Institute of Nuclear Medicine, Università Cattolica del Sacro Cuore, Rome, Italy
Radioiodinated metaiodobenzylguanidine (MIBG) was developed in the late 1970’s, at the Michigan University Medical Center, for imaging of the adrenal medulla and its diseases. Soon after, MIBG was shown to depict a wide range of tumors of neural crest origin other than pheochromocytomas/paragangliomas (Pheo/PGL) with the result that its use rapidly spread to many countries. After more than 30 years of clinical application, MIBG continues to be the most widespread radiopharmaceutical for the functional imaging of Pheo/PGL in spite of the emergent role of PET agents for detection of these tumors. In this paper we review the evolution in the use of MIBG over more than 30 years of experimental and clinical applications, with particular focus on the uptake mechanisms, pharmacokinetics, biodistribution and drug interaction as well as on clinical studies in Pheo/PGL also in comparison to other gamma-emitters tracers and PET radiopharmaceuticals.
Metaiodobenzylguanidine, Nuclear medicine, Paraganglioma, Pheochromocytoma
1. Introduction
Radioiodinated metaiodobenzylguanidine (MIBG) was developed in the late 1970’s at the Michigan University Medical Center after years of attempts to develop a radiolabeled precursor of epinephrine as an imaging agent and was the first radiopharmaceutical to be used for imaging the adrenal medulla and its diseases.1 After its introduction into clinical practice for the identification and localization of pheochromocytoma in 1981, MIBG rapidly demonstrated its ability to depict a wide range of tumors of neural crest origin other than pheochromocytoma, such as neuroblastoma, carcinoid tumor and medullary thyroid carcinoma.2-4 Because of its good selective uptake and retention by these tumors, the therapeutic potential of MIBG was early on explored and targeted therapy with 131I-MIBG was applied in the same tumors of neural crest origin.5,6 In addition, investigation into the use of MIBG to assess the functional status of the adrenergic autonomic innervation was soon undertaken, with lasting interest for the field of cardiology.7,8
Both 131I- and 123I-MIBG were developed in the USA and rapidly spread to many countries where they are today commercially available for routine use. 131I-MIBG gained FDA approval and became commercially available in 1994 for imaging pheochromocytoma and neuroblastoma. 123I-MIBG has been commercially available in Europe since the mid-nineties, while in the US it was used under an IND (Investigation New Drug) of the practice of pharmacy until September 2008 (when it gained FDA approval), this limiting its widespread use in the USA.9,10 Since the introduction of MIBG about 30 years ago, numerous experimental and clinical studies have been carried out and there have also been great developments in radiochemistry and instrumentation. At the present time, 131/123I-MIBG scintigraphy is still the most widespread functional imaging modality for the identification and localization of catecholamine secreting tumors. This review will focus on the evolution of the use of radioiodinated MIBG scintigraphy in Pheo/PGL.
2. Mechanisms of uptake and pharmacokinetics
Radioiodinated MIBG is a guanethidine analog structurally resembling norepinephrine (NE). Besides passive diffusion, MIBG is actively transported into tissues with sympathetic innervation by the norepinephrine transporter (NET).11 This active uptake mechanism, also known as uptake-1 system, is a sodium dependent process characterized by high affinity, low capacity, saturability and temperature and ouabain sensitivity, and occurs only in cells that express NET.12 By contrast, passive diffusion is non-specific, energy independent and unsaturable and takes place in all cells.12 Once in the cytoplasm, MIBG is stored into neurosecretory granules via vesicular monoamine transporters 1 and 2 (VMAT 1 and 2). After intravenous administration, radioiodinated MIBG is rapidly cleared from the vascular compartment (a small amount remains in the blood, mainly in platelets through the serotonin transporter) and accumulated in adrenergic tissues without binding to postsynaptic adrenergic receptors.13 The highest uptake in the heart is reached after 2-3 hours, whereas in tumors it is reached after 24-96 hours.12 The majority of the tracer is excreted unaltered via the kidneys (~50% within 24 hours; ~90% after 4 days); a minimal amount is excreted in the faeces, saliva, sweat and exhaled breath.13
3. Developments in radiochemistry and instrumentation
Radiochemistry
Initially, MIBG was labeled with 131I. However, this tracer has suboptimal dosimetry and imaging characteristics; this led to a preferential use of 123I-MIBG. Both 131I-MIBG and 123I-MIBG are commercially available as a “ready-to use formulation” in a sterile solution for i.v. injection. The effective dose in adults is 0.013 mSv/MBq for 123I-MIBG and 0.14 mSv/MBq for 131I-MIBG.14,15 The first experiences with 123I-MIBG go back to 1986 when Shulkin and colleagues reported the superiority of 123I-MIBG versus the 131I labeled radiopharmaceutical in a primary extra-adrenal pheochromocytoma.16 Today, 123I-MIBG is the tracer of choice both for tumor and non-tumor (cardiac) imaging. It allows better quality images, better photon detection and greater sensitivity. The higher photon flow allows high quality SPECT to be carried out. However, the main drawbacks of 123I-MIBG as far as the USA is concerned are the high cost and limited availability in this country for many years (until FDA approval in 2008), resulting in the routine use of 131I-MIBG in centers that did not have access to 123I-MIBG.
Regarding the evolution in MIBG radiochemistry, the main progress was the development of no-carrier-added MIBG, a radiolabeled agent with very high specific activity (>1200 mCi/μmol vs. approximately 1 mCi/mmol for commercially available 123I-MIBG and 1600 mCi/μmol vs. 1-10 mCi/μmol for commercially available 131I-MIBG for therapy) and negligible “cold” MIBG content.12,17 No-carrier-added (n.c.a.) MIBG is today available for experimental and clinical purposes, this having significant effects on both efficacy and safety of the preparation. Meanwhile, the main advantage of n.c.a. MIBG is evident in its therapeutic application due to the reduced molar amount of drug injected and consequently its reduced pharmacological side effects.18 In addition, MIBG analogs labeled with the alpha-emitter 211At for therapy and MIBG analogs labeled with positron emitting radionuclides such as 124I, 18F and 76Br for positron emission tomography (PET) have been developed.11,19
Instrumentation
From the first experiences with MIBG scintigraphy, technology was also evolved from classic planar scintigraphy with gamma cameras to single photon emission computed tomography (SPECT) imaging up to hybrid machines, which combine a dual head gamma camera with CT, allowing fusion of SPECT images with CT. In particular, SPECT increases reader confidence and improves visualization of lesions that may be missed on planar images due to their small size and/or superimposition of physiological (mainly liver and bladder) or pathological uptake or excretion.14 Moreover, the integration of anatomical and functional imaging by hybrid machines provides precise anatomical localization and metabolic characterization of pathological processes, while it improves SPECT interpretation with a significant impact on diagnostic accuracy;20 for example, SPECT-CT helps to distinguish between soft tissue and bone lesions, this information being important for tumor staging and therapeutic management.14,21 However, attention must be paid to CT-based attenuation correction that may cause enhanced physiological visualization of adrenal medulla, thus leading to misinterpretation (false positive results).22 Diagnostic accuracy can be further improved by using the recently available scanners incorporating spiral CT (diagnostic) technology compared to low-dose CT (non-diagnostic) hybrid machines.
4. Interfering drugs
A wide range of drugs may interfere with MIBG uptake and/or retention via various mechanisms of interaction, as demonstrated by pharmacologic ‘in vitro’ and ‘in vivo’ studies and clinical observations.13,14,23,24 Table 1 includes drugs known to interfere or suspected of interfering with MIBG uptake and/or retention divided into drug categories and the time of their withdrawal prior to imaging.14 With the exception of labetalol (suggested time of withdrawal 10 days), depot form of antipsychotics (suggested time of withdrawal 1 month) and a few other drugs, all medications have to be discontinued for 1-3 days.14,22,23 Most of the interfering drugs reduce MIBG uptake through various mechanisms and must be discontinued to avoid false negative results. On the other hand, calcium blockers such as nifedipine can cause prolonged retention of the tracer.24,25 A complete list of interfering medications is available in review articles and procedure guidelines in the literature.14,24 In hypertensive patients with metabolically active tumors, the decision to withdraw alfa- and beta-blockers should be taken in agreement with the referring physician.14 In our personal experience, the combination of doxazosin (alfa-receptor blockade) and atenolol (beta-receptor blockade) per os does not significantly reduce MIBG uptake. Recently, drugs that increase MIBG uptake by increasing the amount of NET expressed in tumors (such as histone deacetylase inhibitors) are under investigation in animal models in an attempt to enhance the therapeutic efficacy of 131I-MIBG therapy in patients with advanced malignant Pheo/PGL.26
5. Scintigraphic technique
Procedure guidelines on the use of MIBG scintigraphy in neuroendocrine tumors have been published in Europe.14 These guidelines summarize the views of European Committees and provide generic “recommendations that should be taken in the context of good practice of nuclear medicine and local regulation”; in any case, they form a helpful basis for nuclear medicine physicians in daily practice. Essentially, the following procedure is followed for both 131I-labeled and 123I-labeled MIBG. 40-80 MBq of 131I-MIBG and 200-400 MBq of 123I-MIBG are administered by slow intravenous injection (over 1-5 min to avoid potential side effects such as tachycardia or hypertensive crisis). Due to the small percentage of free radioiodine both present in the preparation (approximately 5%) and released “in vivo” after injection (approximately 3%), there is the need of thyroid blockade with potassium iodide administered orally (130 mg/day beginning 1 day before tracer injection and continued for 2 days for 123I-MIBG and 5 days for 131I-MIBG); in emergencies or in iodine-allergic patients, potassium perchlorate is used (400 mg/day started 1-4 hrs before tracer injection and continued for 2 days).14 Imaging is performed using a large field of view (LFOV) gamma camera equipped with a high-energy (131I-MIBG) or low-energy high-resolution (123I-MIBG) parallel-hole collimator. The standard time of imaging with 123I-MIBG is at 20-24 hours after injection (with optional images at 4-6 hours and/or 48 hours), compared to 24 and 48 hours when 131I-MIBG is used (with optional images at 72 hours); delayed images are useful if non-specific tracer accumulation is suspected in the kidneys and/or in the bowel. Anterior and posterior spot views of the head and neck, thorax, abdomen and pelvis (>150 kcounts for 131I-MIBG; about 500 kcounts for 123I-MIBG) are obtained; alternatively, anterior and posterior whole-body imaging may be performed (4-5 cm/min) with additional spot views in selected regions. Whenever possible, SPECT or SPECT-CT should be performed at 24h after 123I-MIBG administration, with acquisition parameters depending on the equipment available. In general, SPECT images are obtained over a 360° orbit, 128 × 128 or 64 × 64 word matrix, 120 projections in steps of 3°, 25-35 sec per step.14
6. Biodistribution and normal scintigraphic pattern
Knowledge of the biodistribution of radioiodinated MIBG is essential in order to avoid misinterpretation in scintigraphic images and false-positive results. In normal subjects, MIBG is accumulated in the myocardium, lungs, salivary glands, liver, spleen, large intestine and urinary bladder. Focal uptake of MIBG can be observed in cerebral tissues (cerebellum, basal nuclei and thalamic regions) after diagnostic or, more frequently, post-therapeutic images. Bilateral symmetrical activity is sometimes evident in the neck and supraclavicular region mainly of children and it is related to uptake in the brown adipose tissue that has an abundant supply of sympathetic nerves.27-29 No bone activity is ever evident. The normal adrenal medulla may show physiological uptake of both 131I- and 123I-MIBG and this finding is more frequently seen when 123I-MIBG is used (up to 75% of patients with 123I-MIBG versus approximately 10% with 131I-MIBG);30 it may thus be difficult with this tracer to assess whether adrenal uptake reflects physiological uptake, adrenal hyperplasia or a small pheochromocytoma.31 Attention must also be paid to MIBG uptake in the contralateral adrenal gland after resection of a pheochromocytoma.32 According to Cecchin and co-workers, the use of a scoring system based on liver uptake as a reference value (scores: 1, uptake absent or less than the liver; 2, equal to the liver; 3, moderately more intense than the liver; 4, markedly more intense than the liver, at images obtained at 24 hrs after 123I-MIBG injection) may help in correctly discriminating physiological adrenal uptake; classifying as positive the score 3-4 and as negative the score 1-2, these authors obtained a sensitivity of 91.5% and a specificity of 100% in the localization of adrenal and extra-adrenal tumors.33
7. Clinical studies
Imaging of pheochromocytoma/paraganglioma (Pheo/PGL) by means of radioiodinated MIBG is based on the expression of plasma membrane and vesicular transporter systems in these tumors. By MIBG scintigraphy, benign tumors are depicted as foci of increased MIBG uptake in adrenal or extra-adrenal sites, whereas multiple areas of uptake outside the adrenal and sympathetic ganglia characterize malignant lesions. One of the main advantages of MIBG scintigraphy is its ability to explore the whole body. Worldwide experience has demonstrated that MIBG scintigraphy can locate Pheo/PGL of all types, including adrenal and extra-adrenal tumors, and metastatic disease as well as Pheo associated with various familial syndromes and simple familial Pheo (Figure 1).30,34,35 Positive results have also been observed in adrenal tumors with low levels of secretion.36,37 When mild adrenal uptake is considered normal, specificity is high (>90%), with the exception of patients with MEN 2 syndrome.38
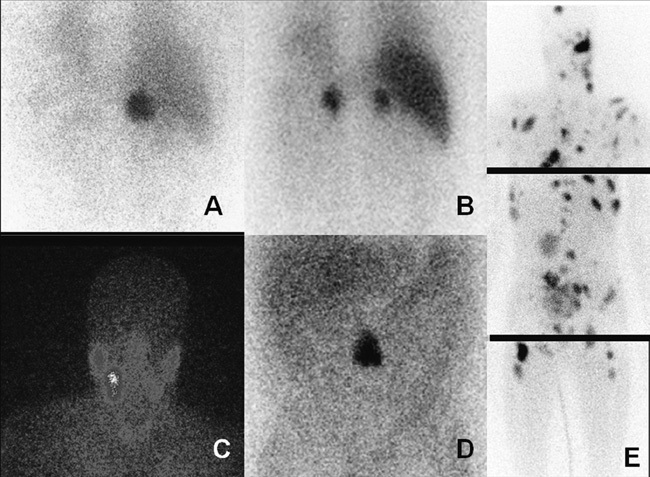
Figure 1. 123I-MIBG scintigraphy in various types of Pheo/PGL. A) Planar scintigraphy (posterior view) in a patient with right adrenal pheochromocytoma. B) Planar scintigraphy (anterior view) in a patient with familial bilateral pheochromocytoma. C) Planar scintigraphy (anterior view) in a patient with right head and neck PGL. D) Planar scintigraphy (anterior view) in a patient with abdominal PGL. E) Planar scintigraphy (spot anterior views) in a patient with metastatic PGL.
The overall sensitivity of 131I-MIBG scintigraphy in Pheo/PGL is high (77-90%), demonstrating better results with the use of 123I-MIBG and SPECT (88-96%).22,30,31 In malignant disease, lesion detection rate further improves at post-therapy high-dose 131I-MIBG scintigraphy, also with respect to 123I-MIBG.39,40 The main limitation of most of the initial studies was the small sample size as well as the paucity of data comparing MIBG and other anatomical or functional imaging techniques. A recent large prospective multicenter trial on the use of 123I-MIBG reported an overall sensitivity of 84% and specificity of 73%; specifically, for Pheos sensitivity and specificity were 84% and 73%, respectively, for PGL 75% and 100%, whereas for metastatic disease sensitivity was 83%.41 The positive results of 123I-MIBG scintigraphy are supported by a recent meta-analysis of 15 well-controlled clinical studies with a calculated sensitivity of 94% and specificity of 92%.42
Besides incorrect withdrawal of drugs that can alter MIBG uptake, false negative results may be caused by technical factors such as limitation in spatial resolution, or by intrinsic tumor characteristics such as low affinity to NET, the lack of storage granules or the loss of NET or VMAT by tumor cell dedifferentiation.31 In particular, the expression of VMAT-1 has been found to be essential for functional imaging of Pheo/PG with MIBG scintigraphy.43 Recent studies indicate that MIBG sensitivity is lower than previously reported, especially for the familial Pheo/PGL syndromes, the malignant lesions (57-79% sensitivity) and the extra-adrenal PGL tumors, particularly the non-secreting in the head and neck region.22,36,41,44,45 According to Fottner et al, clinical predictors for MIBG negativity are a predominant norepinephrine/normetanephrine secretion, age <45 years and a hereditary cause.43 In familial Pheos MIBG scintigraphy contributes little to patient management; in this group of patients, Taieb et al reported a sensitivity of 61%.36 Another limitation of MIBG scintigraphy when studying familial Pheos is the lack of specificity, namely the inability to distinguish between medullary hyperplasia and a tumor, since the mean diameter of familial tumors is usually smaller than that of sporadic Pheos. In the experience of De Graaf et al, in patients with MEN 2 syndrome specificity was as low as 17%.38
Recently, several gene mutations have been discovered in Pheo/PGL with different clinical phenotypes. In this setting, MIBG scintigraphy was found to show different behaviors in the various clinical syndromes, with reduced sensitivity in some familial PGL syndromes, malignant disease and extra-adrenal PGL. In particular, lower sensitivity of MIBG was found in von Hippel-Lindau syndrome (VHL), probably due to the low expression of NET in VHL-related pheochromocytoma cells.46 Similarly, a sensitivity of only 65% for 123I-MIBG was reported by Timmers et al in patients with mutations in SDHB genes.47 According to Fonte et al, all patients with false negative MIBG SPECT should be tested for SDHB mutations.48
8. Comparison with other gamma-emitter tracers and PET radiopharmaceuticals
Somatostatin receptor scintigraphy (SRS) is a sensitive whole body imaging in neuroendocrine tumors, but unlike MIBG it is not specific, providing information only on somatostatin receptor status. The sensitivity of 111In-DTPA-octreotide is inferior to radioiodinated MIBG in Pheo and functioning (sympathetic) PGL, whereas it has been shown to be superior to MIBG in head and neck (parasympathetic) PGL.49 In metastatic Pheo/PGL, a complementary role of SRS and MIBG scintigraphy has been suggested, as in a minority of these patients SRS can uncover more lesions.50-52
A wide range of PET tracers, both specific and non-specific for chromaffin tumors, have been applied for imaging Pheo/PGL.35,53,54 Generally considered, PET agents provide excellent tumor imaging and show a tumor-to-background ratio greater than 123I-MIBG; moreover, with PET technology a higher spatial resolution is obtained. Accordingly, with PET more lesions are detected with higher contrast. Other advantages versus MIBG scintigraphy are: less radiation exposure, no need of thyroid blockade or of withdrawing medication for many of them and immediate imaging after injection. However, except for 18F-FDG, PET agents do not yet have widespread clinical use due to their high cost and limited availability. Specific agents (124I-MIBG, 11C-hydroxyephedrine, 18F-DOPA, 18F-dopamine) show diagnostic specificity similar to MIBG. Among these, 124I-MIBG is used mainly for dosimetric estimates before 131I-MIBG therapy.55 Non-specific PET agents that can be used for imaging Pheo/PGL include 68Ga-somatostatin analogs (somatostatin receptor status) and 18F-FDG (glucose metabolism).54,56 Table 2 depicts diagnostic profiles from studies comparing PET tracers and MIBG in Pheos/PGL on a per-patient and on per-lesion basis.43-47,52,57-76 According to recently published EANM guidelines for radionuclide imaging of Pheo/PGL, MIBG is as sensitive as PET agents (mainly the specific ones) in patients with non-metastatic sporadic Pheo even in rare non-hypersecreting tumors.22 Therefore, in this group of patients PET agents should be used only for MIBG negative cases and/or in the presence of interfering medications (Figure 2).22 A real advantage of PET agents over MIBG seems to be with regard to malignant disease, partly due to the ability of PET-CT scanners to detect and localize very small lesions throughout the body. Literature data support the superiority of various PET tracers (in particular, 18F-Dopamine, 18F-DOPA and 18F-FDG) over 123I-MIBG to assess disease extension in patients with malignant Pheo/PGL, even though with different behavior in the different clinical syndromes on the basis of specific gene mutations.45,47,67,70,77 In any case, in these patients MIBG imaging provides unique information, allowing the selection of patients suitable for 131I-MIBG therapy. For head and neck paragangliomas, where MIBG scintigraphy usually fails, 18F-DOPA yields excellent results being most sensitive for small tumors.45 Preliminary data seem to indicate a primary role of 68Ga-labeled somatostatin analogs not only in patients with head and neck PGL (as expected) but also in those at high risk of PGL and metastatic disease.75,76
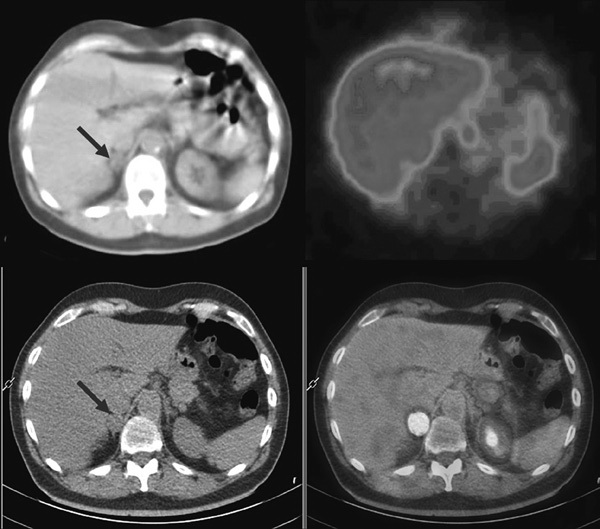
Figure 2. Top: 123I-MIBG SPECT-CT in a patient with Pheo in the right adrenal gland. The right adrenal mass is evident at low-dose CT (left image, red arrow) and shows no significant uptake at MIBG SPECT (right image). Bottom: 18F-DOPA PET-CT showing increased tracer uptake in the right adrenal mass (right image).
9. Conclusion
After more than 30 years of clinical application, MIBG continues to be the most widespread radiopharmaceutical for the functional imaging of Pheo/PGL in spite of the emergent role of PET agents in these tumors; 123I-MIBG is the recommended agent for diagnostic purposes. At the moment, MIBG is still a valid option in patients with non-metastatic sporadic Pheo due to its high sensitivity and wide availability; however, MIBG shows limited utility in patients with familial PGL as well as extra-adrenal and/or metastatic PGL. Thus, the real advantage of PET agents over MIBG seems to be for extra-adrenal PGL and malignant disease, with different behavior of the various PET agents in the different clinical syndromes on the basis of specific gene mutations. In any case, in malignant disease MIBG imaging provides unique information, enabling the selection of patients suitable for 131I-MIBG therapy.
Conflicts of interest: The authors declare no conflicts of interest.
Disclosures: None.
REFERENCES
1. Wieland DM, Wu JL, Brown LE, Mangner TJ, Swanson DP, Beierwaltes WH, 1980 Radiolabeled adrenergic neuron blocking agents: adrenomedullary imaging with 131I-iodobenzylguanidine. J Nucl Med 21: 349-353.
2. Sisson JC, Frager MS, Valk TW, et al, 1981 Scintigraphic localization of pheochromocytoma. N Engl J Med 305: 12-17.
3. Geatti O, Shapiro B, Sisson JC, et al, 1985 Iodine-131 metaiodobenzylguanidine scintigraphy for the location of neuroblastoma: preliminary experience in ten cases. J Nucl Med 26: 736-742.
4. Von Moll L, McEwan AJ, Shapiro B, et al, 1987 Iodine-131 MIBG scintigraphy of neuroendocrine tumors other than pheochromocytoma and neuroblastoma. J Nucl Med 28: 979-988.
5. Sisson JC, Shapiro B, Beierwaltes WH, et al, 1984 Radiopharmaceutical treatment of malignant pheochromocytoma. J Nucl Med 25: 197-206.
6. McEwan AJ, Shapiro B, Sisson JC, et al, 1985 Radioiodobenzylguanidine for the scintigraphic location and therapy of adrenergic tumors. Semin Nucl Med 5: 132-153.
7. Sisson JC, Shapiro B, Meyers L, et al, 1987 Metaiodobenzylguanidine to map scintigraphically the adrenergic nervous system in man. J Nucl Med 28: 1625-1636.
8. Agostini D, Verberne HJ, Hamon, Manrique A, 2008 Cardiac 123I-mIBG scintigraphy in heart failure. Q J Nucl Med Mol Imaging 52: 369-377.
9. Eisenhofer G, Pacak K, Goldstein DS, Chen C, Shulkin B, 2000 123I-MIBG scintigraphy of catecholamine systems: impediments to applications in clinical medicine [letter]. Eur J Nucl Med 27: 611-612.
10. Rufini V, Shulkin B, 2008 The evolution in the use of MIBG in more than 25 years of experimental and clinical applications. Q J Nucl Med Mol Imaging 52: 341-350.
11. Vaidyanathan G, 2008 Meta-iodobenzylguanidine and analogues: chemistry and biology. Q J Nucl Med Mol Imaging 52: 351-368.
12. Vallabhajosula S, Nikolopoulou A, 2011 Radioiodinated metaiodobenzylguanidine (MIBG): radiochemistry, biology, and pharmacology. Semin Nucl Med 41: 324-333.
13. Wafelman AR, Hoefnagel CA, Maes RA, Beijnen JH, 1994 Radioiodinated metaiodobenzylguanidine: a review of its biodistribution and pharmacokinetics, drug interaction, cytotoxicity and dosimetry. Eur J Nucl Med 21: 545-559.
14. Bombardieri E, Giammarile F, Aktolun C, et al, 2010 European Association for Nuclear Medicine. 131I/123I-metaiodobenzylguanidine (mIBG) scintigraphy: procedure guidelines for tumour imaging. Eur J Nucl Med Mol Imaging 37: 2436-2446.
15. ICRP Publication 80, 1998 Radiation dose to patients from radiopharmaceuticals. Annals of ICRP. Oxford, Pergamon Press.
16. Shulkin BL, Shapiro B, Francis IR, Dorr R, Shen SW, Sisson JC, 1986 Primary extra-adrenal pheochromocytoma: positive I-123 MIBG imaging with negative I-131 MIBG imaging. Clin Nucl Med 11: 851-854.
17. Barrett JA, Joyal JL, Hillier SM, et al, 2010 Comparison of high-specific-activity Ultratrace 123/131I-MIBG and carrier-added 123/131I-MIBG on efficacy, pharmacokinetics, and tissue distribution. Cancer Biother Radiopharm 25: 299-308.
18. Matthay KK, Weiss B, Villablanca JG, et al, 2012 Dose escalation study of no-carrier-added 131I-metaiodobenzylguanidine for relapsed or refractory neuroblastoma: new approaches to neuroblastoma therapy consortium trial. J Nucl Med 53: 1155-1163.
19. Watanabe S, Hanaoka H, Liang JX, Iida Y, Endo K, Ishioka NS, 2010 PET imaging of norepinephrine transporter-expressing tumors using 76Br-meta-bromobenzylguanidine. J Nucl Med 51: 1472-1479.
20. Rozovsky K, Koplewitz BZ, Krausz Y, et al, 2008 Added value of SPECT/CT for correlation of MIBG scintigraphy and diagnostic CT in neuroblastoma and pheochromocytoma. AJR Am J Roentgenol 190: 1085-1090.
21. Meyer-Rochow GY, Schembri GP, Benn DE, et al, 2010 The utility of metaiodobenzylguanidine single photon emission computed tomography/computed tomography (MIBG SPECT/CT) for the diagnosis of pheochromocytoma. Ann Surg Oncol 17: 392-400.
22. Taïeb D, Timmers HJ, Hindié E, et al, 2012 EANM 2012 guidelines for radionuclide imaging of phaeochromocytoma and paraganglioma. Eur J Nucl Med Mol Imaging 39: 1977-1995.
23. Khafagi FA, Shapiro B, Fig LM, Mallette S, Sisson JC, 1989 Labetalol reduces iodine-131-MIBG uptake by pheochromocytoma and normal tissues. J Nucl Med 30: 481-489.
24. Solanki KK, Bomanji J, Moyes J, Mather SJ, Trainer PJ, Britton KE, 1992 A pharmacological guide to medicines which interfere with the biodistribution of radiolabelled meta-iodobenzylguanidine (MIBG). Nucl Med Commun 13: 513-521.
25. Blake GM, Lewington VJ, Fleming JS, Zivanovic MA, Ackery DM, 1988 Modification by nifedipine of 131I-meta-iodobenzylguanidine kinetics in malignant phaeochromocytoma. Eur J Nucl Med 14: 345-348.
26. Martiniova L, Perera SM, Brouwers FM, et al, 2011 Increased uptake of [123I]meta-iodobenzylguanidine, [18F]fluorodopamine and [3H]norepinephrine in mouse pheochromocytoma cells and tumors after treatment with the histone deacetylase inhibitors. Endocr Related Cancer 18: 143-157.
27. Okuyama C, Ushjima Y, Kubota T, et al, 2003 123I-metaiodobenzylguanidine uptake in the nape of the neck of children: likely visualization of brown adipose tissue. J Nucl Med 44: 1421-1425.
28. Gelfand MJ, 2004 123I-MIBG uptake in the neck and shoulders of a neuroblastoma patient: damage to sympathetic innervation blocks uptake in brown adipose tissue. Pediatr Radiol 34: 577-579.
29. Hadi M, Chen CC, Whatley M, Pacak K, Carrasquillo JA, 2007 Brown fat imaging with (18)F-6-fluorodopamine PET/CT, (18)F-FDG PET/CT, and (123)I-MIBG SPECT: a study of patients being evaluated for pheochromocytoma. J Nucl Med 48: 1077-1083.
30. Ilias I, Divgi C, Pacak H, 2011 Current role of metaiodobenzylguanidine in the diagnosis of pheochromocytoma and medullary thyroid cancer. Semin Nucl Med 41: 364-368.
31. Havekes B, Lai EW, Corssmit PM, Romijn JA, Timmers HJLM, Pacak K, 2008 Detection and treatment of pheochromocytomas and paragangliomas: current standing of MIBG scintigraphy and future role of PET imaging. Q J Nucl Med Mol Imaging 52: 419-429.
32. Boersma HH, Wensing JW, Kho TL, de Brauw LM, Liem IH, van Kroonenburgh MJ, 2000 Transient enhance uptake of 123I-metaiodobenzylguanidine in the contralateral adrenal region after resection of an adrenal pheochromocytoma. N Engl J Med 342: 1450-1451.
33. Cecchin D, Lumachi F, Marzola MC, et al, 2006 A meta-iodobenzylguanidine scintigraphic scoring system increases accuracy in the diagnostic management of pheochromocytoma. Endocr Relat Cancer 13: 525-533.
34. Shapiro B, Copp JE, Sisson JC, Eyre PL, Wallis J, Beierwaltes WH, 1985 131-Iodine-metaiodobenzylguanidine for the locating of suspected pheochromocytoma: experience in 400 cases. J Nucl Med 26: 576-585.
35. Rufini V, Calcagni ML, Baum RP, 2006 Imaging of neuroendocrine tumors. Semin Nucl Med 36: 228-247.
36. Taïeb D, Sebag F, Hubbard JG, et al, 2004 Does iodine-131 meta-iodobenzylguanidine (MIBG) scintigraphy have an impact on the management of sporadic and familial phaeochromocytoma? Clin Endocrinol 61: 102-108.
37. Maurea S, Klain M, Mainolfi C, Ziviello M, Salvatore M, 2001 The diagnostic role of radionuclide imaging in evaluation of patients with non-hypersecreting adrenal masses. J Nucl Med 42: 884-892.
38. De Graaf JS, Dullaart RP, Kok T, Piers DA, Zwierstra RP, 2000 Limited role of meta-iodobenzylguanidine scintigraphy in imaging phaeochromocytoma in patients with multiple endocrine neoplasia type II. Eur J Surg 166: 289-292.
39. Fukuoka M, Taki J, Mochizuki T, Kinuya S, 2011 Comparison of diagnostic value of I-123 MIBG and high-dose I-131 MIBG scintigraphy including incremental value of SPECT/CT over planar image in patients with malignant pheochromocytoma/paraganglioma and neuroblastoma. Clin Nucl Med 36: 1-7.
40. Kayano D, Taki J, Fukuoka M, et al, 2011 Low-dose (123)I-metaiodobenzylguanidine diagnostic scan is inferior to (131)I-metaiodobenzylguanidine posttreatment scan in detection of malignant pheochromocytoma and paraganglioma. Nucl Med Commun 32: 941-946.
41. Wiseman GA, Pacak K, O’Dorisio MS, et al, 2009 Usefulness of 123I-MIBG scintigraphy in the evaluation of patients with known or suspected primary or metastatic pheochromocytoma or paraganglioma: results from a prospective multicenter trial. J Nucl Med 50: 1448-1454.
42. Jacobson AF, Deng H, Lombard J, Lessig HJ, Black RR, 2010 123I-meta-iodobenzylguanidine scintigraphy for the detection of neuroblastoma and pheochromocytoma: results of a meta-analysis. J Clin Endocrinol Metab 95: 2596-2606.
43. Fottner C, Helisch A, Anlauf M, et al, 2010 6-18F-fluoro-L-dihydroxyphenylalanine positron emission tomography is superior to 123I-metaiodobenzyl-guanidine scintigraphy in the detection of extraadrenal and hereditary pheochromocytomas and paragangliomas: correlation with vesicular monoamine transporter expression. J Clin Endocrinol Metab 95: 2800-2810.
44. Ilias I, Yu J, Carrasquillo JA, et al, 2003 Superiority of 6-[18F]-fluorodopamine positron emission tomography versus [131I]-metaiodobenzylguanidine scintigraphy in the localization of metastatic pheochromocytoma. J Clin Endocrinol Metab 88: 4083-4087.
45. King KS, Chen CC, Alexopoulos DK, et al, 2011 Functional imaging of SDHx-related head and neck paragangliomas: comparison of 18F-fluorodihydroxyphenylalanine, 18F-fluorodopamine, 18F-fluoro-2-deoxy-D-glucose PET, 123I-metaiodobenzylguanidine scintigraphy, and 111In-pentetreotide scintigraphy. J Clin Endocrinol Metab 96: 2779-2785.
46. Kaji P, Carrasquillo JA, Linehan WM, et al, 2007 The role of 6-[18F]fluorodopamine positron emission tomography in the localization of adrenal pheochromocytoma associated with von Hippel-Lindau syndrome. Eur J Endocrinol 156: 483-487.
47. Timmers HJ, Kozupa A, Chen CC, et al, 2007 Superiority of fluorodeoxyglucose positron emission tomography to other functional imaging techniques in the evaluation of metastatic SDHB-associated pheochromocytoma and paraganglioma. J Clin Oncol 25: 2262-2269.
48. Fonte JS, Robles JF, Chen CC, et al, 2012 False-negative ¹²³I-MIBG SPECT is most commonly found in SDHB-related pheochromocytoma or paraganglioma with high frequency to develop metastatic disease. Endocr Relat Cancer 19: 83-93.
49. Koopmans KP, Jager PL, Kema IP, Kerstens MN, Albers F, Dullaart RP, 2008 111In-octreotide is superior to 123I-metaiodobenzylguanidine for scintigraphic detection of head and neck paragangliomas. J Nucl Med 49: 1232-1237.
50. Tenenbaum F, Lumbroso J, Schlumberger M, 1995 Comparison of radiolabeled octreotide and meta-iodobenzylguanidine (MIBG) scintigraphy in malignant pheochromocytoma. J Nucl Med 36: 1-6.
51. van der Harst E, de Herder WW, Bruining HA, et al, 2001. [(123)I]metaiodobenzylguanidine and [(111)In]octreotide uptake in begnign and malignant pheochromocytomas. J Clin Endocrinol Metab 86: 685-693.
52. Ilias I, Chen CC, Carrasquillo JA, et al, 2008 Comparison of 6-18F-fluorodopamine PET with 123I-metaiodobenzylguanidine and 111In-pentetreotide scintigraphy in localization of nonmetastatic and metastatic pheochromocytoma. J Nucl Med 49: 1613-1619.
53. Pacak K, Eisenhofer G, Goldstein DS, 2004 Functional imaging of endocrine tumors: role of positron emission tomography. Endocr Rev 25: 568-580.
54. Cuccurullo V, Mansi L, 2012 Toward tailored medicine (and beyond): the phaeochromocytoma and paraganglioma model. Eur J Nucl Med Mol Imaging 39: 1262-1265.
55. Lopci E, Chiti A, Castellani MR, et al, 2011 Matched pairs dosimetry: 124I/131I metaiodobenzylguanidine and 124I/131I and 86Y/90Y antibodies. Eur J Nucl Med Mol Imaging 38: Suppl 1: 28-40.
56. Taïeb D, Sebag F, Barlier A, et al, 2009 18F-FDG avidity of pheochromocytomas and paragangliomas: a new molecular imaging signature? J Nucl Med 50: 711-717.
57. Shulkin BL, Wieland DM, Schwaiger M, et al, 1992 PET scanning with hydroxyephedrine: an approach to the localization of pheochromocytoma. J Nucl Med 33: 1125-1131.
58. Franzius C, Hermann K, Weckesser M, et al, 2006 Whole-body PET/CT with 11C-meta-hydroxyephedrine in tumors of the sympathetic nervous system: feasibility study and comparison with 123I-MIBG SPECT/CT. J Nucl Med 47: 1635-1642.
59. Mann GN, Link JM, Pham P, et al, 2006 [11C]metahydroxyephedrine and [18F]fluorodeoxyglucose positron emission tomography improve clinical decision making in suspected pheochromocytoma. Ann Surg Oncol 13: 187-197.
60. Mamede M, Carrasquillo JA, Chen CC, et al, 2006 Discordant localization of 2-[18F]-fluoro-2-deoxy-D-glucose in 6-[18F]-fluorodopamine- and [123I]-metaiodobenzylguanidine-negative metastatic pheochromocytoma sites. Nucl Med Commun 27: 31-36.
61. Zelinka T, Timmers HJ, Kozupa A, et al, 2008 Role of positron emission tomography and bone scintigraphy in the evaluation of bone involvement in metastatic pheochromocytoma and paraganglioma: specific implications for succinate dehydrogenase enzyme subunit B gene mutations. Endocr Relat Cancer 15: 311-323.
62. Timmers HJ, Chen CC, Carrasquillo JA, et al, 2009 Comparison of 18F-fluoro-L-DOPA, 18F-fluoro-deoxyglucose, and 18F-fluorodopamine PET and 123I-MIBG scintigraphy in the localization of pheochromocytoma and paraganglioma. J Clin Endocrinol Metab 94: 4757-4767.
63. Timmers HJ, Eisenhofer G, Carrasquillo JA, et al, 2009 Use of 6-[18F]-fluorodopamine positron emission tomography (PET) as first-line investigation for the diagnosis and localization of non-metastatic and metastatic phaeochromocytoma (PHEO). Clin Endocrinol (Oxf) 71: 11-17.
64. Hoegerle S, Nitzsche E, Altehoefer C, et al, 2002 Pheochromocytomas: detection with 18F DOPA whole body PET-initial results. Radiology 222: 507-512.
65. Taïeb D, Tessonnier L, Sebag F, et al, 2008 The role of 18F-FDOPA and 18F-FDG-PET in the management of malignant and multifocal phaeochromocytomas. Clin Endocrinol (Oxf) 69: 580-586.
66. Fiebrich HB, Brouwers AH, Kerstens MN, et al, 2009 6-[F-18]Fluoro-L-dihydroxyphenylalanine positron emission tomography is superior to conventional imaging with (123)I-metaiodobenzylguanidine scintigraphy, computer tomography, and magnetic resonance imaging in localizing tumors causing catecholamine excess. J Clin Endocrinol Metab 94: 3922-3930.
67. Rufini V, Treglia G, Castaldi P, et al, 2011 Comparison of 123I-MIBG SPECT-CT and 18F-DOPA PET-CT in the evaluation of patients with known or suspected recurrent paraganglioma. Nucl Med Commun 32: 575-582.
68. Shulkin BL, Thompson NW, Shapiro B, Francis IR, Sisson JC, 1999 Pheochromocytomas: imaging with 2-[fluorine-18]fluoro-2-deoxy-D-glucose PET. Radiology 212: 35-41.
69. Takano A, Oriuchi N, Tsushima Y, et al, 2008 Detection of metastatic lesions from malignant pheochromocytoma and paraganglioma with diffusion-weighted magnetic resonance imaging: comparison with 18F-FDG positron emission tomography and 123I-MIBG scintigraphy. Ann Nucl Med 22: 395-401.
70. Timmers HJ, Chen CC, Carrasquillo JA, et al, 2012 Staging and functional characterization of pheochromocytoma and paraganglioma by 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography. J Natl Cancer Inst 104: 700-708.
71. Nakano S, Tsushima Y, Higuchi T, Taketomi-Takahashi A, Amanuma M, 2012 Contrast-and non-contrast-enhanced ultrasonography (US) findings of hepatic metastasis from malignant pheochromocytoma/paraganglioma. Jpn J Radiol 30: 310-316.
72. Win Z, Al-Nahhas A, Towey D, et al, 2007 68Ga-DOTATATE PET in neuroectodermal tumours: first experience. Nucl Med Commun 28: 359-363.
73. Kroiss A, Putzer D, Uprimny C, et al, 2011 Functional imaging in phaeochromocytoma and neuroblastoma with 68Ga-DOTA-Tyr 3-octreotide positron emission tomography and 123I-metaiodobenzylguanidine. Eur J Nucl Med Mol Imaging 38: 865-873.
74. Naji M, Zhao C, Welsh SJ, et al, 2011 68Ga-DOTA-TATE PET vs. 123I-MIBG in identifying malignant neural crest tumours. Mol Imaging Biol 13: 769-775.
75. Naswa N, Sharma P, Nazar AH, et al, 2012 Prospective evaluation of 68Ga-DOTA-NOC PET-CT in phaeochromocytoma and paraganglioma: preliminary results from a single centre study. Eur Radiol 22: 710-719.
76. Maurice JB, Troke R, Win Z, et al, 2012 A comparison of the performence of 68Ga-DOTATATE PET/CT and 123I-MIBG SPECT in the diagnosis and follow-up of phaeochromocytoma and paraganglioma. Eur J Nucl Med Mol Imaging 39: 1266-1270.
77. Taïeb D, Rubello D, Al-Nahhas A, Calzada M, Marzola MC, Hindié E, 2011 Modern PET imaging for paragangliomas: relation to genetic mutations. Eur J Surg Oncol 37: 662-668.
Address for correspondence:
Vittoria Rufini, MD, Istituto di Medicina Nucleare, Università
Cattolica del Sacro Cuore, Largo Gemelli, 8, Zip code: 00168,
Roma, Italy, Tel.: +39 0630154978, Fax: +39 063058185,
e-mail: v.rufini@rm.unicatt.it
Received 06-09-12, Accepted 14-12-12