1Cultural and Educational Center “G. & H. Krassas”, 2Department of Endocrinology, St. Paul Hospital, Thessaloniki, Greece
The most common thyroid diseases during pregnancy are hyper- and hypothyroidism and their variants including isolated hypothyroxinemia (hypo-T4), autoimmune thyroid disease (AITD) and different types of goiter. AITD represents the main cause of hypothyroidism during pregnancy ranging in prevalence between 5 and 20% with an average of 7.8%. The incidence of isolated hypo-T4 is about 150 times higher compared to congenital hypothyroidism. Prevalence of Graves’ disease (GD) ranges between 0.1% and 1% and the Transient Gestational Hyperthyroidism Syndrome between 1 and 3%. Thyroid stimulating hormone (TSH) is a sensitive marker of thyroid dysfunction during pregnancy. Normal values have been modified recently with a downward shift. Thus, the upper normal range is now considered to be 2.5 mUI/mL in the first trimester and 3.0 mUI/mL for the remainder of pregnancy. Most studies have shown that children born to women with hypothyroidism during gestation had significantly lower scores in neuropsychological tests related to intelligence, attention, language, reading ability, school performance and visual motor performance. However, some studies have not confirmed these findings. On the other hand, multiple retrospective studies have shown that the risks of maternal and fetal/neonatal complications are directly related to the duration and inadequate control of maternal thyrotoxicosis. The latter is associated with a risk of spontaneous abortion, congestive heart failure, thyrotoxic storm, preeclampsia, preterm delivery, low birth weight and stillbirth. Despite the lack of consensus among professional organizations, recent studies, which are based on sophisticated analyses, support universal screening in all pregnant women in the first trimester for thyroid diseases.
Autoimmune thyroiditis, Hyperthyroidism, Hypothyroidism, Pregnancy, Screening during pregnancy, Thyroid disease
INTRODUCTION
Numerous hormonal changes as well as alterations in metabolic demands occur during pregnancy, resulting in profound and complex effects on thyroid function. Because thyroid disorders are much more prevalent in women of childbearing age than in men of the same age group, it is not surprising that common thyroid disorders, such as chronic autoimmune thyroiditis, hypothyroidism, Graves’ disease (GD), etc, are relatively frequently observed in pregnant women. The main change in thyroid function associated with the pregnant state is the requirement for an increased production of thyroid hormone which depends directly upon adequate availability of dietary iodine and a normal and functional thyroid gland. Indeed, the physiological adaptation to the pregnant state can only take place when the iodine intake is appropriate. When iodine intake is deficient, however, pregnancy can reveal an underlying iodine deficiency.1 Iodine deficiency (ID) during pregnancy and infancy may impair growth and neurodevelopment of the offspring and increase infant mortality. It is noteworthy that assessment of iodine status in pregnancy is difficult. Meanwhile, it remains unclear whether iodine intakes are sufficient in this group, leading to calls for iodine supplementation during pregnancy in several industrialized countries.2 The economy of the thyroid is modified by several complex physiological changes such as the marked increase in both serum thyroid binding globulin (TBG) concentrations and extrathyroidal thyroxine (T4) distribution space that take place during the first half of gestation. To maintain the homeostasis of T4 concentrations, the thyroid machinery must produce more T4 until a new steady state is reached around mid-gestation. Thus, the main change in thyroid function associated with the pregnant state is the requirement of an increased production of thyroid hormone which, in turn, depends directly upon the adequate availability of dietary iodine and integrity of the thyroid gland. Therefore, any functional perturbation of normal thyroid function may have consequences for pregnancy outcome, and conversely, pregnancy by itself may affect the presentation and course of most thyroid disorders.3-9
THYROID FUNCTION TESTING DURING PREGNANCY
The normal reference range of serum thyroid stimulating hormone (TSH) is modified during pregnancy, implying the need to define trimester-specific normative TSH reference values. Of note, serum free T4 (FT4) estimates, as measured by most – if not all – commonly available FT4 assays, are flawed during pregnancy.
TSH is a sensitive marker of thyroid dysfunction during pregnancy.10,11 This is true despite the significant effect of human chorionic gonadotropin (hCG) on TSH concentrations, especially in the first trimester. During the first trimester, hCG induces a decrease in circulating TSH and, as a consequence, reference values have been modified accordingly. As a result, utilizing normal values for the non-pregnant population leads to diagnostic errors. Due to the interference of pregnancy-modified plasma proteins, the measurement of maternal FT4 with the usual techniques presents some difficulties. Certain FT4 immunoassays have a high correlation with equilibrium dialysis, which is considered today as the gold standard for FT4 measurement during pregnancy.12,13
The determination of total T4 (TT4) has also been proposed as an alternative method for evaluating thyroid function during pregnancy, as TT4 measurements are performed by a more robust methodology than those used for FT4. The increase in TT4 due to the increase in placental hCG is more predictable and it appears that the reference values established in different populations are more comparable and probably more reliable than those obtained for FT4. Of interest, it has recently been suggested that the normal levels for TT4 may be reliably obtained by multiplying by 1.5 the reference values of non-pregnant women.14 However, the validity of such a method has been questioned because of the close relationship between TT4 and the variability of TBG.15
FT4 index (FT4I) which is based on two estimates, namely a measurement of T3 resin uptake and an immunoassay estimate of TT4, could still be a good method for situations when TBG concentrations are dynamically modified, as occurs during pregnancy.16 However, FT4I frequently fails to completely correct for the TBG-induced increase in TT4.
The recent American Thyroid Association (ATA) guideline recommends that in the absence of local reference values and after taking into account the importance of potential variations between the technical methods of measurement, the upper normal value for TSH in pregnant women should be 2.5 mUI/mL in the first trimester and 3.0 mUI/mL in the second and third trimesters. This value of 2.5 mUI/mL was chosen not only because it is close to the 97.5 percentile but also because higher values are associated with higher fetal morbidity.5,17 Figure 1 illustrates the downward shift of the TSH reference range according to the trimesters of pregnancy on the basis of data from three relevant publications.
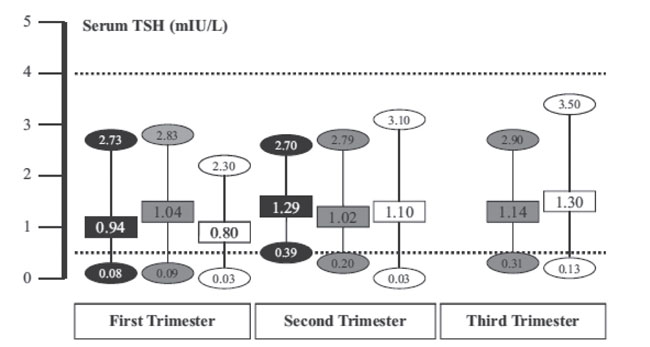
Figure 1. The figure illustrates the downward shift of the TSH reference range, according to trimesters of pregnancy. The graph shows the median TSH values (boxes) and the upper and lower reference limits of normal (ovals). The upper and lower limits show the 5th and 95th centiles. For the black set of symbols see ref. 30. For the grey set see ref. 72 and for the white set see ref. 73. The horizontal dotted lines show the non-pregnant serum TSH reference range (0.40–4.0 mIU/L). Modified from ref. 4
THYROID DISEASES AND PREGNANCY
Thyroid dysfunction during pregnancy includes overt hypothyroidism (OH) and subclinical hypothyroidism (SCH), with a relative incidence of approximately 0.4% for OH and 3% for SCH, as well as overt and subclinical thyrotoxicosis, with relative incidences of approximately 0.2% for the former and 2.5% for the latter. Finally, concerning autoimmune thyroid disease (AITD), it was shown to range between 5 and 20%, with an average of 7.8%. AITD represents the main cause leading to hypothyroidism in pregnant women.
OH is defined as a low serum FT4 with elevated serum TSH concentration. With regard to SCH in pregnancy, the upper normal limit of serum TSH values is shifted downward because of the indirect dampening effect of elevated serum hCG levels of TSH secretion, hCG acting as a TSH-like agonist on the thyroid gland. Due to the downward shifting of TSH, a significant proportion of pregnant women with only slight serum TSH elevations may not be diagnosed.4,8
Besides the classical presentations of maternal thyroid failure as OH and SCH, another condition has been reported specifically in the context of pregnancy, namely isolated hypothyroxinemia (hypo-T4), which is defined as a lowering in serum FT4 without concomitant serum TSH elevation. This biochemical condition was first described in pregnant women residing in areas with mild to moderate ID, which is today believed to possibly be responsible for relative hypo-T4 with normal TSH.18 However, the causes of the latter currently remain not well understood and even somewhat enigmatic.
The incidence of isolated hypo-T4 is about 150 times more frequent than congenital hypothyroidism.19
Regarding hyperthyroidism, prevalence of GD with ranges between 0.1% and 1%20 and transient gestational hyperthyroidism syndrome, with ranges between 1 and 3%, have been reported.21 Multipleretrospective studies have shown that adverse outcomes of pregnancy, namely risks of maternal and fetal/neonatal complications, were directly related to the duration and inadequate control of maternal thyrotoxicosis.22-28 Poor control of thyrotoxicosis is associated with a risk of spontaneous abortion, congestive heart failure, thyrotoxic storm, preeclampsia, preterm delivery, low birth weight and stillbirth. In a study from Australia by Smith et al,29 the outcome of pregnancy was evaluated in women with undiagnosed GD. Results demonstrated severe prematurity (mean delivery time, 30 wks) associated with very low birth weight (<2 kg) and neonatal hyperthyroidism requiring antithyroid drugs (ATDs) treatment. In contrast, pregnancy outcome was excellent for women with GD in whom diagnosis was made early and ATD treatment was started promptly.29
The overall goal of therapy is to control maternal thyrotoxicosis as early as possible. With the use of ATDs, which constitute the first-line therapy option for GD in pregnancy, women should be maintained at a high euthyroid or borderline hyperthyroid level.24,30,31 Because all ATDs cross the placenta, it is recommended to use the smallest possible ATD dose that will allow for controlling maternal thyrotoxicosis without the risk of harming the fetus because ATDs also inhibit fetal thyroid function. Combined administration of ATD and levothyroxine (LT4) to the mother should be avoided because the transplacental passage of ATD is high, whereas it is negligible for thyroid hormones; hence, addition of LT4 will not protect the fetus from ATD-induced hypothyroidism. ATDs belong to the thionamide drug family, the compounds prescribed being propylthiouracil (PTU), methimazole (MTZ) or carbimazol (CMZ), the pharmacological precursor of MTZ.31-33 MTZ has been reported to be the cause of congenital abnormalities, so called “MTZ embryopathy”, which includes aplasia cutis and choanal or esophageal atresia.34-36 Aplasia cutis, which is the absence of skin and accessory structures over the scalp, has so far not been reported in mothers exposed to PTU. Skin defects are estimated to occur in one of approximately 4,000–10,000 pregnancies and, from the scarce data available, it is considered that this incidence does not exceed background incidence in pregnant women who have received MTZ.37 Choanal or esophageal atresia is a severe congenital anomaly requiring major surgery to repair and is considered to have a higher incidence than expected in fetuses exposed to MTZ in the first trimester.38 The relative risk of choanal atresia in pregnant women receiving MTZ was estimated to be approximately 17-fold greater than in the general population, although it should be noted that such congenital birth defects could also be attributed to thyrotoxicosis per se rather than to the administration of MTZ.39
With regard to PTU, a controversy has recently come to light after the alarming report that the use of PTU for treatment of pediatric GD was associated with a significant and therefore unacceptable risk of liver failure.40-42 Soon thereafter, The Endocrine Society alerted its members about the risk of hepatotoxicity and recommended that PTU use be stopped in the pediatric population.43 An additional concern is that liver failure related to PTU administration is idiosyncratic and, therefore, no biomarkers can be used to predict liver toxicity.44 In the specific context of the first trimester of pregnancy, however, PTU remains the drug of choice because of the potential adverse effects of MTZ on the fetus, described above.45,46 Although there have been a few isolated reports of maternal liver injury associated with the use of PTU in pregnancy, there are presently no clear data that would allow to us evaluate the relative risks of MTZ-induced fetal anomalies vs PTU-associated liver failure. A possible modification of currently available recommendations could be to limit the use of PTU to the first trimester of gestation –during which time completion of organogenesis takes place– and possibly to switch women with active GD to MΤΖ treatment thereafter. Finally, pregnant women under PTU should report any new symptom (such as anorexia, nausea, etc.) to their physician and, although liver toxicity may appear abruptly, it seems reasonable to recommend monitoring of liver function tests.
Guidelines for GD treatment in pregnancy are depicted in Table 1.
Recently, Bulmus et al47 conducted a prospective clinical study with 998 pregnant women between the ages of 17–48 years. In the first step, a detailed medical history was obtained and a detailed thyroid gland examination was performed in all subjects (n=998). In pregnant women diagnosed with thyroid disease (TD) or considered to have TD with these results (n=107), TD was evaluated via thyroid function tests and imaging methods. In the second step, TSH, FT4 and free triiodothyronine (FT3) were measured in the first antenatal examination of the pregnant cases considered not to have TD after medical history and examinations (n=891). Thyroid antibodies (Abs) were measured in cases with abnormal thyroid indices. It was found that the incidence of hyper- and hypothyroidism during pregnancy in the whole study group came to 71 cases, 67 of whom had TD before pregnancy. They concluded that detailed medical history and family history obtained during the first trimester of pregnancy helped them to identify 67/71 cases who had TD before pregnancy.
The authors emphasize the importance of detailed first prenatal examination regarding the thyroid.
MATERNAL THYROID DISEASES AND THEIR ASSOCIATION WITH MATERNAL, FETAL, NEONATAL AND INFANT DISTURBANCES
Several studies have shown an association between abnormal thyroid status and different diseases which would support the recommendation of universal screening for TD in pregnancy.
The generation R study48 investigated 5153 pregnant women in early pregnancy. Serum TSH, FT4 and thyroid peroxidase (TPO) Abs levels were determined, while the association of thyroid function with the risk of hypertensive disorders was studied. The authors demonstrated that biochemical hyperthyroidism and also high-normal FT4 levels during early pregnancy are associated with an increased risk of hypertensive disorders. Τhese associations are even seen for a mild variation in thyroid function within the normal range.48
A prospective population based cohort study in China49 investigated the association between thyroid abnormalities in pregnancy and subsequent fetal and infant development. They investigated 1017 women with singleton pregnancies. Maternal serum samples in the first 20 wks of pregnancy were tested for TSH and FT4. Pregnant women were classified by hormone status into percentile categories based on laboratory assay and were compared accordingly. The authors found that OH was associated with increased fetal loss, low birth weight and congenital circulation system malformations. SCH was associated with increased fetal distress, preterm delivery, poor vision development and neurodevelopmental delay. Isolated hyperthyroxinemia was related to fetal distress, small-for-gestational-age and musculoskeletal malformations. Isolated hyperthyroxinemia was associated with spontaneous abortion. Finally, clinical hyperthyroidism was associated with hearing dysplasia. The conclusion is that thyroid dysfunction in the first 20 wks of pregnancy may result in fetal loss and dysplasia and some congenital malformations.
Ashoor and his group50 investigated 102 singleton pregnancies that subsequently developed preeclampsia (PE) and compared the FT4 and TSH results to the values of 4318 normal pregnancies. They found that in late-PE (delivery after 34 wks) pregnancies, the median TSH was significantly increased and the median FT4 was decreased. Logistic regression analysis demonstrated that TSH provided a significant contribution to the prediction of late-PE.
In a study from the United Arab Emirates by Agarwal et al,51 the aim of which was to determine the prevalence of abnormal thyroid function and antithyroid antibodies during early pregnancy in a population at high risk for Gestational Diabetes Mellitus (GDM), the authors investigated, by measuring FT3, FT4 and TSH, 301 pregnant women who underwent routine “universal screening” for GDM. In 255 of the above women TPO Abs were also measured. GDM was confirmed in 80 of these women. No difference was found between the 80 women with and the 221 women without GDM for any of the above thyroid function tests. In the cohort tested for antiTPO Abs, the 51 (20%) women with positive Abs had higher mean TSH than negative women (p <0.001). Seventeen women had low FT4, while 12 women had high TSH. Twenty-eight women had low serum TSH, among whom three also had high FT4. The authors concluded that higher prevalence of hypo-T4 and TPO Abs titers than generally reported warrants routine screening for thyroid abnormalities. This screening would result in improved obstetric care.
In a study from Crete, Greece, the objective of which was to examine the association of thyroid function and autoimmunity in early pregnancy with adverse pregnancy and birth outcomes, Karakosta et al52 investigated a total of 1170 women with singleton pregnancies. They measured serum FT4, FT3, TSH and thyroid Abs in the first trimester of pregnancy. The main outcome measures included gestational diabetes, gestational hypertension/preeclampsia, cesarean section, preterm delivery, low birth weight and small-for-gestational-age neonates.
They found that the combination of high TSH and thyroid autoimmunity in early pregnancy was associated with a 4-fold increased risk for gestational diabetes and a 3-fold increased risk for low birth weight neonates after adjustment for several confounders. Women positive for thyroid Abs without elevated TSH levels in early pregnancy were at high risk for spontaneous preterm delivery, whereas the combined effect of high TSH and positive thyroid Abs did not show an association with preterm birth. They concluded that high TSH levels and thyroid autoimmunity in early pregnancy may detrimentally affect pregnancy and birth outcomes.
Haddow et al53 observed that at 9 years of age, children of women with undiagnosed hypothyroidism (TSH >98th percentile) during pregnancy had significantly lower scores in neuropsychologic tests related to intelligence, attention, language, reading ability, school performance and visual motor performance. Other studies linked SCH, AITD or hypo-T4 in the mothers with poorer results on tests of intelligence and motor skills in their children.54 The effect of maternal hypo-T4 has generated an extensive debate about its causal involvement in impaired neuropsychological development of the progeny. Although several authors55-59 have shown that maternal hypo-T4, defined as FT4 <10th percentile with a normal TSH, during the first trimester of gestation is associated with decreased neuropsychological development of the children, a recent study by Craig et al60 has not confirmed these findings. However, it has to be remembered that Morreale de Escobar et al61 already in 2000 presented epidemiological and experimental data strongly suggesting that hypo-T4 detected during the first trimester, irrespective of whether TSH was normal or elevated, was associated with a higher risk of poor neuropsychological development of the offspring, mostly due to a decreased availability of T4 to the developing fetal brain tissues.61
In 2009, Berbel et al,62 using Stringent selection criteria for the assessment of neuropsychological parameters in their cohort of patients, clearly demonstrated the relationship between hypo-T4 and impaired functional brain maturation.
Mannisto et al63recently reported that hyper- or hypothyroidism in pregnant mothers was associated with multiple adverse outcomes in the offspring, like sepsis, respiration distress syndrome, transient tachepnea, tachycardia and apnea. Changes in TSH levels in the children of mothers with TD have also been described, as children of hypothyroid mothers show significantly higher TSH levels than controls and children of hyperthyroid mothers have lower levels of TSH. It is unknown if these alterations may in the long term increase the risk of TD in these children.
An important study was published recently which provides robust data regarding the above issue. Lazarus et al64 conducted a randomized trial in which 21,846 pregnant women at 15 weeks 6 days or less gestation provided blood samples for TSH and T4 measurements. The women were assigned to a screening group (in which measurements were obtained immediately) or a control group (in which serum was stored and measurements were obtained shortly after delivery). TSH levels >97.5th percentile, FT4 <2.5th percentile or both were considered a positive screening result. Women with positive results in the screening group were assigned to 150 μg LT4 per day. The primary outcome was IQ at 3 years of age in children of women with positive results, as measured by psychologists who were unaware of the group of assignments. They concluded that antenatal screening (at a median gestational age of 12 weeks 3 days) and maternal treatment for hypothyroidism did not result in improved cognitive function in children of 3 years of age. This study supports the current ATA guidelines, which do not recommend routine antenatal screening for hypothyroidism in pregnancy.65
SCREENING FOR THYROID DISEASES: A PERMANENT CONFRONTATION BETWEEN EXPERTS
A question which is invariably raised at different meetings regarding this important issue is whether thyroid function should be screened in all pregnant women. Many major scientific societies have considered this issue. The American Association of Clinical Endocrinologists has recommended routine TSH measurements during the first trimester (or before pregnancy) in all women.66
The American Endocrine Society and all four world thyroid associations have endorsed the international guidelines recommending screening of pregnant women, especially those in high-risk groups.5
The “middle-way” position taken by the endocrine and thyroid societies resulted from a compromise within the ad hoc committee that prepared the guidelines, mainly to satisfy the opposite views held by our Ob-Gyn colleagues. In the end, the American College of Obstetricians and Gynecologists did not endorse the guidelines, considering that routine screening of thyroid function in pregnant women could not be recommended because of the lack of studies showing a proven benefit, even if asymptomatic women with SCH were identified and treated.67
The American Endocrine Society guidelines are considering high-risk categories of women justifying screening, especially those with a personal or family history of TD, symptoms of thyroid dysfunction, history of other autoimmune diseases, infertility, type 1 diabetes, history of head and neck radiation, and obviously also women with positive thyroid Abs.5 However, the study by Vaidya et al68 has shown that targeting high-risk groups is not a panacea because in the experience of these authors, targeted screening would still have missed about one third of all pregnant women with hypothyroidism.
To the question “Should women be screened for TPO antibodies before or during pregnancy with the goal of treating TPO Ab+ euthyroid women with LT4 to decrease the rate of spontaneous miscarriage”, the ATA Taskforce on Thyroid Disease during Pregnancy and Postpartum points out that there is insufficient data for or against screening all women for thyroid antibodies in the first trimester of pregnancy.65
To the question “Should women with recurrent abortion be screened for thyroid antibodies before or during pregnancy with the goal of treating TPO Abs + euthyroid women with LT4 or intravenous immunoglobulin therapy (IVIG) to decrease the rate of recurrent spontaneous abortion”, the answer is that there is also insufficient evidence to recommend for or against screening for thyroid antibodies, or treating in the first trimester of pregnancy with LT4 or IVIG, in euthyroid women with sporadic or recurrent abortion or in women undergoing in vitro fertilization (IVF).65
To the question “Should women undergoing IVF be screened for TPO antibodies before or during pregnancy”, the answer is that there is still no universal agreement regarding the mandatory screening for thyroid abnormality in pregnant women. However, there is a general agreement regarding thyroid screening in pregnant women who belong to high-risk groups.65
Most of the above statements which do not recommend universal antenatal screening for TD are based on the high cost of the programme. However, only a few studies up to now have investigated this issue in depth.
Dosiou et al,69 in their first report on the issue, developed a state-transition Markov model and performed a cost-effectiveness analysis of screening pregnant women for AITD. Three strategies were compared: 1) no screening, 2) one-time screening using antiTPO Abs and 3) one-time screening using TSH. Screening tests were added to the laboratory tests of the first prenatal visit. Abnormal screening tests were followed by further testing and subsequent thyroxine treatment of hypothyroid women. They found that screening pregnant women in the first trimester using TSH was cost-saving compared with no screening. Screening antiTPO Abs was cost-effective compared with TSH screening with an incremental cost-effectiveness ratio of $15.182 per quality-adjusted life-year. Screening using TSH remained cost-saving across a wide range of ages regarding screening, costs of treatment and probabilities of adverse outcomes.
The cost-effectiveness of antiTPO screening compared with TSH screening was mostly influenced by the probability of diagnosing hypothyroidism in unscreened subjects or subjects with a normal screening test. Screening remained highly cost-effective in scenarios where they assumed no improvement of child IQ outcomes by thyroxine treatment.
They concluded that screening all pregnant women for AITD in the first trimester is cost-effective compared with no screening.
A few years later the same first author, with different collaborators,70 published a more sophisticated report on the same issue.
The objective of the study was to compare the cost-effectiveness of universal screening of pregnant women for AITD with screening only high-risk women and with no screening.
A decision-analytic model compared the incremental cost per quality-adjusted life-year (QALY) gained among the following: 1) universal screening, 2) high-risk screening and 3) no screening. Screening consisted of a first-trimester TSH and TPO Abs measurements. Women with abnormal results underwent further testing and, when indicated, LT4 therapy. Randomized controlled trials provided probabilities for adverse obstetrical outcomes. The model accounted for the development of postpartum thyroiditis and OH. Additional scenarios in which therapy prevented cases of decreased child intelligence quotient were explored.
The results indicated that risk-based screening and universal screening were both cost-effective relative to no screening, with incremental cost-effectiveness ratios (ICERs) of $6,753/QALY and $7,138/QALY, respectively. Universal screening was cost-effective compared with risk-based screening, with an ICER of $7,258/QALY. Screening remained cost-effective in various clinical scenarios, including when only OH was assumed to have adverse obstetrical outcomes. Universal screening was cost-saving in the scenario of untreated maternal hypothyroidism resulting in decreased child intelligence, with LT4 therapy being preventive. They concluded that universal screening of pregnant women in the first trimester for AITD is cost-effective, not only compared with no screening but also compared with screening of high-risk women.
SUMMARY
TDs during pregnancy includes mainly hyper- and hypothyroidism and their variants, isolated hypo-T4, AITD and different types of goiter. AITD represents the main cause and ranges between 5 and 20% with an average of 7.8%.
TSH is a very sensitive marker of thyroid dysfunction during pregnancy. Normal values have been modified during gestation with a downward shift. The ATA recommends the upper normal range for TSH to be 2.5 mUI/ml in the first trimester and 3.0 mUI/ml for the remainder of pregnancy. Most of the studies have shown that children of pregnant women with hypothyroidism during pregnancy had significantly lower scores in neuropsychological tests related to intelligence, attention, language, reading ability, school performance and visual motor performance. However, some studies have not confirmed these findings. On the other hand, poor control of thyrotoxicosis is associated with a risk of spontaneous abortion, congestive heart failure, thyrotoxic storm, preeclampsia, preterm delivery, low birth weight and stillbirth.
An important question is whether universal screening for TD is justified or not. Despite the lack of consensus among professional organizations, recent studies which have been performed on the basis of sophisticated analyses have found that screening all pregnant women for AITD in the first trimester is cost-effective compared with no screening or with screening of high-risk women. This is in line with the everyday experience of most physicians who deal with such cases. For example, in the USA, and specifically in the area of Maine, it has been found that 76% of urban obstetric practices are investigated via TSH testing.71
REFERENCES
1. Vanderpump M, 2014 Thyroid and iodine nutritional status: a UK perspective. Clin Med 14: Suppl 6: s7-s11.
2. Zimmermann MB, 2009 Iodine deficiency. Endocr Rev 30: 376-408.
3. Glinoer D, 1997 Regulation of thyroid function in pregnancy: pathways of endocrine adaptation from physiology to pathology. Endocr Rev 18: 404-433.
4. Krassas GE, Poppe K, Glinoer D, 2010 Thyroid function and human reproductive health. Endocr Rev 31: 702-755.
5. Abalovich M, Amino N, Barbour LA, et al, 2007 Management of thyroid dysfunction during pregnancy and postpartum: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 92: S1-S47.
6. Krassas GE, Perros P, Kaprara A, 2008 Thyroid autoimmunity, infertility and miscarriage. Expert Rev Endocrinol Metab 3: 127-136.
7. Kaprara A, Krassas GE, 2008 Thyroid autoimmunity and miscarriage. Hormones (Athens) 7: 294-302.
8. Glinoer D 2005 Thyroid disease during pregnancy. In: Braverman L, Utiger R, eds. Werner’s and Ingbar’s the thyroid: a fundamental and clinical text. 9th ed, Lippincott Williams & Wilkins, Philadelphia; pp; 1086-1108.
9. Glinoer D, 2007 The importance of iodine nutrition during pregnancy. Public Health Nutr 10: 1542-1546.
10. Tortosa F, 2011 Subclinical thyroid dysfunction in pregnancy. Endocrinología y Nutrición 58: 255-257.
11. Glinoer D, Spencer CA, 2010 Serum TSH determinations in pregnancy: how, when and why? Nat Rev Endocrinol 6: 526-529.
12. Anckaert E, Poppe K, Van Uytfanghe K, Schiettecatte J, Foulon W, Thienpont LM, 2010 FT4 immunoassays may display a pattern during pregnancy similar to the equilibrium dialysis ID–LC/tandem MS candidate reference measurement procedure in spite of susceptibility towards binding protein alterations. Clinica Chimica Acta 411: 1348-1353.
13. Kahric-Janicic N, Soldin SJ, Soldin OP, West T, Gu J, Jonklaas J, 2007 Tandem mass spectrometry improves the accuracy of free thyroxine measurements during pregnancy. Thyroid 17: 303-311.
14. Mandel SJ, Spencer CA, Hollowell JG, 2005 Are detection and treatment of thyroid insufficiency in pregnancy feasible? Thyroid 15: 44-53.
15. Midgley JE, Hoermann R, 2013 Measurement of total rather than free thyroxine in pregnancy: the diagnostic implications. Thyroid 23: 259-261.
16. Lee RH, Spencer CA, Mestman JH, et al, 2009 Free T4 immunoassays are flawed during pregnancy. Am J Obstet Gynecol 200: 260-266.
17. Negro R, Schwartz A, Gismondi R, Tinelli A, Mangieri T, Stagnaro-Green A, 2010 Increased pregnancy loss rate in thyroid antibody negative women with TSH levels between 2.5 and 5.0 in the first trimester of pregnancy. J Clin Endocrinol Metab 95: E44-E48.
18. Glinoer D, de Nayer P, Bourdoux P, et al, 1990 Regulation of maternal thyroid during pregnancy. J Clin Endocrinol Metab 71: 276-287.
19. Morreale de Escobar G, Obregón MJ, Escobar del Rey F, 2000 Is neuropsychological development related to maternal hypothyroidism or to maternal hypothyroxinemia? J Clin Endocrinol Metab 85: 3975-3987.
20. Patil-Sisodia K, Mestman JH, 2010 Graves hyperthyroidism and pregnancy: a clinical update. Endocr Pract 16: 118-129.
21. Goodwin TM, Montoro M, Mestman JH, Pekary AE, Hershman JM, 1992 The role of chorionic gonadotropin in transient hyperthyroidism of hyperemesis gravidarum. J Clin Endocrinol Metab 75: 1333-1337.
22. LeBeau SO, Mandel SJ, 2006 Thyroid disorders during pregnancy. Endocrinol Metab Clin North Am 35: 117-136, vii.
23. Mestman JH, 1997 Hyperthyroidism in pregnancy. Clin Obstet Gynecol 40: 45-64.
24. Mestman JH, 2004 Hyperthyroidism in pregnancy. Best Pract Res Clin Endocrinol Metab 18: 267-288.
25. Davis LE, Lucas MJ, Hankins GD, Roark ML, Cunningham FG, 1989 Thyrotoxicosis complicating pregnancy. Am J Obstet Gynecol 160: 63-70.
26. Millar LK, Wing DA, Leung AS, Koonings PP, Montoro MN, Mestman JH, 1994 Low birth weight and preeclampsia in pregnancies complicated by hyperthyroidism. Obstet Gynecol 84: 946-949.
27. Phoojaroenchanachai M, Sriussadaporn S, Peerapatdit T, et al, 2001 Effect of maternal hyperthyroidism during late pregnancy on the risk of neonatal low birth weight. Clin Endocrinol (Oxf) 54: 365-370.
28. Sheffield JS, Cunningham FG, 2004 Thyrotoxicosis and heart failure that complicate pregnancy. Am J Obstet Gynecol 190: 211-217.
29. Smith C, Thomsett M, Choong C, Rodda C, McIntyre HD, Cotterill AM, 2001 Congenital thyrotoxicosis in premature infants. Clin Endocrinol (Oxf) 54: 371-376.
30. Haddow JE, Knight GJ, Palomaki GE, McClain MR, Pulkkinen AJ, 2004 The reference range and within-person variability of thyroid stimulating hormone during the first and second trimesters of pregnancy. J Med Screen 11: 170-174.
31. Chan GW, Mandel SJ, 2007 Therapy insight: management of Graves’ disease during pregnancy. Nat Clin Pract Endocrinol Metab 3: 470-478.
32. Momotani N, Ito K, 1991 Treatment of pregnant patients with Basedow’s disease. Exp Clin Endocrinol 97: 268-274.
33. Mandel SJ, Cooper DS, 2001 The use of antithyroid drugs in pregnancy and lactation. J Clin Endocrinol Metab 86: 2354-2359.
34. Clementi M, Di Gianantonio E, Pelo E, Mammi I, Basile RT, Tenconi R, 1999 Methimazole embryopathy: delineation of the phenotype. Am J Med Genet 83: 43-46.
35. Bournaud C, Orgiazzi J, 2003 Antithyroid agents and embryopathies. Ann Endocrinol (Paris) 64: 366-369.
36. Wolf D, Foulds N, Daya H, 2006 Antenatal carbimazole and choanal atresia: a new embryopathy. Arch Otolaryngol Head Neck Surg 132: 1009-1011.
37. Van Dijke CP, Heydendael RJ, De Kleine MJ, 1987 Methimazole, carbimazole, and congenital skin defects. Ann Int Med 106: 60-61.
38. Di Gianantonio E, Schaefer C, Mastroiacovo PP, et al, 2001 Adverse effects of prenatal methimazole exposure. Teratology 64: 262-266.
39. Barbero P, Valdez R, Rodríguez H, et al, 2008 Choanal atresia associated with maternal hyperthyroidism treated with methimazole: a case-control study. Am J Med Genet A 146A: 2390-2395.
40. Rivkees SA, Mattison DR, 2009 Ending propylthiouracil-induced liver failure in children. N Engl J Med 360: 1574-1575.
41. Rivkees SA, Mattison DR, 2009 Propylthiouracil (PTU) hepatoxicity in children and recommendations for discontinuation of use. Int J Pediatr Endocrinol 2009: 132041.
42. Karras S, Tzotzas T, Krassas GE, 2009 Antithyroid drugs used in the treatment of hyperthyroidism during breast feeding. An update and new perspectives. Hormones 8: 254-257.
43. Rivkees SA, Szarfman A, 2010 Dissimilar hepatotoxicity profiles of propylthiouracil and methimazole in children. J Clin Endocrinol Metab 95: 3260-3267.
44. Rivkees SA, Mattison DR, Cooper DS, 2009 AACE Patient Safety–Editorials Propylthiouracil (PTU) Hepatotoxicity and Graves’ Disease Therapy. http://www.aacepatientsafetyexchange.com/editorial/index.php?id=25.
45. Bahn RS, Burch HS, Cooper DS, et al, 2009 The role of propylthiouracil in the management of Graves’ disease in adults: report of a meeting jointly sponsored by the American Thyroid Association and the Food and Drug Administration. Thyroid 19: 673-674.
46. FDA Statement (Medwatch Online) 2009 Information for healthcare professionals—propylthiouracil-induced liver failure. http://www.fda.gov/Drugs/DrugSafety/ Postmarket DrugSafetyInformationforPatientsandProviders/ DrugSafetyInformationforHeathcareProfessionals/ucm162701.htm (accessed June 17, 2010)
47. Bulmus N, Ustuner I, Seda Guvendag Guven E, Kir Sahin F, Senturk S, Baydur Sahin S, 2013 Thyroid diseases in pregnancy: The importance of anamnesis. Pak J Med Sci 29: 1187-1192.
48. Medici M, Korevaar TI, Schalekamp-Timmermans S, et al, 2014 Maternal early-pregnancy thyroid function is associated with subsequent hypertensive disorders of pregnancy: the generation R study. J Clin Endocrinol Metab 99: E2591-E2598.
49. Su PY, Huang K, Hao JH, et al, 2011 Maternal thyroid function in the first twenty weeks of pregnancy and subsequent fetal and infant development: a prospective population-based cohort study in China. J Clin Endocrinol Metab 96: 3234-3241.
50. Ashoor G, Maiz N, Rotas M, Kametas NA, Nicolaides KH, 2010 Maternal thyroid function at 11 to 13 weeks of gestation and subsequent development of preeclampsia. Prenat Diagn 30: 1032-1038.
51. Agarwal MM, Dhatt GS, Punnose J, Bishawi B, Zayed R, 2006 Thyroid function abnormalities and antithyroid antibody prevalence in pregnant women at high risk for gestational diabetes mellitus. Gynecol Endocrinol 22: 261-266.
52. Karakosta P, Alegakis D, Georgiou V, et al, 2012 Thyroid dysfunction and autoantibodies in early pregnancy are associated with increased risk of gestational diabetes and adverse birth outcomes. J Clin Endocrinol Metab 97: 4464-4472.
53. Haddow JE, Palomaki GE, Allan WC, et al, 1999 Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. New England Journal of Medicine 341: 549-555.
54. Li Y, Shan Z, Teng W, et al, 2010 Abnormalities of maternal thyroid function during pregnancy affect neuropsychological development of their children at 25-30 months. Clin Endocrinol 72: 825-829.
55. Pop VJ, Kuijpens JL, van Baar AL, et al, 1999 Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin Endocrinol 50: 149-155.
56. Henrichs J, Bongers-Schokking JJ, Schenk JJ, et al, 2010 Maternal thyroid function during early pregnancy and cognitive functioning in early childhood: the generation R study. J Clin Endocrinol Metab 95: 4227-4234.
57. Kooistra L, Crawford S, van Baar AL, Brouwers EP, Pop VJ 2006 Neonatal effects of maternal hypothyroxinemia during early pregnancy. Pediatrics 117: 161-167.
58. Finken MJ, van Eijsden M, Loomans EM, Vrijkotte TG, Rotteveel J, 2013 Maternal hypothyroxinemia in early pregnancy predicts reduced performance in reaction time tests in 5- to 6-year-old offspring. J Clin Endocrinol Metab 98: 1417-1426.
59. Julvez J, Alvarez-Pedrerol M, Rebagliato M, et al, 2012 Thyroxine levels during pregnancy in healthy women and early child neurodevelopment. Epidemiology 24: 150-157.
60. Craig WY, Allan WC, Kloza EM, et al, 2012 Mid-gestational maternal free thyroxine concentration and offspring neurocognitive development at age two years. J Clin Endocrinol Metab 97: E22-E28.
61. Morreale de Escobar G, Obregón MJ, Escobar del Rey F, 2000 Is neuropsychological development related to maternal hypothyroidism or to maternal hypothyroxinemia? J Clin Endocrinol Metab 85: 3975-3987.
62. Berbel P, Mestre JL, Santamaria A, et al, 2009 Delayed neurobehavioral development in children born to pregnant women with mild hypothyroxinemia during the first month of gestation: the importance of early iodine supplementation. Thyroid 19: 511-519.
63. Mannisto T, Mendola P, Reddy U, Laughon SK, 2013 Neonatal outcomes and birth weight in pregnancies complicated by maternal thyroid disease. American J Epidemiol 178: 731-740.
64. Lazarus JH, Bestwick JP, Channon S, et al, 2012 Antenatal thyroid screening and childhood cognitive function. N Engl J Med 366: 493-501.
65. Stagnaro-Green A, Abalovich M, Alexander E, et al, 2011 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid 21: 1081-125.
66. Baskin HJ, Cobin RH, Duick DS, et al, 2002 American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocr Pract 8: 457-469.
67. ACOG news release 2007 Routine thyroid screening not recommended for pregnant women. www.acog.org/from_home/publications/press_releases/nr10-01-07-1.cfm
68. Vaidya B, Anthony S, Bilous M, et al, 2007 Detection of thyroid dysfunction in early pregnancy: universal screening or targeted high-risk case finding? J Clin Endocrinol Metab 92: 203-207.
69. Dosiou C, Sanders GD, Araki SS, Crapo LM, 2008 Screening pregnant women for autoimmune thyroid disease: a cost-effectiveness analysis. Eur J Endocrinol 158: 841-851.
70. Dosiou C, Barnes J, Schwartz A, Negro R, Crapo L, Stagnaro-Green A, 2012 Cost-effectiveness of universal and risk-based screening for autoimmune thyroid disease in pregnant women. J Clin Endocrinol Metab 97: 1536-1546.
71. Haddow JE, McClain MR, Palomaki GE, Kloza EM, Williams J, 2006 Screening for thyroid disorders during pregnancy: results of a survey in Maine. Am J Obstet Gynecol 194: 471-474.
72. Stricker R, Echenard M, Eberhart R, et al, 2007 Evaluation of maternal thyroid function during pregnancy: the importance of using gestational age-specific reference intervals. Eur J Endocrinol 157: 509-514.
73. Panesar NS, Li CY, Rogers MS, 2001 Reference intervals for thyroid hormones in pregnant Chinese women. Ann Clin Biochem 38: 329-332.
Address for correspondence:
Prof. Gerasimos Krassas MD, PhD, FRCP (Lond), 92 Tsimiski Str., 54622 Thessaloniki, Greece, E-mail: gkra34@otenet.gr
Received:22-05-2014, Accetpted:13-01-2015