1Second Medical Clinic, Ippokration Hospital; 2Unit of Reproductive Endocrinology, First Department of Obstetrics and Gynecology, Papageorgiou Hospital; 3Division of Endocrinology and Human Reproduction, Second Department of Obstetrics and Gynecology, Ippokration Hospital; Aristotle University of Thessaloniki, Thessaloniki, Greece
OBJECTIVE: Insulin resistance contributes to the pathogenesis of both polycystic ovary syndrome (PCOS) and non-alcoholic fatty liver disease (NAFLD). The main aim of the present study was the evaluation of non-invasive indices of hepatic steatosis and fibrosis in PCOS women with or without metabolic syndrome (MetS).
DESIGN: In this cross-sectional study, three non-invasive indices for hepatic steatosis [NAFLD liver fat score, lipid accumulation product (LAP) and hepatic steatosis index (HIS)] and four for fibrosis [FIB-4, aspartate aminotransferase (AST)-to-Platelet Ratio Index (APRI), body mass index (BMI)-Age-Alanine aminotransferase (ALT)-Triglycerides (BAAT) and BMI AST/ALT Ratio Diabetes (BARD)] were calculated in 314 PCOS women (77 with, 237 without MetS) and 78 controls.
RESULTS: All steatosis indices were significantly higher in the PCOS than the control group (NAFLD liver fat score: -0.139±0.117 vs. -0.976±0.159, p<0.001; LAP: 43.3±1.9 vs. 34.7±3.1, p=0.036; HIS: 44.6±0.5 vs. 42.1±0.8, p=0.016). FIB-4 and BAAT [fibrosis stage (F)2-4] were higher in the PCOS group (0.480±0.020 vs. 0.400±0.013, p<0.001; and 15.6% vs. 5.1%, respectively), whereas APRI and BARD were not. All steatosis indices were significantly higher in PCOS women with than without MetS (NAFLD liver fat score: 1.874±0.258 vs. -0.793±0.099, p<0.001; LAP: 76.8±4.9 vs. 33.4±1.4, p<0.001; and HIS: 49.8±1 vs. 43±0.5, p<0.001). Of the fibrosis indices, only BAAT (F2-4: 50.6% vs. 4.2%) was higher in PCOS women with MetS.
CONCLUSIONS: Non-invasive indices of hepatic steatosis were significantly higher in PCOS, especially in the presence of MetS, whereas indices of hepatic fibrosis yielded controversial results. Further studies are warranted to evaluate the long-term outcomes of hepatic steatosis and fibrosis indices in PCOS women.
APRI, FIB-4, Hepatic steatosis index, Insulin resistance, Lipid accumulation product, Metabolic syndrome, NAFLD liver fat score, Steatosis
INTRODUCTION
Polycystic ovary syndrome (PCOS) is the commonest endocrine disorder in women of reproductive age,1 its prevalence having been reported to be up to 18% in accordance with the ESHRE/ASRM criteria.2 Multiple metabolic aberrations, such as insulin resistance (IR) and hyperinsulinemia, visceral obesity, inflammation and endothelial dysfunction, hypertension, dyslipidemia, high incidence of impaired glucose tolerance and a lifetime risk of type 2 diabetes mellitus (T2DM) and cardiovascular diseases are associated with the syndrome.3
Non-alcoholic fatty liver disease (NAFLD), the commonest hepatic disorder and the leading cause of cryptogenic cirrhosis in Western countries, constitutes a major, global, public health problem.4 Its prevalence has been reported to be 10-46% and 6-35% in the US and the rest of the world, respectively,5 and it increases mortality mainly due to hepatic and cardiovascular disease.6
Although the pathogenesis of either disorder has not yet been fully elucidated, IR has been implicated, at least partly, in the pathogenesis of both diseases.2,7-8 IR has been proposed as the common pathogenetic mechanism in conditions clustered under the term of metabolic syndrome (MetS), including obesity, T2DM, dyslipidemia, hypertension, PCOS and NAFLD, all of which increase the risk for the abovementioned cardiovascular diseases and mortality, the endpoints of MetS.8 Based on the observation that IR is the common underlying factor, studies evaluating the association between PCOS and NAFLD are increasing and are systematically summarized elsewhere.9 It seems that women with PCOS are possibly at risk for developing NAFLD and, conversely, NAFLD may be a risk factor for PCOS.9 However, the majority of relevant studies to date are not based on liver biopsy, regarded as the gold standard for the diagnosis of NAFLD,10 because it raises obvious ethical issues. Therefore, the use of non-invasive indices of hepatic injury is gaining increasing interest, as they offer an estimation of NAFLD stage and grade and thus may serve as accurate tools, at least for selection of patients for liver biopsy.11
The primary endpoints of this study were the evaluation of non-invasive indices of hepatic steatosis and fibrosis in women: a) with PCOS vs. without PCOS; and b) with PCOS associated or not with MetS. The secondary endpoint of this study was the association of non-invasive indices of hepatic steatosis and fibrosis with variables related to sex hormones or IR.
PATIENTS AND METHODS
Patients
This was a one-center, cross-sectional study. Data were prospectively collected based on protocols of previous studies,7,12-13 but retrospectively reviewed for this study. Premenopausal women with PCOS and apparently healthy female controls were recruited on an outpatient basis at the Gynecological Endocrinology Infirmary of the Second Department of Obstetrics and Gynecology, Aristotle University of Thessaloniki, Greece. Women with PCOS were referred to the Gynecological Endocrinology Infirmary for diagnostic evaluation and/or treatment; controls were healthy volunteers. All participants provided informed consent. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the institutional review board.
The inclusion criteria for the PCOS group were based on the revised criteria of ESHRE/ASRM, which require the presence of at least two of the following three features: 1) oligo- or anovulation (<8 spontaneous hemorrhagic episodes/year); 2) biochemical hyperandrogenemia [defined in our population as early follicular phase serum total testosterone >2.1 nmol/L, corresponding to the mean + 2 standard deviations (SD) testosterone concentrations in 200 control subjects measured in our laboratory] or clinical manifestations of hyperandrogenemia; and 3) polycystic ovary morphology on ultrasound (≥12 small follicles in at least one ovary and/or ovarian volume >10 cm3).2 According to this definition, PCOS can be subdivided into four different phenotypes of PCOS: 1) severe PCOS (when all three criteria are included); 2) oligo- or anovulation and hyperandrogenemia; 3) ovulatory PCOS (hyperandrogenemia and polycystic ovary morphology); and 4) mild PCOS (oligo- or anovulation and polycystic ovary morphology).1
The PCOS group was further subdivided based on the presence of MetS according to the definitions proposed by the International Diabetes Federation (IDF). The reason for choosing this definition was that the same definition was applied for the estimation of the NAFLD liver fat score, which is one of the non-invasive indices used in this study.
Exclusion criteria for both PCOS and control groups were: 1) congenital adrenal hyperplasia (a short Synachten test was performed in all women with basal serum 17α-hydroxyprogesterone concentrations >1.5 ng/mL); 2) Cushing’s syndrome; 3) galactorrhea; 4) androgen-secreting tumors; 5) ethanol consumption >20 g/day; 6) history of liver cirrhosis or other liver disease (viral hepatitis, autoimmune hepatitis, primary sclerosing cholangitis, primary biliary cirrhosis, drug-induced liver disease, hemochromatosis, Wilson’s disease, α1-antitrypsin deficiency); 7) type 1 diabetes mellitus; 8) uncontrolled hypothyroidism or hyperthyroidism; 9) adrenal insufficiency; 10) renal failure; 11) cancer; 12) pregnancy; 13) premature ovarian failure; 13) addiction to any drug; 14) use of the following medications within the last semester before screening: estrogens, androgens, anti-androgens, progestins, glucocorticosteroids, spironolactone, insulin, thiazolidinediones, ursodeoxycholic acid, ferrum, interferon, tamoxifene, amiodarone, biologic agents, any medication against tuberculosis, epilepsy or viruses, or any medication affecting hemostasis, such as antiplatelet agents or oral anticoagulants.
Methods
In all women, weight, height, waist circumference (WC) and hip circumference (HC) were measured. Baseline blood samples were collected after an overnight fast between days 3 and 7 of the menstrual cycle in the control group and after a spontaneous bleeding episode in the PCOS group. The serum concentrations of follicle stimulating hormone (FSH), luteinizing hormone (LH), prolactin, total testosterone, Δ4-androstenedione, dehydroepiandrosterone-sulphate (DHEAS), sex hormone-binding globulin (SHBG), insulin, glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C), triglycerides, uric acid, aspartate transaminase (AST), alanine transaminase (ALT), gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP) and platelets count were measured. The methodology used for serum measurements has been previously described in detail.13 The same experienced sonographer performed transvaginal ultrasonography on the same day as the blood was drawn; the volume and the number of small follicles (measuring 2–9 mm in diameter) of each ovary were determined.
The diagnoses of T2DM or impaired fasting glucose (IFG) were based on the American Diabetes Association (ADA) definition; T2DM: either fasting plasma glucose ≥7 mmol/L or 2-h plasma glucose ≥11 mmol/L during an oral glucose tolerance test (75 g anhydrous glucose dissolved in water), which was performed in all the participants; IFG: fasting plasma glucose 5.5-6.9 mmol/L.
Body mass index (BMI) was calculated by the formula: body weight (kg)/height2 (m2). The waist-to-hip ratio (WHR), LH-to-FSH ratio and AST-to-ALT ratio were also calculated. Low-density lipoprotein cholesterol (LDL-C) was estimated using the Friedewald formula. Free androgen index (FAI) was calculated by the formula: FAI = total testosterone (nmol/L)/SHBG (nmol/L) X 100. IR, β-cell function and insulin sensitivity were quantified by the homeostatic model of assessment (HOMA)-IR, HOMA-β and quantitative insulin sensitivity check index (QUICKI), respectively, by the formulas: HOMA-IR = fasting glucose (mmol/L) x fasting insulin (μU/mL) / 22.5; HOMA-β = [20 * fasting insulin (μU/mL)] / [fasting glucose (mmol/L) – 3.5); QUICKI = 1/(log(fasting insulin; μU/mL) + log[fasting glucose; mg/dL)].
Based on the available data, three non-invasive indices for hepatic steatosis [NAFLD liver fat score,14 lipid accumulation product (LAP)15 and hepatic steatosis index (HIS)16] and four indices for hepatic fibrosis [FIB-4,17 AST-to-Platelet Ratio Index (APRI),18 BMI Age ALT Triglycerides (BAAT)19 and BMI Age ALT Triglycerides (BARD)20] were estimated for all women with PCOS and controls. The required parameters and equations for each of these indices are presented in detail in Table 1.
Statistical Analysis
Continuous data were presented as mean ± standard error of the mean (SEM). Categorical data were presented as frequencies. The Kolmogorov-Smirnov test was used to check the normality of distributions of continuous variables. The independent sample T-test or Mann-Whitney test was used for between-group comparisons in cases of two groups of continuous variables. One-way analysis of variance (ANOVA) or the Kruskal-Wallis test (with Bonferroni post-hoc adjustment) was used for between-group comparisons in cases of more than two groups of continuous variables. Analysis of covariance (ANCOVA) was used to adjust between-group comparisons for covariates. The chi-square or Fisher exact test was used for between-group comparisons of categorical variables. Spearman’s coefficient (rs) was used for binary correlations. Statistical analysis was performed with SPSS 21.0 for Macintosh (IBM Corp., Armonk, NY). Significance was set at a level of p<0.05.
RESULTS
Comparison between women with PCOS and controls
Three hundred and fourteen women with PCOS and 78 controls were included in this study. The PCOS group was divided into: 1) severe (n=164); 2) oligo- or anovulation and hyperandrogenemia (n=89); 3) ovulatory (n=30); and 4) mild PCOS (n=31). Comparative data of the study groups are presented in Table 2. The control group was of significantly higher age compared with the PCOS group. As expected, BMI, WC and HC, testosterone, DHEAS, Δ4-androstenedione and FAI, LH, LH to FSH ratio were significantly higher, whereas SHBG and FSH were lower in the PCOS compared with the control group. Regarding liver function tests, ALP was significantly higher in the PCOS compared with the control group, whereas AST, ALT, GGT and AST to ALT ratio were similar between groups. Total and HDL-cholesterol were significantly higher in the control group, whereas LDL-C and triglycerides were not different between groups. Interestingly, serum uric acid was significantly higher in the PCOS compared with the control group. Regarding IR, insulin, HOMA-IR, HOMA-β and frequency of MetS were significant higher, whereas QUICKI was lower in the PCOS compared with the control group (Table 2).
Regarding non-invasive indices, data for continuous ones are presented in Table 2 and Figure 1 and those for categorical (BAAT and BARD) in Table 3. All indices of hepatic steatosis (NAFLD liver fat score, LAP and HIS) were significantly higher in the PCOS than the control group (Table 2; Figure 1). Regarding hepatic fibrosis, FIB-4 and BAAT were higher in the PCOS group, whereas APRI and BARD were not (Tables 2 and 3). In any case, based on the indices of hepatic fibrosis, only a minority of PCOS women had advanced fibrosis: according to BAAT, there were only two (0.6%) PCOS women at stage 3 and none at stage 4, whereas, according to BARD, only three (1%) PCOS women were at stage 4. By applying the thresholds for advanced fibrosis (F3-F4) in FIB-4 and APRI, two (0.6%) and four (1.3%) PCOS women, respectively, were expected to have advanced fibrosis and no women in the control group.
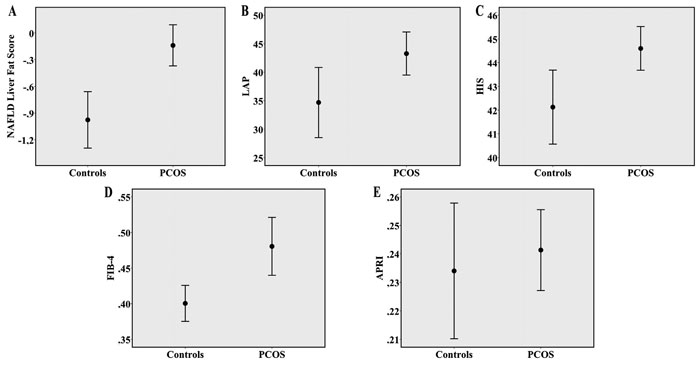
Figure 1. Error bars (mean and 95% Confidence Interval) depicting: (A) NAFLD liver fat score; (B) LAP; (C) HIS; (D) FIB-4 and (E) APRI scores between PCOS women and controls. APRI: AST to Platelet Ratio Index; AST: aspartate transaminase; HIS: hepatic steatosis index; LAP: lipid accumulation product; NAFLD: non-alcoholic fatty liver disease.
When patients with mild PCOS (without hyperandrogenemia; n=31) were excluded, the results were essentially unchanged for all indices. Notably, statistical significance did not change when continuous non-invasive indices were adjusted for age (Table 4). FIB-4 was not adjusted for age as the latter was included in its calculation.
Correlations between the continuous non-invasive indices and parameters related to sex hormones and IR are summarized in Table 5.
Comparison between women with and without MetS
Comparative data between PCOS women with (n=77) and without (n=237) MetS groups are presented in Table 6. Age was essentially similar between groups. As expected, data regarding anthropometric, metabolic (lipid profile and uric acid), IR and sex hormone (testosterone, SHBG and FAI) parameters were more favorable in PCOS women without MetS.
Regarding the liver function tests, ALT, AST, GGT, but not ALP and AST to ALT ratio, were higher in PCOS women with MetS. All indices of hepatic steatosis (NAFLD liver fat score, LAP and HIS) were significantly higher in PCOS patients with than without MetS (Table 6). Regarding hepatic fibrosis, only BAAT was higher in PCOS patients with MetS, whereas FIB-4, APRI and BARD were similar (Tables 6 and 7). When patients with mild PCOS (without hyperandrogenemia; n=31) were excluded, the results were essentially unchanged for all indices. Notably, statistical significance did not change when continuous non-invasive indices were adjusted for age (data not shown).
Similarly, when the comparison was performed among four groups [control/non-MetS (n=68) vs. control/MetS (n=10) vs. PCOS/non-Mets (237) vs. PCOS/MetS (n=77)], all indices of hepatic steatosis showed a trend towards higher values when moving towards PCOS and MetS [NAFLD liver fat score: p<0.001; LAP: p<0.001; HIS: p<0.001].
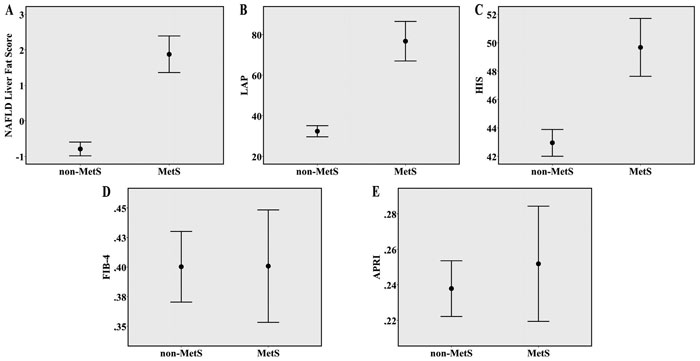
Figure 2. Error bars (mean and 95% Confidence Interval) depicting: (A) NAFLD liver fat score; (B) LAP; (C) HIS; (D) FIB-4 and (E) APRI scores between PCOS patients with and without MetS. APRI: AST to Platelet Ratio Index; AST: aspartate transaminase; HIS: hepatic steatosis index; LAP: lipid accumulation product; MetS: metabolic syndrome; NAFLD: non-alcoholic fatty liver disease.
DISCUSSION
To our knowledge, this is the first study reporting data on combined non-invasive indices of hepatic steatosis and fibrosis in women with PCOS, which are of relatively low cost and can be calculated on a routine basis. Indices of hepatic steatosis (NAFLD liver fat score, LAP and HIS) were significantly higher in the PCOS than the control group, as well as in PCOS women with than without MetS. However, regarding indices of hepatic fibrosis, FIB-4 and BAAT, but not APRI and BARD, were higher in the PCOS than the control group. In PCOS women with MetS, only BAAT was higher compared with PCOS women without MetS. When non-invasive indices were adjusted for age, the statistical significance remained essentially unchanged for all comparisons.
The results of this series seem to be clear for indices of hepatic steatosis, but not for those of fibrosis. This discordance in fibrosis indices may be partly attributed to the low rates of hepatic fibrosis expected in this cohort of relatively young women (26.1±0.4 years) with a low rate of T2DM (1.9%) and relatively low concentrations of triglycerides (1.11±0.03 mmol/L). As shown, only a minority of women with PCOS in this cohort were expected to have advanced fibrosis according to the thresholds of all fibrosis indices. However, the high rates of hepatic steatosis, together with higher IR (HOMA-IR, HOMA-β, QUICKI) and obesity (BMI, WC) in their third decade, may possibly render PCOS women at higher risk for hepatic inflammation and fibrosis later in their life, given that approximately 15-30% of patients with non-alcoholic simple steatosis evolve to non-alcoholic steatohepatitis (NASH).6 Simple steatosis consists mainly of steatosis, whereas NASH in addition to steatosis features progressive inflammation and fibrosis;6 this distinction is of importance, since simple steatosis progresses to cirrhosis in less than 5% of cases, whereas NASH progresses to cirrhosis in 10-15% of cases over 10 years and in 25-30% of cases in the presence of advanced fibrosis.5,21 Furthermore, NASH, but not simple steatosis, predisposes to hepatocellular carcinoma.5,21
Regarding the indices of hepatic steatosis, similarly to our findings other studies have reported higher rates of ultrasound-proven hepatic steatosis in PCOS (31-73%) than non-PCOS controls (18-47%).22-25 Steatosis was also shown to be positively associated with HOMA-IR23,26 and FAI23 but negatively with SHBG,23 as in our study. Hepatic fat, quantified by proton-magnetic resonance spectroscopy, was also found to be higher in women with PCOS (n=29) than in controls (n=22).27 Interestingly, hyperandrogenic women with PCOS (n=19) had higher IR and hepatic fat than non-hyperandrogenic ones (n=10).27 Likewise, in our study, women with PCOS with MetS had higher IR, higher testosterone concentrations and FAI and higher rates of indices for hepatic steatosis compared with PCOS women without MetS.
Contrary to our findings and existing literature, there is one study of 17 lean women with PCOS (with normal aminotransferases) and 17 controls in whom hepatic steatosis was not detected (by ultrasound and computed tomography) in any woman.28 The reasons for this inconsistency are not well understood, but the selection bias (lean women with normal liver function tests) may be a plausible cause. However, although obesity certainly predisposes to NAFLD,8 high rates of NAFLD (approximately 40%) have been reported even in lean and young PCOS women.26,29
Data regarding hepatic fibrosis in women with PCOS are rather scarce because this requires liver biopsy which has to date been performed only on a very limited number of patients in relevant studies. In one study, six (of a total of 200 retrospectively evaluated) women with PCOS and persistent aminotransferase elevation underwent liver biopsy: all had NASH with fibrosis (3%).30 This low fibrosis rate is in accordance with our findings, based on non-invasive fibrosis indices. By contrast, in another study partly based on liver biopsy, women with PCOS (n=34; 25 subjected to liver biopsy; 11 with NASH) had a non-statistical trend towards higher rates for NASH than non-PCOS controls (n=32; 25 subjected to liver biopsy; 5 with NASH).31 Different rates of NASH between these studies may possibly reflect population differences and/or selection bias; further, large-scale studies are needed to clarify the prevalence of NASH in women with PCOS. However, irrespective of the rate of NASH, some women with PCOS have advanced fibrosis, even early in their lives, and they may benefit if NASH is diagnosed. Persistent aminotransferase elevation alone may provide an indication, but combined non-invasive indices (some of which contain AST and/or ALT) are currently regarded as more sensitive tools to select these patients for liver biopsy.11
Data evaluating the reverse issue, being the rates of PCOS in NAFLD populations, are currently rather limited. In one study, 10 of 14 (71%) premenopausal women with NAFLD (biopsy-proven in 7, ultrasound-proven in 7) were reported to have PCOS.32 In another study, patients with biopsy-proven simple steatosis and PCOS (n=12) had higher serum cytokeratin-18 (M30) compared with those with simple steatosis but without PCOS (n=12), despite similar serum liver function tests, serum lipids and IR.31 However, the small sample size of both studies possibly resulted in low statistical power.
Indices of hepatic steatosis correlated with each other (Table 5). FIB-4 also correlated with APRI, NAFLD liver fat score and HIS, but not LAP. APRI did not correlate with indices of hepatic steatosis. All indices of hepatic steatosis correlated with insulin and IR in this study; FIB-4 also correlated with insulin and IR, although less strongly. This was expected given that IR contributes to the pathogenesis of hepatic steatosis, but also, possibly to a lower degree, to the progression from the hepatic steatosis to NASH, which usually features fibrosis.8 Furthermore, all indices of hepatic steatosis correlated positively with testosterone and FAI but inversely with SHBG. Interestingly, testosterone, DHEAS, Δ4-androstenedione and FAI inversely correlated with FIB-4. Likewise, higher concentrations of calculated free testosterone, bioavailable testosterone and FAI, whereas lower concentrations of SHBG, were shown in postmenopausal women with NAFLD than age- and BMI-matched controls.33 Although correlations cannot prove a cause-effect association, it could be speculated that hyperandogenemia, which is usually observed in women with PCOS, may play a permissive role in the pathogenesis of hepatic steatosis but a potential protective role in the progression to NASH. This hypothesis is in agreement with low fibrosis rates in PCOS women observed in this study, but requires validation from studies of different design.
This study has certain strengths and limitations. It is a relatively large-scale study and the first based on seven different non-invasive indices of hepatic steatosis and fibrosis. On the other hand, the limitations of the study are the following. 1) Data were retrospectively reviewed for the need of this study; however, they had been previously prospectively and systematically recorded for the need of other studies. 2) The participants were not subjected to liver biopsy, regarded as the diagnostic gold standard;34 however, the performance of liver biopsy in young women with low or no burden of metabolic co-morbidity (i.e., T2DM, dyslipidemia) and low hepatic fibrosis risk raises obvious ethical considerations. 3) Other liver diseases were excluded on the basis of history and self-reporting and no specific tests were used for this aim; however, the rates of other liver diseases in this cohort of young women are expected to be very low. 4) The control group were of higher age than the PCOS group; however, although the prevalence of NAFLD increases with increasing age,5 women with PCOS had higher non-invasive indices, especially those related to hepatic steatosis, but also to FIB-4 and BAAT (which also increase with increasing age), even if they were of lower age than controls. In any case, adjustment for age did not change the results. 5) The control group had lower BMI and WC than the PCOS group; however, most of the presenting non-invasive indices (with the exemption of FIB-4 and APRI) include a variable of adiposity (i.e., BMI, WC or metabolic syndrome), which implies a type of internal adjustment. 6) Data for GGT should be cautiously interpreted because they were available only for a subgroup of participants (66.7% of the control and 45.9% of the PCOS group, respectively). 7) Other non-invasive indices of hepatic steatosis or fibrosis [i.e., fatty liver index (FLI), NAFLD fibrosis score, etc] were not calculated because at least one of the required parameters for their estimation was not available or was available only for a subset of the participants. 8) Liver ultrasound was not performed in this study, mainly due to its retrospective nature; however, the sensitivity (60-94%) and specificity (66-100%) of liver ultrasound varies, being lower in obesity,35 which usually co-exists with PCOS and NAFLD. In addition, it has low inter- and intra-observer variability and, most importantly, the ultrasound cannot provide information about hepatic fibrosis.35
In conclusion, indices of hepatic steatosis (NAFLD liver fat score, LAP and HIS) were all significantly higher in the PCOS than the control group, as well as in PCOS women with rather than without MetS in this study, indicating a common link, especially in the presence of MetS. Regarding indices of hepatic fibrosis, controversial results were retrieved, possibly because of the expected low rates of hepatic fibrosis in this series of young women with low relevant co-morbidity. However, despite the low rate of hepatic fibrosis, the selection of these PCOS women with high NASH probability would be of paramount importance, since these women are expected to benefit the most from histological validation and subsequent lifestyle and pharmacological interventions. Further studies are warranted to validate hepatic steatosis and fibrosis indices in PCOS populations and to evaluate the long-term outcomes of these indices in PCOS women.
FUNDING
This study did not receive any funding.
DISCLOSURE STATEMENT
There is no conflict of interest related to this manuscript.
REFERENCES
1. Panidis D, Tziomalos K, Papadakis E, Vosnakis C, Chatzis P, Katsikis I, 2013 Lifestyle intervention and anti-obesity therapies in the polycystic ovary syndrome: impact on metabolism and fertility. Endocrine 44: 583-590.
2. Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group, 2004 Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 19: 41-47.
3. Carmina E, 2012 PCOS: metabolic impact and long-term management. Minerva Ginecol 64: 501-505.
4. Sanyal AJ, 2011 NASH: A global health problem. Hepatol Res 41: 670-674.
5. Vernon G, Baranova A, Younossi ZM, 2011 Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 34: 274-285.
6. Polyzos SA, Kountouras J, Zavos C, Deretzi G, 2012 Nonalcoholic fatty liver disease: multimodal treatment options for a pathogenetically multiple-hit disease. J Clin Gastroenterol 46: 272-284.
7. Panidis D, Macut D, Tziomalos K, et al, 2013 Prevalence of metabolic syndrome in women with polycystic ovary syndrome. Clin Endocrinol (Oxf) 78: 586-592.
8. Polyzos SA, Kountouras J, Zavos C, 2009 Nonalcoholic fatty liver disease: the pathogenetic roles of insulin resistance and adipocytokines. Curr Mol Med 9: 299-314.
9. Baranova A, Tran TP, Birerdinc A, Younossi ZM, 2011 Systematic review: association of polycystic ovary syndrome with metabolic syndrome and non-alcoholic fatty liver disease. Aliment Pharmacol Ther 33: 801-814.
10. Chalasani N, Younossi Z, Lavine JE, et al, 2012 The diagnosis and management of non-alcoholic fatty liver disease: practice Guideline by the American Association for the study of liver diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 55: 2005-2023.
11. Machado MV, Cortez-Pinto H, 2013 Non-invasive diagnosis of non-alcoholic fatty liver disease. A critical appraisal. J Hepatol 58: 1007-1019.
12. Panidis D, Tziomalos K, Misichronis G, et al, 2012 Insulin resistance and endocrine characteristics of the different phenotypes of polycystic ovary syndrome: a prospective study. Hum Reprod 27: 541-549.
13. Piouka A, Farmakiotis D, Katsikis I, Macut D, Gerou S, Panidis D, 2009 Anti-Mullerian hormone levels reflect severity of PCOS but are negatively influenced by obesity: relationship with increased luteinizing hormone levels. Am J Physiol Endocrinol Metab 296: E238-243.
14. Kotronen A, Peltonen M, Hakkarainen A, et al, 2009 Prediction of non-alcoholic fatty liver disease and liver fat using metabolic and genetic factors. Gastroenterology 137: 865-872.
15. Bedogni G, Kahn HS, Bellentani S, Tiribelli C, 2010 A simple index of lipid overaccumulation is a good marker of liver steatosis. BMC Gastroenterol 10: 98.
16. Lee JH, Kim D, Kim HJ, et al, 2010 Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis 42: 503-508.
17. McPherson S, Stewart SF, Henderson E, Burt AD, Day CP, 2010 Simple non-invasive fibrosis scoring systems can reliably exclude advanced fibrosis in patients with non-alcoholic fatty liver disease. Gut 59: 1265-1269.
18. Kruger FC, Daniels CR, Kidd M, et al, 2011 APRI: a simple bedside marker for advanced fibrosis that can avoid liver biopsy in patients with NAFLD/NASH. S Afr Med J 101: 477-480.
19. Ratziu V, Giral P, Charlotte F, et al, 2000 Liver fibrosis in overweight patients. Gastroenterology 118: 1117-1123.
20. Harrison SA, Oliver D, Arnold HL, Gogia S, Neuschwander-Tetri BA, 2008 Development and validation of a simple NAFLD clinical scoring system for identifying patients without advanced disease. Gut 57: 1441-1447.
21. Musso G, Gambino R, Cassader M, Pagano G, 2011 Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med 43: 617-649.
22. Cerda C, Perez-Ayuso RM, Riquelme A, et al, 2007 Nonalcoholic fatty liver disease in women with polycystic ovary syndrome. J Hepatol 47: 412-417.
23. Vassilatou E, Lafoyianni S, Vryonidou A, et al, 2010 Increased androgen bioavailability is associated with non-alcoholic fatty liver disease in women with polycystic ovary syndrome. Hum Reprod 25: 212-220.
24. Qu ZY, Shi YH, Zhao DN, Jiang JJ, Ma ZX, Chen ZJ, 2010 Effect of obesity on nonalcoholic fatty liver disease in Chinese women with polycystic ovary syndrome. Zhonghua Yi Xue Za Zhi 90: 2036-2039.
25. Zueff LF, Martins WP, Vieira CS, Ferriani RA, 2012 Ultrasonographic and laboratory markers of metabolic and cardiovascular disease risk in obese women with polycystic ovary syndrome. Ultrasound Obstet Gynecol 39: 341-347.
26. Gambarin-Gelwan M, Kinkhabwala SV, Schiano TD, Bodian C, Yeh HC, Futterweit W, 2007 Prevalence of nonalcoholic fatty liver disease in women with polycystic ovary syndrome. Clin Gastroenterol Hepatol 5: 496-501.
27. Jones H, Sprung VS, Pugh CJ, et al, 2012 Polycystic ovary syndrome with hyperandrogenism is characterized by an increased risk of hepatic steatosis compared to nonhyperandrogenic PCOS phenotypes and healthy controls, independent of obesity and insulin resistance. J Clin Endocrinol Metab 97: 3709-3716.
28. Markou A, Androulakis, II, Mourmouris C, et al, 2010 Hepatic steatosis in young lean insulin resistant women with polycystic ovary syndrome. Fertil Steril 93: 1220-1226.
29. Ciotta L, Pagano I, Stracquadanio M, Formuso C, 2011 Polycystic ovarian syndrome incidence in young women with non-alcoholic fatty liver disease. Minerva Ginecol 63: 429-437.
30. Setji TL, Holland ND, Sanders LL, Pereira KC, Diehl AM, Brown AJ, 2006 Nonalcoholic steatohepatitis and nonalcoholic Fatty liver disease in young women with polycystic ovary syndrome. J Clin Endocrinol Metab 91: 1741-1747.
31. Baranova A, Tran TP, Afendy A, et al, 2013 Molecular signature of adipose tissue in patients with both Non-alcoholic fatty liver disease (NAFLD) and polycystic ovarian syndrome (PCOS). J Transl Med 11: 133.
32. Brzozowska MM, Ostapowicz G, Weltman MD, 2009 An association between non-alcoholic fatty liver disease and polycystic ovarian syndrome. J Gastroenterol Hepatol 24: 243-247.
33. Polyzos SA, Kountouras J, Tsatsoulis A, et al, 2013 Sex steroids and sex hormone-binding globulin in postmenopausal women with nonalcoholic fatty liver disease. Hormones (Athens) 12: 405-416.
34. Sanyal AJ, Brunt EM, Kleiner DE, et al, 2011 Endpoints and clinical trial design for non-alcoholic steatohepatitis. Hepatology 54: 344-353.
35. Schwenzer NF, Springer F, Schraml C, Stefan N, Machann J, Schick F, 2009 Non-invasive assessment and quantification of liver steatosis by ultrasound, computed tomography and magnetic resonance. J Hepatol 51: 433-445.
Address for correspondence:
Stergios A. Polyzos, MD, MSc, PhD, Endocrinologist, 13 Simou Lianidi Str., 551 34 Thessaloniki, Greece, Tel./Fax: +302310424710; E-mail: stergios@endo.gr
Received 27-11-2013, Accepted 20-02-2014