1Clinic for Endocrinology, Diabetes and Diseases of Metabolism, Clinical Center of Serbia, 2Clinic for Endocrinology, Diabetes and Diseases of Metabolism, Clinical Center of Serbia, Faculty of Medicine, University of Belgrade, Belgrade, Serbia, 3Endocrine Unit, Evgenidion Hospital, University of Athens Medical School, Athens, Greece, 4Institute for Medical Statistics and Informatics, Faculty of Medicine, University of Belgrade, Belgrade, Serbia
OBJECTIVE: The aim of our study was to evaluate the relationship between the elevated TSH and fracture risk in postmenopausal women with subclinical hypothyroidism for evaluation of individuals with a high risk for osteoporotic fractures. DESIGN: FRAX score calculation (10-year estimated risk for bone fracture) and measurement of bone markers (osteocalcin and beta cross-laps) were performed in 82 postmenopausal women with newly discovered subclinical hypothyroidism (mean age 59.17±7.07, mean BMI 27.89±3.46kg/m2, menopause onset in 48.05±4.09 years of age) and 51 matched controls (mean age 59.69±5.72, mean BMI 27.68±4.66kg/m2, menopause onset in 48.53±4.58 years of age) with normal thyroid function. RESULTS: The main FRAX score was significantly higher in the group with subclinical hypothyroidism than in the controls (6.50±4.58 vs. 4.35±1.56; p=0.001). Hip FRAX score was significantly higher in the group with subclinical hypothyroidism (1.11±1.94 vs. 0.50±0.46; p=0.030). There was no significant difference in bone markers: osteocalcin (23.99±12.63 vs. 21.79±5.34 ng/mL; p=0.484) and beta cross-laps (365.76±184.84 vs. 306.88±110.73 pg/mL; p=0.21) between the two groups. CONCLUSIONS: Postmenopausal patients with subclinical hypothyroidism, in particular of autoimmune origin, have higher FRAX scores and a thus greater risk for low-trauma hip fracture than euthyroid postmenopausal women. Our results point to the need to monitor postmenopausal patients with subclinical hypothyroidism for avoidance of fractures.
Bone markers, FRAX score, Postmenopausal women, Subclinical hypothyroidism, TSH
INTRODUCTION
Thyroid hormones play a critical role in the development of the skeleton, while it has been ascertained that the bone remodelling cycle in adulthood is closely related to thyroid function.1-3 It has thus been demonstrated that in adults thyroid hormone exerts catabolic effects on bone due to their effect on osteoclastic activity, while TSH can affect bone turnover via a direct pathway, independently of thyroid hormone levels.4 A lack of thyroid hormones during childhood will impair normal growth, but adult hypothyroid patients tend to exhibit higher bone density. Despite somewhat higher bone quantity, hypothyroidism is accompanied by increased fracture risk before and after diagnosis.6 Some data have suggested that even mild or moderate thyroid disease is a risk factor for osteoporosis, especially in postmenopausal women.7 Therefore, identification of subjects with a high risk for osteoporotic fractures is becoming an issue of major concern among populations with subclinical hypothyroidism (SCH), and particularly in postmenopausal women who are at increased risk for osteoporosis.
There is however no consensus regarding the importance of various prognostic tools for assessment of the risk in individual patients. Bone mineral density (BMD) is one of the most important predictive factors for fractures, but a number of independent risk factors, including age, history of maternal hip fracture, low body weight, height, previous hyperthyroidism, previous fracture, poor health and benzodiazepine use, may provide additional information about the 10-year fracture risk.8-13 Recently, the World Health Organization Collaborating Centre for Metabolic Bone Diseases in Sheffield (UK) has recommended the FRAX score as a fracture risk assessment tool enabling a 10-year prediction for possible fractures, which moreover includes clinical data and bone mineral density measured by dual-energy-X-ray absorptiometry-DXA.14,15 Based on this assessment, the National Foundation for Osteoporosis drew up guidelines for individuals with fracture risk.16-18 Treatment of osteoporosis should be considered for patients with low bone mineral density and a 10-year risk of hip fracture of ≥3% or a ≥20% 10-year risk of a major osteoporosis-related fracture as assessed with FRAX.19-21 Since fracture rates vary by ethnicity and race, it was suggested that it is desirable to examine data concerning fractures for each major ethnic group in each country in order to establish a calculator for that country. However, FRAX calculators are available for only 31 countries worldwide. In order to compensate for this lack of data on hip fracture incidence in very many countries, the WHO recommended using the FRAX model from a surrogate country with similarity in country-specific mortality rates so as to construct a country-specific FRAX model. On the other hand, some results indicate that the FRAX model from a surrogate country might lead to bias and that each country preferably requires its own FRAX.22
The aim of our study was to evaluate the usefulness of the FRAX score and bone markers determination in postmenopausal women with SCH in order to potentially identify individuals at high risk for osteoporotic fractures, and to investigate possible associations with serum TSH and fT4 levels.
SUBJECTS AND METHODOLOGY
FRAX score calculation (10-year estimated risk for bone fracture) and measurement of bone markers, osteocalcin and beta cross-laps (β cross-laps) were performed in a group of 82 postmenopausal women with SCH (mean age 59.17±7.07, mean BMI 27.89 ±3.46kg/m2, menopause onset at 48.81±6.76 years of age and 51 euthyroid controls matched for age (59.69±5.72), BMI (27.68±4.66kg/m2) and menopause onset (at 48.53±4.58 years of age). Investigation was performed after obtaining the approval of the local Ethics Committee. The inclusion criteria for the studied group were: postmenopausal women 40-70 years of age, TSH between 5mIU/L and 10mIU/L, fT4 within normal range, with no history of thyroid disease, no steroid therapy longer than 6 months, no osteoarthritis and no absorption bowel disease. Both groups were assessed via a questionnaire about independent risk factors for osteoporosis, such as parental fractures, previous fractures, current cigarette smoking and onset of menopause (Table 1).
TSH and fT4 were measured by the CMIA (chemiluminescent microparticle immunoassay) method by Abbott, on ARCHITECT ci8200. Reference ranges for TSH were 0.35-4.94 mIU/mL with analytical sensitivity of ≤0.1 µIU/mL and for Free T4 9.0-19.1pmol/L with analytical sensitivity of ≤0.4ng/dL. TPOAb were measured by CMIA (chemiluminescent microparticle immunoassay) by Abbott for the quantitative determination of the IgG class of thyroid peroxidase autoantibodies in human serum and plasma on the ARCHITECT i system. Normal values were <5.61 IU/mL. Bone markers, osteocalcin and β cross-laps were determined by the ECLIA method (electrochemiluminescence immunoassay) by Roche, on Cobas e601, with reference range for osteocalcin 15-46ng/ml, and for β cross-laps 104-1008 pg/ml, which is the range for postmenopausal women. Bone mineral density was measured by dual energy X-ray bone densitometer Lunar DPX. Measuring was performed on the lumbar spine (from L1 to L4) and left femoral neck. BMD was expressed as standard deviation (SD) in T-score. Fracture risk was calculated by the FRAX score assessment for Turkey.23,22
STATISTICAL ANALYSIS
Data were expressed as mean values with standard deviations (mean ± SD). All the data sets were normally distributed, except the FRAX score which was skewed. Categorical data were presented by absolute numbers with percentages and analyzed using a chi-square test and Fisher’s exact test, as appropriate. For continuous variables, the Student’s t-test or the Mann-Whitney U-test (for skewed data) was used. Differences were considered significant at a value of p<0.05. Odds ratios (ORs) for vertebral and hip fracture in relation to TSH, FT4 and TPOAb were determined using unadjusted and adjusted (for age, BMI and BMD expressed as T-score) logistic regression. A multivariate logistic regression was used to assess the most relevant risk fracture model. The ability of the TSH and FRAX score to discriminate between bone fracture positive and negative postmenopausal female subjects with SCH was calculated by the ROC (Receiver Operating Characteristic) curve method. The curves were drawn by plotting the sensitivity against the false positive rate (1-specificity), for varying cut-off levels of TSH and FRAX score. The area under the curve (AUC) was calculated and represents a quantitative measure of predictive value of TSH and FRAX score for bone fracture. A non-discriminating test would follow the diagonal line of the figure, whereas a 100% accurate test would coincide with the upper left corner of the box. Stepwise adjusted regression analysis of relationships between thyroid status and BMD (expressed as T-score), bone turnover and FRAX score was performed after adjustment for age, BMI and smoking. In logistic and linear regression analysis, all the individuals from both groups were included as a population in the model.
The study was conducted according to the principles expressed in the Declaration of Helsinki and was approved by the institutional Ethics Board of the Faculty of Medicine, University of Belgrade.
RESULTS
Age, body mass index, percentage of fat mass, TSH, FT4, TPO antibodies, current smoking, presence of diabetes type 2, previous fractures, parents’ hip fractures, bone markers and FRAX score of both examined groups are presented in Table 2. There were no significant differences in mean values of the evaluated descriptive parameters as regards parents’ fractures, current smoking, type 2 diabetes mellitus as well as treatment with corticosteroids. TSH was significantly higher in patients with SCH in comparison with the control group (8.00±2.82 mU/L vs. 2.23±0.94mU/L; p<0.001), while the free T4 was significantly lower (12.34±2.05pmol/L vs. 13.82±1.31pmol/L; p<0.001). TPO antibodies were more prevalent in subclinical hypothyroid individuals than in healthy subjects (48.7% vs. 3.9%; p<0.001). There were no significant differences between the two groups concerning age, body mass index and percentage of fat mass. Patients with SCH had more previous fractures in comparison with the control group (22% vs. 2%; p<0.001). The previous fractures recorded were morphometric vertebral fractures (n=8), wrist fractures (n=4), ankle fractures (n=4), hip fracture (n=1) and rib fracture (n=1) and all of them were detected by the X-ray examination. The control group included one individual with a hip fracture. The fractures had occurred at least 6 months before the examination of thyroid status. Hip T-score was lower in the examined group than in controls (-1.08±0.73 SD vs. -0.68±0.90SD; p=0.005). There was no significant difference in lumbar spine bone mineral density (-1.24±1.10 SD vs. -1.13±1.59SD; p=0.63) and bone markers - osteocalcin (23.99±12.63 ng/ml vs. 21.79±5.34ng/mL; p=0.484) and β cross-laps (365.76±184.84 pg/ml vs. 306.88±110.73pg/mL; p=0.21) between the two groups (Table 2). The main and hip FRAX scores were significantly higher in the group with subclinical hypothyroidism than in the disease-free group (6.50±4.58 vs. 4.35±1.56; p=0.001 and 1.11±1.94 vs. 0.50±0.46; p=0.030, respectively).
Unadjusted and adjusted (for age, BMI and lumbar spine T-score) logistic regression analysis indicated that TSH levels were related to fracture (p=0.019; OR=1.164; 95% CI for OR=1.026-1.322 and p=0.024; OR=1.173; 95% CI for OR=1.021-1.346, respectively), while the FT4 levels and presence of TPOAb did not further improve the regression model (p>0.05) (Table 3). The ability of the TSH and FRAX score to discriminate between bone fracture positive and negative postmenopausal female subjects is presented by area under the curve (AUC) plotted via ROC analysis (Table 4 and Figure 1). The determined FRAX score cut-off value by this analysis is 6%, with estimated sensitivity and specificity of 95% and 75.9%, respectively (Figure 1). The relationship between thyroid function tests, T-score, markers of bone turnover and FRAX scores after adjustment for age or age, BMI and smoking is presented in Table 5 and Figure 2. A significant association was found between serum osteocalcin level and TSH (OR=0.067; 95% CI for OR=0.007-0.128; p= 0.029) and between presence of TPOAb and FRAX score levels >6% (OR=4.471; 95% CI for OR=1.847-10.821; p=0.001).
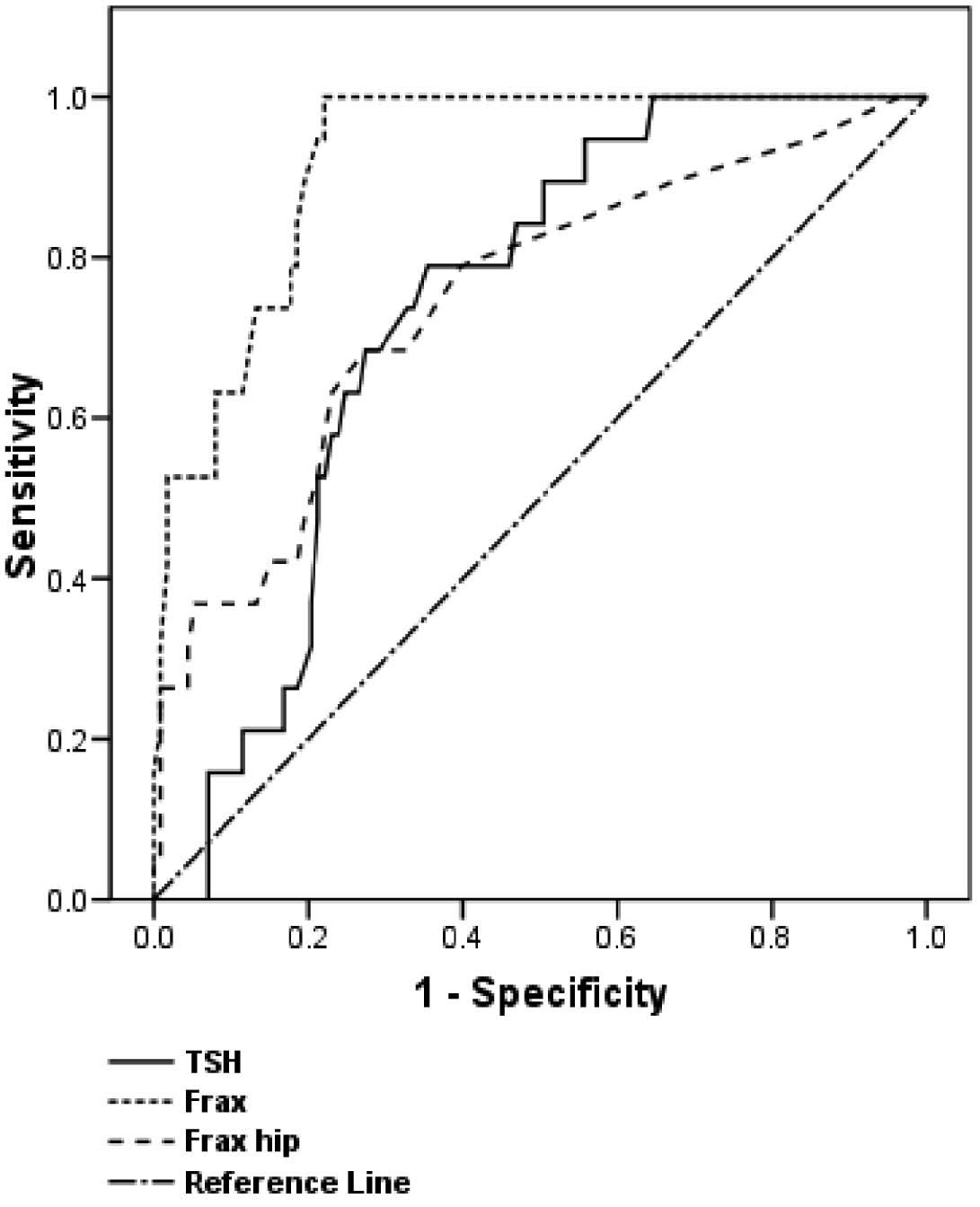
Figure 1. The ability of the TSH to discriminate between bone fracture positive and negative postmenopausal female subjects

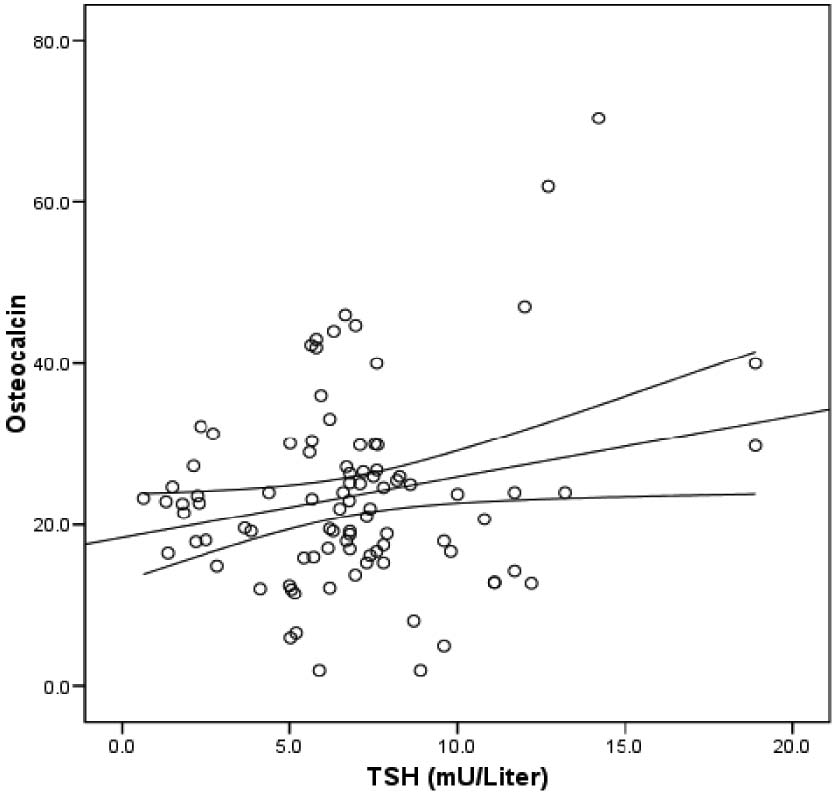
Figure 2. Relationship between thyroid function tests and T-score and FRAX score.
DISCUSSION
We compared the 10-year fracture risk in 82 postmenopausal SCH women with euthyroid women matched by age, BMI, years of onset of menopause and percentage of fat mass. We found that SCH women had higher risk for low-trauma hip fracture than euthyroid postmenopausal women and that TSH levels were related to fractures.
A few studies have pointed out that even a slight variation in normal thyroid function may have an impact on bone health.24-27 Recently, it has been hypothesized that TSH inhibits, via TSH receptors expressed on osteoblasts and osteoclasts, bone remodeling,28-31 and that by suppressing osteoclasts and osteoblasts, as evidenced in the TSHR knock-out mouse,3 TSH could be defined as a bone-suppressing hormone. However, other studies did not find any association between TSH, within normal range, and BMD, and others reported a prolongation of bone remodeling cycle in a thyroid deficiency state.32,33 TSH, operating as a possible molecular “switch” in the control of bone turnover, inhibits osteoclast survival by attenuating JNK/c-jun and NFkappaB signaling and osteoblast differentiation, while it also inhibits type 1 collagen expression in a Runx-2 by down-regulating Wnt and VEGF signaling.34 In untreated hypothyroid patients, high fracture risk has been reported to begin about 8 years prior to diagnosis. A study in which ultrasound bone densitometry was performed on the calcaneus in postmenopausal SCH women clearly showed that elevation of serum TSH concentration was associated with the decrease of calcaneus osteo sono assessment indices (OSI), concluding that elevated TSH in SCH affects bone turnover.35
Furthermore, a study in thyroidectomized patients due to thyroid carcinoma found that TSH is related to indicators of bone remodeling independently of thyroid hormone levels, this possibly pointing to a functional role of the TSHR in bone in humans. TSHR could thus eventually become a drug target in osteoporosis treatment. Despite several investigations, as regards whether TSH is involved or not in controlling bone metabolism, the results remain inconclusive.36,37 A recent study indicated that the combination of L-T4 and L-T3 in the treatment of primary hypothyroidism leads to a higher rate of bone resorption.38 Our previous findings suggest that even monotherapy with levothyroxine increases bone turnover in postmenopausal, but not in premenopausal women.39 Our study demonstrated an association between TSH levels and fractures (OR=1.179). The level of FT4 was within the normal range and was not associated with fractures.
Previous studies have shown that exposure to T3 decreased alkaline phosphatase activity in femoral bone marrow cell cultures but not in vertebral bone marrow cells.40 A study on an animal model found that gene expression markers for osteoblast and osteoclast in L-T4 treated rats were increased in the femoral bone but not in the lumbar spine.41 Meanwhile, another study showed that gravitational force might account for lower BMD in the femoral neck than in the lumbar spine in overt hypothyroidism.42 A study of a subclinical thyroid disease group (both, hyperthyroid and hypothyroid), compared with euthyroid controls, indicated that the effect of thyroid hormones on bone metabolism differs according to the site on the bone.43 A small study from our country on women with subclinical hypothyroidism did not show lower BMD than in the control group of euthyroid women.44 In this line of evidence, our results demonstrated that DXA-measured hip T-score was lower in the SCH group than in the control group. There was no difference in lumbar spine bone mineral density. Although the BMD yields valuable information about the skeleton in postmenopausal women,45 it is not a sensitive enough predictive fracture risk factor, since more than 50% of postmenopausal women with vertebral fractures have BMD within normal range or they do not meet the criteria for osteoporosis.19,46,47 In addition, some younger women with low bone density but no other risk factors have relatively low fracture rates. The limitation of DXA to predict fractures has led to the development of fracture assessment tools which combine bone density and clinical risk factors to improve the prediction of osteoporotic, low-trauma fractures.20 FRAX score as a computer-based algorithm for calculation of 10-year fracture probability evaluated clinical risk factors, habits and hip BMD. In our study, ROC analysis showed association between FRAX score and TSH. The SCH group had significantly higher prevalence of TPOAb (48.7% vs. 3.9%) and previous fractures (22% vs. 2%) than the control group. Vestergaard P et al in 2005 showed an increase in the fracture risk within the first 5 years after diagnosis of hyperthyroidism and the first 10 years after diagnosis of hypothyroidism. They also concluded that a previous fracture, corticosteroid use and alcohol abuse were significant risk factors for fractures.48 We have found an association between TPOAb presence and FRAX score (p=0.001). Events occurring in a patient may arise through complex interactions between individual genetic background and environmental exposure to infectious agents that generate a pro-inflammatory status, with subsequent perturbation of normal bone homeostasis and which are known to induce bone loss. Bone impairment in autoimmune disease is complex and is a consequence of an immunoregulatory imbalance. Alterations in homeostatic mechanisms might account for an imbalance of osteoblast activity leading to osteopenia. Alterations in the immune system may be responsible for mechanisms leading to osteoporosis. The immune mediators found to play a role in most autoimmune disorders could also be involved in promoting bone loss. Imbalanced osteclastic activity in new bone formation might occur as a consequence of chronic inflammatory autoimmune disorders leading to osteopenia. Inflammatory cytokines are major regulators of bone destructions (RANKL L - proosteoclastogenic (activated T-cells), OPG, IFN γ, IL 4 (antiosteclastogenic).31 Higher inflammatory response during bone regeneration was observed in immunologically restricted patients.49 Increased risk for fractures around the time of diagnosis has been reported in older men with subclinical hyper- or hypothyroidism50 as well as in the nationwide follow-up study performed by Vestergaard on patients with overt hyper- and hypothyroidism. Fracture risk decreased after surgical treatment of hyperthyroidism but further increased after diagnosis and treatment of hypothyroidism. This may be due to poor bone quality despite increase in BMD.51 Previous findings showed that poor bone quality in subclinical hypothyroidism is associated with elevations of osteoprotegerin (OPG), an inhibitor of osteoclastogenesis; this could be a compensatory response to the osteoclastic bone resorption, similarly to women with osteoporosis.52
Although the bone remodeling process was able to predict bone fracture independently of BMD, the FRAX score did not include bone markers in the calculation of fracture risk. In some studies, the contribution of bone markers in fracture risk was investigated, but the results were inconsistent, probably due to different markers and methods used in the studies.53-59 Meanwhile, several studies reported elevation of bone markers in post- and premenopausal women,4 whereas other investigators did not find any changes.60 Moreover, there were no differences among markers of bone turnover between patients with subclinical thyroid dysfunction and euthyroid controls.44 In our study, though we did not find any difference in the levels of osteocalcin and β cross-laps between the two groups (p >0.05), an association was registered between the serum osteocalcin level and TSH (p=0.029), this likely being due to more previous fractures in the SCH group and higher osteoblastic activity, which was recently shown in a meta-analysis.61
The limitations of our study are related to the cross-sectional design and to the fact that we were not aware of the duration of SCH before it was diagnosed. The retrospective data used in the questionnaire was validated by the patients’ personal medical history.
CONCLUSION
In our study, postmenopausal women with subclinical hypothyroidism, in particular of autoimmune origin, have higher FRAX scores and a thus greater risk for low-trauma hip fracture than euthyroid postmenopausal women. Although the pathogenesis is not as yet fully elucidated, and especially as regards the involvement of TSH, our results point to the need to monitor these patients for avoidance of fractures.
REFERENCES
2. Wojcicka A, Bassett JH, Williams GR, 2012 Mechanism of action of thyroid hormones in the skeleton. Biochim Biophys Acta (in press) http://dx.doi.org/10.1016/j.bbagen.2012.05.005
3. Williams GR, 2011 Extrathyroidal expression of TSH receptor. Ann Endocrinol 72: 68-73.
4. Mazziotti G, Porcelli T, Patelli I,Vescovi PP, Giustina A, 2010 Serum TSH values and risk of vertebral fractures in euthyroid post-menopausal women with low bone mineral density. Bone 46: 747-751.
5. Bassett JH, Williams GR, 2008 Critical role of the hypothalamic-pituitary-thyroid axis in bone. Bone 43: 418-426.
6. Lakatos P, 2003 Thyroid hormones: beneficial or deleterious for bone? Calcif Tissue Int 73: 205-209.
7. Duntas LH, 2003 Subclinical thyroid disorders: the menace of the Trojan horse. J Endocrinol Invest 26: 472-480.
8. Unnanuntan A, Gladnick BP, Donnelly E, Lane JM, 2010 The assessement of fracture risk. J Bone Joint Surg Am 92: 743-753.
9. Pearce EN, 2007 Thyroid dysfunction in perimenopausal and postmenopausal women. Menopause Int 13: 8-13.
10. Shidara K, Inaba M, 2009 Bone metabolic marker for osteoporosis. Nihon Rinsho 67: 927-931.
11. Kamel HK, Hussain MS, Tariq S, Perry HM, Morley JE, 2000 Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med 109: 326-328.
12. Follin SL, Black JN, McDermott MT, 2003 Lack of diagnosis and treatment of osteoporosis in men and women after hip fracture. Pharmacotherapy 23: 190-198.
13. Gardner MJ, Brophy RH, Demetrakopoulos D, et al, 2005 Interventions to improve osteoporosis treatment following hip fracture. A prospective, randomized trial. J Bone Joint Surg Am 87: 3-7.
14. Kanis JA, Johnell O, Oden A, Johansson H, McCloskey E, 2008 FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int 19: 385-397.
15. Baddoura R, Hoteit M, El-Hajj Fuleihan G, 2011 Osteoporotic fracture, DXA, and fracture risk assessment: Meeting future challenges in the Eastern Mediterranean region. J Clin Densitom 14: 384-394.
16. Leslie WD, Lix LM, Langsetmo L, et al, 2011 Construction of a FRAX® model for the assessment of fracture probability in Canada and implications for treatment. Osteoporos Int 22: 817-827.
17. Dawson-Hughes B, Tosteson AN, Melton LJ 3rd, et al, 2008 Implications of absolute fracture risk assessment for osteoporosis practice guidelines in the USA, Osteoporos Int 19: 449-458.
18. Lau B, Cole SR, Gange SJ, 2009 Competing risk regression models for epidemiologic data. Am J Epidemiol 170: 244-256.
19. Kanis JA, Johnell O, Johansson H, et al, 2006 Prior clinical vertebral fractures are a particularly strong predictor of hip fracture: a meta-analysis. Osteoporosis Int 17: Suppl 3: 365.
20. Watts NB, 2011 The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt) 20: 525-531.
21. Leslie WD, Majumdar SR, Lix LM, 2012 High fracture probability with FRAX usually indicates densitometric osteoporosis: implications for clinical practice. Osteoporos Int 23: 391-397.
22. Illias I, Spanoudi F, Koukkou E, Nikopoulou SC, 2012 Use of FRAX calculator with and without bone mineral density in Greek women. Hormones (Athens) 11: 222-223.
23. Cauley JA, El-Hajj Fuleihan G, Arabi A, et al, 2011 Official Positions for FRAX® clinical regarding International differences from Joint Official Positions Development Conference of the International Society for Clinical Densitometry and International Osteoporosis Foundationma on FRAX. J Clin Densitom 14: 240-262.
24. Pirro M, Manfredelli MR, Scarponi AM, et al, 2012 Association between thyroid hormone levels, the number of circulating osteoprogenitor cells, and bone mineral density in euthyroid postmenopausal women. Metabolism 61: 569-576.
25. Lippuner K, Johansson H, Kanis JA, Rizzoli R, 2009 Remaining lifetime and absolute 10-year probabilities of osteoporotic fracture in Swiss men and women. Osteoporos Int 20: 1131-1140.
26. Mayer J, 2007 The association between serum thyroid-stimulating hormones in its reference range and bone status in postmenopausal American women. Bone 40: 1128-1134.
27. Greet R, Bruno L, Stefan G, 2011 Thyroid hormone status within the physiological range affects bone mass and density in healthy men at the age of peak bone mass. Europ J Endocinol 164: 1027-1034.
28. Novack, D, 2003 TSH, the bone suppressing hormone. Cell 115: 129-130.
29. Sun L, Davies TF, Blair HC, Abe E, Zaidi M, 2006 TSH and bone loss. Ann N Y Acad Sci 1068: 309-318.
30. Bauer DC, 2009 Bone: does TSH concentration influence skeletal health? Nat Rev Endocrinol 5: 245-246.
31. Schett G, David JP, 2010 The multiple faces of autoimmune mediated bone loss. Nat Rev Endocrinol 6: 698-706.
32. Grimnes G, Emaus N, Joakimsen RM, Figenschau Y, Yorde R, 2008 The relationship between serum TSH and bone mineral density in men and postmenopausal women: the Tromso study. Thyroid 18: 1147-1155.
33. Mosekilde L, Eriksen EF, Charles P, 1990 Effects of thyroid hormones on bone and mineral metabolism. Endocrinol Metab Clin North Am 19: 35-63.
34. Abe E, Marians RC, Yu W, et al, 2003 TSH is a negative regulator of skeletal remodeling. Cell 115: 151-162.
35. Nagata M, Suzuki A, Sekiguchi S, et al, 2007 Subclinical hypothyroidism is related to lower heel QUS in postmenopausal women. Endocr J 54: 625-630.
36. Zaidi M, Davies TF, Zallone A, et al, 2009 Thyroid-stimulating hormone, thyroid hormones, and bone loss. Curr Osteoporos Rep 7: 47-52.
37. Heemstra KA, van der Deure WM, Peeters RP, et al, 2008 Thyroid hormone independent associations between serum TSH levels and indicators of bone turnover in cured patients with differentiated thyroid carcinoma. Eur J Endocrinol. 159(1):69-76.
38. Fadejev VV, Morgunova TB, Melnichenko GA, Dedov II, 2010 Combined therapy with L-Thyroxine and L-Triiodthyronine compared to L-Thyroxine alone in the treatment of primary hypothyroidism. Hormones (Athens) 9: 245-252.
39. Polovina S, Tucic-Nemet K, Pletikosic I, Obradovic A, 2009 The influence of thyroxin replacement therapy on bone mineral density in hypothyroid subjects. Endocrine Abstracts 11th European Congress of Endocrinology, 25-29 April, Istanbul, Turkey 20, p58.
40. Milne M, Kang MI, Quail JM, Baran DT, 1998 Thyroid hormone excess increases insulin-like growth factor I transcripts in bone marrow cell cultures: divergent effects on vertebral and femoral cell cultures. Endocrinology 139: 2527-2534.
41. Suwanwalaikorn S, Ongphiphadhanakul B, Bravenman LE, Baran DT, 1996 Differential responses of femoral and vertebral bones to long-term excessive L-Thyroxine administration in adults’ rats. Eur J Endocrinol 134: 655-659.
42. Bertoli A, Fusco A, Andreoli A, et al, 2002 Effect of subclinical hypothyroidism and obesity on whole-body and regional bone mineral content. Horm Res 57: 79-84.
43. Lee WY, Oh KW, Rhee EJ, et al, 2006 Relationship between subclinical thyroid dysfunction and femoral neck bone mineral density in women. Arch Med Res 37: 511-516.
44. Rasic-Milutinovic Z, Milicevic D, Gluvic Z, et al, 2008 Subclinical hypothyroidism and bone mineral density. Endocrine abstracts 16:75.
45. Johnell O, Kanis JA, Oden A, et al, 2005 Predictive value of BMD for hip and other fractures. J Bone Miner Res 20: 1185-1194.
46. De Laet C, Oden A, Johnell O, Jonsson B, Kanis JA, 2005 The impact of the use of multiple risk factors on case finding strategies: a mathematical framework. Osteoporosis Int 16: 313-318.
47. De Laet C, Kanis JA, Oden A, et al, 2005 Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporosis Int 16: 1330-1338.
48. Vestergaard P, Rejnmark L, Mosekilde L, 2005 Influence of hyper and hypothyroidism, and the effects of treatment with antithyroid drugs and levothyroxine on fracture risk. Calcif Tissue Int 77: 139-144.
49. Singh A, Mehdi AA, Srivastava RN, Verma NS, 2012 Immunoregulation of bone remodeling. Int J Crit Illn Inj Sci 2: 75-81.
50. Lee JS, Buzkova P, Fink HA, et al, 2010. Subclinical thyroid dysfunction and incident hip fracture in older adults. Arch Intern Med 170: 1876-83.
51. Vestergaard P, Mosekilde L, 2002 Fractures in patients with hyperthyroidism and hypothyroidism: a nationwide follow-up study in 16,249 patients. Thyroid 12: 411-419.
52. Botella-Carretero JI, Alvarez-Blasco F, San Millan JL, et al, 2007 Thyroid hormone deficiency and postmenopausal stratus independently increase serum osteoprotegerin concentrations in women. Europ J of Endocinol 156: 539-545.
53. Lee J, Vasikaran S, 2012 Current recommendation for laboratory testing and use of bone turnover markers on management of osteoporosis. Ann Lab Med 32: 105-112.
54. Tromp AM, Ooms ME, Popp-Snijders C, Roos JC, Lips P, 2000 Predictors of fractures in elderly women. Osteoporos Int 11: 134-140.
55. Ross PD, Kress BC, Parson RE, Wasnich RD, Armour KA, Mizrahi IA, 2000 Serum bone alkaline phosphatase and calcaneus bone density predict fractures: a prospective study. Osteoporos Int 11: 76-82.
56. Van Daele PL, Seibel MJ, Burger H, et al, 1996 Case-control analysis of bone resorption markers, disability, and hip fracture risk: the Rotterdam study. BMJ 312: 482-483.
57. Chapurlat RD, Garnero P, Bréart G, Meunier PJ, Delmas PD, 2000 Serum type I collagen breakdown product (serum CTX) predicts hip fracture risk in elderly women: the EPIDOS study. Bone 27: 283-286.
58. Greenfield DM, Hannon RA, Eastell R 2001 The association between bone turnover and fracture risk (The Sheffield Osteoporosis study). In: Eastell R, Baumann M, Hoyle N, Wieczorek L (eds) Bone markers - biochemical and clinical perspectives. Martin Dunitz, London; pp, 225-236.
59. Bauer DC, Garnero P, Harrison SL, et al, 2009 Biochemical markers of bone turnover, hip bone loss, and fracture in older men: the MrOS study. J Bone Miner Res 24: 2032-2038.
60. Martini G, Gennari L, De Paola V, et al, 2008 The effects of recombinant TSH on bone turnover markers and serum osteoprotegerin and RANKL levels. Thyroid 18: 455-460.
61. Biver E, Chopin F, Coiffeier G. et al. 2012 Bone turnover markers for osteoporotic status assessment? A systemic review of their diagnosis value at baseline in osteoporosis. Joint Bone Spine 79: 20-25.
Address for correspondence:
Prim Snezana Polovina, MD, M.Sc., Clinic for Endocrinology, Diabetes and
Diseases of Metabolism, Clinical Center of Serbia, Dr Subotica 13,
11000 Belgrade, Serbia, Tel.: +381 11 2 656 527, Fax: +381 11 2 685 357,
E-mail: snezanapolovina@gmail.com
Received 09-10-2012, Accepted 07-03-2013