone on skeletal muscle. J Endocrinol 170: 27-38.
41. Nieschlag E. 2010 Male contribution to contraception. In: Andrology, 3rd Edition edn. Springer-Verlag, Berlin Heidelberg; pp. 557-564
42. Raynaud JP, 2006 Prostate cancer risk in testosterone-treated men. J Steroid Biochem Mol Biol 102: 261-266.
43. Campagnoli C, Clavel-Chapelon F, Kaaks R, Peris C, Berrino F, 2005 Progestins and progesterone in hormone replacement therapy and the risk of breast cancer. J Steroid Biochem Mol Biol 96: 95-108.
44. Sjögren B, Gottlieb C, 2001 Testosterone for male contraception during one year: attitudes, well-being and quality of sex life. Contraception 64: 59-65.
45. Liu PY, Swerdloff RS, Christenson PD, Handelsman DJ, Wang C, Group HMCS, 2006 Rate, extent, and modifiers of spermatogenic recovery after hormonal male contraception: an integrated analysis. Lancet 367: 1412-1420.
46. Behre HM, Nashan D, Hubert W, Nieschlag E, 1992 Depot gonadotropin-releasing hormone agonist blunts the androgen-induced suppression of spermatogenesis in a clinical trial of male contraception. J Clin Endocrinol Metab 74: 84-90.
47. Swerdloff RS, Bagatell CJ, Wang C, et al, 1998 Suppression of spermatogenesis in man induced by Nal-Glu gonadotropin releasing hormone antagonist and testosterone enanthate (TE) is maintained by TE alone. J Clin Endocrinol Metab 83: 3527-3533.
48. Tomera K, Gleason D, Gittelman M, et al, 2001 The gonadotropin-releasing hormone antagonist abarelix depot versus luteinizing hormone releasing hormone agonists leuprolide or goserelin: initial results of endocrinological and biochemical efficacies in patients with prostate cancer. J Urol 165: 1585-1589.
49. Herbst KL, Coviello AD, Page S, Amory JK, Anawalt BD, Bremner WJ, 2004 A single dose of the potent gonadotropin-releasing hormone antagonist acyline suppresses gonadotropins and testosterone for 2 weeks in healthy young men. J Clin Endocrinol Metab 89: 5959-5965.
50. Struthers RS, Xie Q, Sullivan SK, et al, 2007 Pharmacological characterization of a novel nonpeptide antagonist of the human gonadotropin-releasing hormone receptor, NBI-42902. Endocrinology 148: 857-867.
51. Schlegel PN, Group HS, 2006 Efficacy and safety of histrelin subdermal implant in patients with advanced prostate cancer. J Urol 175: 1353-1358.
52. Meriggiola MC, Costantino A, Saad F, et al, 2005 Norethisterone enanthate plus testosterone undecanoate for male contraception: effects of various injection intervals on spermatogenesis, reproductive hormones, testis, and prostate. J Clin Endocrinol Metab 90: 2005-2014.
53. Kamischke A, Venherm S, Plöger D, von Eckardstein S, Nieschlag E, 2001 Intramuscular testosterone undecanoate and norethisterone enanthate in a clinical trial for male contraception. J Clin Endocrinol Metab 86: 303-309.
54. Gui YL, He CH, Amory JK, et al, 2004 Male hormonal contraception: suppression of spermatogenesis by injectable testosterone undecanoate alone or with levonorgestrel implants in chinese men. J Androl 25: 720-727.
55. Barfield A, Melo J, Coutinho E, et al, 1979 Pregnancies associated with sperm concentrations below 10 million/ml in clinical studies of a potential male contraceptive method, monthly depot medroxyprogesterone acetate and testosterone esters. Contraception 20: 121-127.
56. Turner L, Conway AJ, Jimenez M, et al, 2003 Contraceptive efficacy of a depot progestin and androgen combination in men. J Clin Endocrinol Metab 88: 4659-4667.
57. Gu YQ, Tong JS, Ma DZ, et al, 2004 Male hormonal contraception: effects of injections of testosterone undecanoate and depot medroxyprogesterone acetate at eight-week intervals in chinese men. J Clin Endocrinol Metab 89: 2254-2262.
58. Meriggiola MC, Bremner WJ, Paulsen CA, et al, 1996 A combined regimen of cyproterone acetate and testosterone enanthate as a potentially highly effective male contraceptive. J Clin Endocrinol Metab 81: 3018-3023.
59. Meriggiola MC, Costantino A, Cerpolini S, et al, 2003 Testosterone undecanoate maintains spermatogenic suppression induced by cyproterone acetate plus testosterone undecanoate in normal men. J Clin Endocrinol Metab 88: 5818-5826.
60. Ilani N, Roth MY, Amory JK, et al, 2012 A new combination of testosterone and nestorone transdermal gels for male hormonal contraception. J Clin Endocrinol Metab 97: 3476-3486.
61. Roth MY, Shih G, Ilani N, et al, 2014 Acceptability of a transdermal gel-based male hormonal contraceptive in a randomized controlled trial. Contraception 90: 407-412.
62. Grimes DA, Lopez LM, Gallo MF, Halpern V, Nanda K, Schulz KF, 2012 Steroid hormones for contraception in men. Cochrane Database Syst Rev 3: CD004316.
63. Anderson RA, Kinniburgh D, Baird DT, 2002 Suppression of spermatogenesis by etonogestrel implants with depot testosterone: potential for long-acting male contraception. J Clin Endocrinol Metab 87: 3640-3649.
64. Thompson IM, Goodman PJ, Tangen CM, et al, 2003 The influence of finasteride on the development of prostate cancer. N Engl J Med 349: 215-224.
65. Sundaram K, Kumar N, 2000 7alpha-methyl-19-nortestosterone (MENT): the optimal androgen for male contraception and replacement therapy. Int J Androl 23 Suppl 2: 13-15.
66. von Eckardstein S, Noe G, Brache V, et al, 2003 A clinical trial of 7 alpha-methyl-19-nortestosterone implants for possible use as a long-acting contraceptive for men. J Clin Endocrinol Metab 88: 5232-5239.
67. Diel P, Friedel A, Geyer H, et al, 2007 Characterisation of the pharmacological profile of desoxymethyltestosterone (Madol), a steroid misused for doping. Toxicol Lett 169: 64-71.
68. Brady BM, Anderson RA, 2002 Advances in male contraception. Expert Opin Investig Drugs 11: 333-344.
69. Amory JK, Anawalt BD, Bremner WJ, Matsumoto AM, 2001 Daily testosterone and gonadotropin levels are similar in azoospermic and nonazoospermic normal men administered weekly testosterone: implications for male contraceptive development. J Androl 22: 1053-1060.
70. Amory JK, Page ST, Anawalt BD, Coviello AD, Matsumoto AM, Bremner WJ, 2007 Elevated end-of-treatment serum INSL3 is associated with failure to completely suppress spermatogenesis in men receiving male hormonal contraception. J Androl 28: 548-554.
71. Kornmann B, Nieschlag E, Zitzmann M, Gromoll J, Simoni M, von Eckardstein S, 2009 Body fat content and testosterone pharmacokinetics determine gonadotropin suppression after intramuscular injections of testosterone preparations in normal men. J Androl 30: 602-613.
72. Eckardstein SV, Schmidt A, Kamischke A, Simoni M, Gromoll J, Nieschlag E, 2002 CAG repeat length in the androgen receptor gene and gonadotrophin suppression influence the effectiveness of hormonal male contraception. Clin Endocrinol (Oxf) 57: 647-655.
73. Mruk DD, Lau AS, Sarkar O, Xia W, 2007 Rab4A GTPase catenin interactions are involved in cell junction dynamics in the testis. J Androl 28: 742-754.
74. Liu GZ, Lyle KC, Cao J, 1987 Experiences with gossypol as a male pill. Am J Obstet Gynecol 157: 1079-1081.
75. Qian SZ, 1987 Tripterygium wilfordii, a Chinese herb effective in male fertility regulation. Contraception 36: 335-345.
76. Lopez LM, Grimes DA, Schulz KF, 2005 Nonhormonal drugs for contraception in men: a systematic review. Obstet Gynecol Surv 60: 746-752.
77. Mruk DD, Wong CH, Silvestrini B, Cheng CY, 2006 A male contraceptive targeting germ cell adhesion. Nat Med 12: 1323-1328.
78. Hild SA, Reel JR, Larner JM, Blye RP, 2001 Disruption of spermatogenesis and Sertoli cell structure and function by the indenopyridine CDB-4022 in rats. Biol Reprod 65: 1771-1779.
79. Hild SA, Reel JR, Dykstra MJ, Mann PC, Marshall GR, 2007 Acute adverse effects of the indenopyridine CDB-4022 on the ultrastructure of sertoli cells, spermatocytes, and spermatids in rat testes: comparison to the known sertoli cell toxicant Di-n-pentylphthalate (DPP). J Androl 28: 621-629.
80. Wolbach SB, Howe PR, 1925 Tissue changes following deprivation of fat-soluble A vitamin. J Exp Med 42: 753-777.
81. Heller CG, Moore DJ, Paulsen CA, 1961 Suppression of spermatogenesis and chronic toxicity in men by a new series of bis(dichloroacetyl) diamines. Toxicol Appl Pharmacol 3: 1-11.
82. Amory JK, Muller CH, Shimshoni JA, et al, 2011 Suppression of spermatogenesis by bisdichloroacetyldiamines is mediated by inhibition of testicular retinoic acid biosynthesis. J Androl 32: 111-119.
83. Schulze GE, Clay RJ, Mezza LE, Bregman CL, Buroker RA, Frantz JD, 2001 BMS-189453, a novel retinoid receptor antagonist, is a potent testicular toxin. Toxicol Sci 59: 297-308.
84. Chung SS, Wang X, Roberts SS, Griffey SM, Reczek PR, Wolgemuth DJ, 2011 Oral administration of a retinoic Acid receptor antagonist reversibly inhibits spermatogenesis in mice. Endocrinology 152: 2492-2502.
85. Kandeel FR, Swerdloff RS, 1988 Role of temperature in regulation of spermatogenesis and the use of heating as a method for contraception. Fertil Steril 49: 1-23.
86. Voegeli M. 1956 Contraception through temporary male sterilization. Unpublished manuscript, Northampton, MA. Text available online at http://www.puzzlepiece.org/bcontrol/voegeli1956.txt.
87. Robinson D, Rock J, Menkin MF, 1968 Control of human spermatogenesis by induced changes of intrascrotal temperature. JAMA 204: 290-297.
88. Voegeli M. 1954 Data on the thermic method for temporary male sterilization. Unpublished manuscript, Northampton, MA. Text available at http://www.puzzlepiece.org/bcontrol/voegeli1954.txt.
89. Fahim MS, Fahim Z, Harman J, Thompson I, Montie J, Hall DG, 1977 Ultrasound as a new method of male contraception. Fertil Steril 28: 823-831.
90. VandeVoort CA, Tollner TL, 2012 The efficacy of ultrasound treatment as a reversible male contraceptive in the rhesus monkey. Reprod Biol Endocrinol 10: 81.
91. Tsuruta JK, Dayton PA, Gallippi CM, et al, 2012 Therapeutic ultrasound as a potential male contraceptive: power, frequency and temperature required to deplete rat testes of meiotic cells and epididymides of sperm determined using a commercially available system. Reprod Biol Endocrinol 10: 7.
92. Kempinas WD, Suarez JD, Roberts NL, et al, 1998 Rat epididymal sperm quantity, quality, and transit time after guanethidine-induced sympathectomy. Biol Reprod 59: 890-896.
93. Kedia K, Markland C, 1975 The effect of pharmacological agents on ejaculation. J Urol 114: 569-573.
94. Amobi N, Guillebaud J, Kaisary A, Lloyd-Davies RW, Turner E, Smith IC, 2003 Contractile actions of imidazoline alpha-adrenoceptor agonists and effects of noncompetitive alpha1-adrenoceptor antagonists in human vas deferens. Eur J Pharmacol 462: 169-177.
95. Homonnai ZT, Shilon M, Paz GF, 1984 Phenoxybenzamine--an effective male contraceptive pill. Contraception 29: 479-491.
96. Heath JW, Evans BK, Gannon BJ, Burnstock G, James VB, 1972 Degeneration of adrenergic neurons following guanethidine treatment: an ultrastructural study. Virchows Arch B Cell Pathol 11: 182-197.
97. Ventura S, Pennefather JN, 1991 Sympathetic co-transmission to the cauda epididymis of the rat: characterization of postjunctional adrenoceptors and purinoceptors. Br J Pharmacol 102: 540-544.
98. Ricker DD, Crone JK, Chamness SL, Klinefelter GR, Chang TS, 1997 Partial sympathetic denervation of the rat epididymis permits fertilization but inhibits embryo development. J Androl 18: 131-138.
99. Cooper TG, 1998 Interactions between epididymal secretions and spermatozoa. J Reprod Fertil Suppl 53: 119-136.
100. Hinton BT, Hernandez H, Howards SS, 1983 The male antifertility agents alpha chlorohydrin, 5-thio-D-glucose, and 6-chloro-6-deoxy-D-glucose interfere with sugar transport across the epithelium of the rat caput epididymidis. J Androl 4: 216-221.
101. Rodríguez CM, Labus JC, Hinton BT, 2002 Organic cation/carnitine transporter, OCTN2, is differentially expressed in the adult rat epididymis. Biol Reprod 67: 314-319.
102. Carr DW, Usselman MC, Acott TS, 1985 Effects of pH, lactate, and viscoelastic drag on sperm motility: a species comparison. Biol Reprod 33: 588-595.
103. Caflisch CR, DuBose TD, 1990 Direct evaluation of acidification by rat testis and epididymis: role of carbonic anhydrase. Am J Physiol 258: E143-150.
104. Jones AR, Cooper TG, 1999 A re-appraisal of the post-testicular action and toxicity of chlorinated antifertility compounds. Int J Androl 22: 130-138.
105. Singh AP, Rajender S, 2015 CatSper channel, sperm function and male fertility. Reprod Biomed Online 30: 28-38.
106. Carlson AE, Burnett LA, del Camino D, et al, 2009 Pharmacological targeting of native CatSper channels reveals a required role in maintenance of sperm hyperactivation. PLoS One 4: e6844.
107. Li H, Ding X, Guan H, Xiong C, 2009 Inhibition of human sperm function and mouse fertilization in vitro by an antibody against cation channel of sperm 1: the contraceptive potential of its transmembrane domains and pore region. Fertil Steril 92: 1141-1146.
108. Hall JC, Perez FM, Kochins JG, et al, 1996 Quantification and localization of N-acetyl-beta-D-hexosaminidase in the adult rat testis and epididymis. Biol Reprod 54: 914-929.
109. Tassi C, Angelini A, Beccari T, Capodicasa E, 2006 Fluorimetric determination of activity and isoenzyme composition of N-acetyl-beta-D-hexosaminidase in seminal plasma of fertile men and infertile patients with secretory azoospermia. Clin Chem Lab Med 44: 843-847.
110. Meinertz H, Linnet L, Fogh-Andersen P, Hjort T, 1990 Antisperm antibodies and fertility after vasovasostomy: a follow-up study of 216 men. Fertil Steril 54: 315-321.
111. Barfield JP, Nieschlag E, Cooper TG, 2006 Fertility control in wildlife: humans as a model. Contraception 73: 6-22.
112. Nieschlag E, Henke A, 2005 Hopes for male contraception. Lancet 365: 554-556.
113. O’rand MG, Widgren EE, Sivashanmugam P, et al, 2004 Reversible immunocontraception in male monkeys immunized with eppin. Science 306: 1189-1190.
114. Qin Y, Han Y, Xiong CL, Li HG, Hu L, Zhang L, 2014 Urokinase-type plasminogen activator: a new target for male contraception? Asian J Androl 16: 623-630.
115. Naz RK, Gupta SK, Gupta JC, Vyas HK, Talwar AG, 2005 Recent advances in contraceptive vaccine development: a mini-review. Hum Reprod 20: 3271-3283.
116. Xu C, Li YC, Yang H, et al, 2014 The preparation and application of N-terminal 57 amino acid protein of the follicle-stimulating hormone receptor as a candidate male contraceptive vaccine. Asian J Androl 16: 623-630.
117. Guha SK, Singh G, Anand S, Ansari S, Kumar S, Koul V, 1993 Phase I clinical trial of an injectable contraceptive for the male. Contraception 48: 367-375.
118. Guha SK, Singh G, Ansari S, et al, 1997 Phase II clinical trial of a vas deferens injectable contraceptive for the male. Contraception 56: 245-250.
119. Lohiya NK, Alam I, Hussain M, Khan SR, Ansari AS, 2014 RISUG: An intravasal injectable male contraceptive. Indian J Med Res 140 Suppl: S63-72.
120. Chaki SP, Das HC, Misro MM, 2003 A short-term evaluation of semen and accessory sex gland function in phase III trial subjects receiving intravasal contraceptive RISUG. Contraception 67: 73-78.
121. Zhao SC, 1990 Vas deferens occlusion by percutaneous injection of polyurethane elastomer plugs: clinical experience and reversibility. Contraception 41: 453-459.
122. Lohiya NK, Manivannan B, Mishra PK, Pathak N, 2001 Vas deferens, a site of male contraception: an overview. Asian J Androl 3: 87-95.
123. Zhao SC, Lian YH, Yu RC, Zhang SP, 1992 Recovery of fertility after removal of polyurethane plugs from the human vas deferens occluded for up to 5 years. Int J Androl 15: 465-467.
124. Amory JK, Muller CH, Page ST, et al, 2007 Miglustat has no apparent effect on spermatogenesis in normal men. Hum Reprod 22: 702-707.
125. Matzuk MM, McKeown MR, Filippakopoulos P, et al, 2012 Small-molecule inhibition of BRDT for male contraception. Cell 150: 673-684.
126. Rahman MS, Lee JS, Kwon WS, Pang MG, 2013 Sperm proteomics: road to male fertility and contraception. Int J Endocrinol 2013: 360986.
Address for correspondence:
George A. Kanakis, 70 Deinokratous Str., 11521 Athens, Greece; Tel. & Fax: +30 2107709248, E-mail: geokan@endo.gr
Received: 07-07-2015, Accepted: 07-09-2015