1Department of Anesthesiology, 2Department of Endocrinology and Metabolic Diseases, 3Depatment of Surgery, Leiden University Medical Center, Leiden, The Netherlands
Dear Sir,
Adrenalectomy for pheochromocytoma (PCC) is generally performed after preparation with α- and β-sympathicolytic agents to blunt the effects of the elevated catecholamine output.1 In patients also known to have hypertrophic cardiomyopathy, this preoperative workup needs more attention than in other patients with PCC. This is illustrated by the following case, in which the perioperative pass through Scylla (blunting catecholamine effects to allow adrenalectomy, potentially inducing hypotension and relative hypovolemia) and Charybdis (preventing hypovolemia and hypotension and maintaining sufficient afterload to prevent aortic outflow obstruction) lead to a hypertensive crisis during adrenalectomy.
A 41-year old woman presented to the Emergency Department with severe dyspnoea, palpitations, nausea and vomiting, from which she had been suffering for a long period. Her medical history included hypertension and a small myocardial infarction five years earlier without significant stenosis on coronary angiogram, for which a diagnosis of transient stress cardiomyopathy (also known as Tako-Tsubo, transient apical ballooning or broken heart syndrome) had been postulated.
At presentation, the patient’s blood pressure was 225/140 mmHg and heart rate 134 beats per minute (bpm), while the electrocardiogram (ECG) revealed a sinus tachycardia without signs of ischemia. She was admitted to the intensive care unit (ICU), where she was treated with intravenous (IV) diuretics, ß-blockade and nitroglycerine. Urinary excretion rates of norepinephrine, epinephrine and normetanephrine were markedly elevated and abdominal computed tomography showed a mass of 6 by 7 cm in the left adrenal gland, suspicious for PCC. Doxazosin 4 mg per os (PO), an α-sympathicolytic agent, was started and two days after admission the patient was discharged, awaiting adrenalectomy.
The day after the patient collapsed. At readmission a loud systolic murmur, grade 4/6, maximum point of loudness 2nd intercostal space on the left, was noticed. Echocardiography showed a severe concentric hypertrophic left ventricle with a systolic obstruction of the left ventricular outflow tract due to a systolic anterior motion (SAM) of the mitral valve with a maximal velocity over the outflow tract of 6 m/s (corresponding to a pressure gradient of 144 mmHg) and a mitral valve insufficiency grade 2-3. With fluid therapy and temporary termination of doxazosin the patient recovered. Preoperative preparation was started in the ward with doxasozin 4 mg PO 2 dd and, in view of the fact the patient had collapsed earlier while taking doxasozin, verapamil 120 mg PO. Metoprolol 50 mg PO 2 dd was added 2 days later because of a persisting heart rate >100 bpm and the patient was intravenously hydrated. Blood pressure and heart rate reached near normal values but still periods of hypertension and tachycardia persisted. Because of the patient’s history of hypertrophic obstructive cardiomyopathy (HOCM) with SAM resulting in cardiovascular collapse, a further increase in the dosage of α- and β-sympathicolytic agents was determined undesirable and the intentional treatment goal, orthostatic hypotension, was not achieved.
On the day of surgery, blood pressure was 125/73 mmHg and heart rate 73 bpm. In spite of high dose anesthetic induction, intubation was accompanied by a period of hypertension reaching 230/110 mmHg with a heart rate of 110 bpm. Anesthesia was continued and surgery proceeded uneventfully until reaching the left adrenal gland, whereupon various periods of hyper-, normo- and hypotension followed. Due to the varying intensity of surgical stimulation, the blood pressure now varied between 230/120 mmHg and 70/40 mmHg, in the presence of extra doses of anesthetics (Figure 1). Surgical pauses were needed to regain hemodynamic control but again were often complicated by hypotension requiring rapid reductions in anesthetic medication. At the time when the PCC had almost been removed, the blood pressure reached 280/140 mmHg accompanied by a tachycardia of 140 bpm, refractory to medication. This ended when the remaining adrenal tumour was swiftly excised. After adrenalectomy, norepinephrine up to 0.6 µg/kg/min was infused to prevent hypotension.
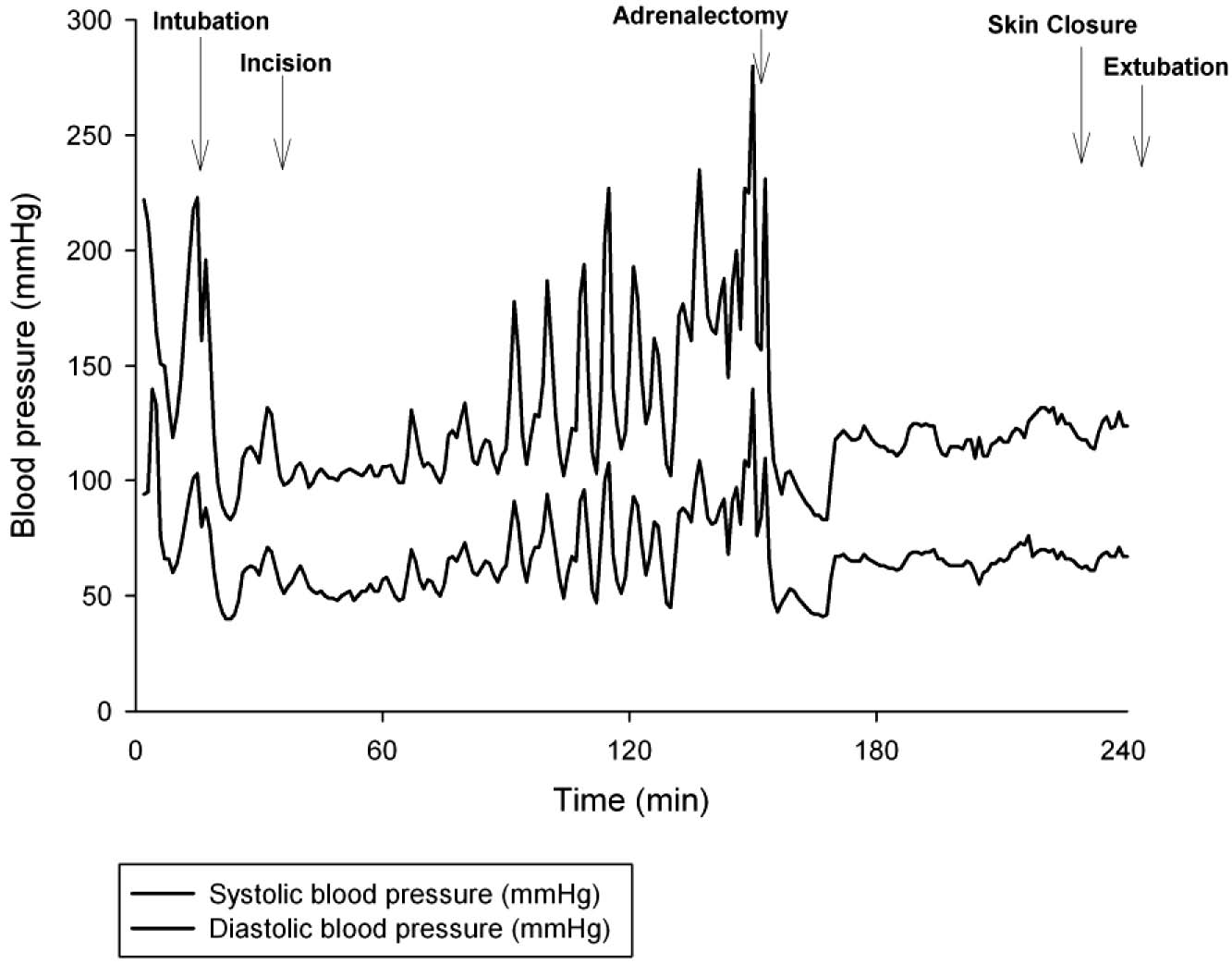
Figure 1. Intra-arterial systolic and diastolic blood pressure during adrenalectomy.
Pathological examination revealed a fully extirpated 6.5-5-6 cm PCC. Postoperative 24 h urinary excretion of catecholamines and metanephrines were normal. Interestingly, evaluation of the patient’s cardiac function one month and one year after surgery repeatedly displayed a non-dilated, normally functioning left ventricle without significant hypertrophy. This suggests that the left ventricular hypertrophy was a reversible condition, initiated by catecholamine excess and reversed after surgical removal of the PCC. A few cases of reversible dilatation of hypertrophied left ventricle in PCC have been reported before.2,3
In conclusion, in this case the preoperative α- and β-sympathicolytic dosage was kept low for fear of another cardiovascular collapse but proved insufficient intraoperatively. In retrospect, in this patient who collapsed preoperatively and was known to have a HOCM and SAM and a transient Tako-Tsubo cardiomyopathy, it would have been better to preoperatively monitor and control blood pressure and fluid volume status invasively at an intensive care unit (ICU) or a post anesthesia care unit (PACU) and thus allow for a more sufficient α- and β-blockade preoperatively.
This case stresses the importance of considering the presence of cardiomyopathy in any patient with PCC. It is reported that cardiomyopathy, resulting from catecholamine-induced myofibrillar damage, occurs in 30% of patients with PCC.4,5 Excess catecholamines contribute to myocardial hypertrophy not only indirectly via adrenergic receptor stimulation but also directly by stimulation of cardiac cell protein synthesis.6 A complete cardiac evaluation is therefore recommended in the preoperative evaluation of a patient with PCC.7
REFERENCES
1. Pacak K, Eisenhofer G, Ahlman H, et al, 2007 Pheochromocytoma: recommendations for clinical practice from the First International Symposium. October 2005. Nat Clin Pract Endocrinol Metab 3: 92-102.2. Lam JB, Shub C, Sheps SG, 1985 Reversible dilatation of hypertrophied left ventricle in pheochromocytoma: serial two-dimensional echocardiographic observations. Am Heart J 109: 613-615.
3. Wiswell JG, Crago RM, 1969 Reversible cardiomyopathy with pheochromocytoma. Trans Am Clin Climatol Assoc 80: 185-195.
4. Gilsanz FJ, Luengo C, Conejero P, Peral P, Avello F, 1983 Cardiomyopathy and phaeochromocytoma. Anaesthesia 38: 888-891.
5. Park JH, Kim KS, Sul JY, et al, 2011 Prevalence and patterns of left ventricular dysfunction in patients with pheochromocytoma. J Cardiovasc Ultrasound 19: 76-82.
6. Hjalmarson A, Kallfelt B, Swedberg K, Waagstein F, Waldenstrom A. Possible adrenergic effects on heart protein metabolism. Acta Med Scand Suppl 672: 85-93.
7. Pacak K, 2007 Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab 92: 4069-4079.
Address for correspondence:
Leonie Theresia van Hulsteijn, e-mail: L.T.van_Hulsteijn@lumc.nl
Received 01-03-2013, Accepted 22-05-2013