1Department of Endocrinology, Aalborg Hospital, Aalborg, 2Medicine Clinics I, Bispebjerg Hospital, Copenhagen, Denmark
Although autoimmune hypothyroidism
has generally been considered to be a disease that mainly develops because of
genetic aberrations and for which adjustment of environment would bring about
but slight risk modification, this understanding is increasingly appearing to
be incorrect. We describe how iodine intake, smoking cessation and alcohol
intake are all strong modifiers of risk that, combined, may influence risk by a
factor of up to 30.
Unfortunately,
promotion of an environment leading to substantial lowering of the risk of
autoimmune hypothyroidism (i.e. improvement of dietary iodine deficiency,
decrease or cessation of smoking, and moderate alcohol intake) is not
incorporated within current public health promoting programs. Nevertheless, it
is increasingly becoming evident that knowledge of the importance of these
factors for disease development is likely to assist in the planning of health
promotion programs, while it will surely also be of value in the care of individual
patients.
Alcohol intake, Hypothyroidism, Iodine, Prevention, Tobacco smoking, Thyroid autoimmunity
While prevention of disease is, according to the available evidence, the most cost-effective way of reducing the burden of disease in a population,1 the concept of prevention of autoimmune thyroid disease is nevertheless frequently met with scepticism. This is because thyroid autoimmunity is considered to be caused predominantly by genetic aberrations.2,3 People are believed to be more or less ‘pre-destined’ to develop disease if they harbour ‘the wrong genes’, with little possibility of prevention.
This understanding does not seem to be correct, as discussed by Rothman and colleagues.4 Practically all diseases have multi-factorial etiology and cases can be attributed to both genes and environment. The fractions of disease attributable to genes and environment overlap and would normally add up to more than 100%. A specific case of disease may be 100% preventable by a change of environment, even if the disease is 100% dependent on a genetic abnormality. An example given by Rothman et al4 is phenylketonuria. In this disease, mental retardation is caused by a genetic disorder, but it is entirely preventable by diet.
Recent studies have identified a number of modifiable environmental risk factors having a strong influence on the occurrence of autoimmune hypothyroidism, and considerable reduction of risk is in principle possible via a program of primary prevention addressing the population as a whole.
Unfortunately, the present combination of effective measures includes: 1. that the population be kept iodine deficient, 2. that smokers be convinced not to quit tobacco, and 3. that people have a relatively high intake of alcohol. However, changes of these conditions in environment would unacceptably increase the risk of other diseases, and a balance has to be found. In this article, the association between these risk factors and the development of overt autoimmune hypothyroidism will be discussed. Additional environmental factors often mentioned in relation to risk of autoimmune thyroid disease, such as stress, minerals and vitamins in diet, various medications and chemicals, fetal life, pregnancy, and infections, are not discussed.
Iodine deficiency and the risk of disease
The iodine intake of a given population has a profound effect on the balance in occurrence of the various thyroid disorders.5 The most serious pattern of disease is observed when iodine deficiency is severe inducing insufficient thyroid hormone production, as can be seen when the median urinary iodine concentration in a population is below 20 µg/L.6 Thyroid hormones are important regulators of brain development, and irreversible brain damage with some degree of mental retardation may affect a considerable proportion of the population. In addition, many members of society will suffer from goitre and thyroid function abnormalities. In mild iodine deficiency (median urinary iodine concentration 50-99 µg/L6) the consequences observed are less severe, the most common being goitre.5
Normally, the thyroid gland will adapt to the low amount of iodine available in mild iodine deficiency. Iodine auto-regulation will lead to an increase in activity of the sodium-iodide-symporter (NIS) as well as many other processes, such as H2O2 generation, involved in thyroid hormone production.7 Thyroid hormone secretion is thereby maintained at sufficient levels. However, the thyroid hyperactivity often leads to goitre and to the development of multiple thyroid nodules with autonomous hormone product. This is why hyperthyroidism from multinodular toxic goitre is very common in elderly people when iodine intake is below the recommended level.
Figure 1 illustrates this latter phenomenon, which represents the age-associated incidence of the three most common types of hyperthyroidism in the Danish population before the initiation of a program of mandatory salt iodization in Denmark.8 Data were collected as part of the DanThyr study, which is the monitoring of the Danish iodization program.9 Particularly to be noted is the steep increase in incidence of multinodular toxic goitre with age in this mildly to moderately iodine deficient population.

Figure 1. Age-specific incidence rates (IR) of the three most common nosological types of hyperthyroidism in Denmark (Graves’ disease, multinodular toxic goitre, and solitary toxic adenoma). Data were obtained by prospectively surveying a population cohort (person-years of observation: 2,027,208) before the implementation of the Danish mandatory iodization of salt. Reproduced from Carlé A et al8 with permission.
As described in detail in the report on iodine deficiency by WHO/UNICEF/ICCIDD,6 it is important that a population have a sufficient iodine intake to prevent the consequences of iodine deficiency. On the other hand, iodine has complex effects on the thyroid gland, as discussed below.
Iodine intake and autoimmune hypothyroidism
The association between iodine intake and thyroid disease is U-shaped,10 excessive iodine intake with a median urinary iodine concentration above 300 µg/L officially labelled as being associated with a higher frequency of thyroid autoimmunity.6 Unfortunately, the increase in incidence of autoimmune hypothyroidism coming with a higher iodine intake starts already at the level of iodine deficiency.5,11
Figure 2 shows the incidence of overt autoimmune hypothyroidism in two Danish regions before the Danish iodization program. The two regions had different levels of iodine deficiency (moderate and mild) caused by a difference in the ground water iodine content.12 In Denmark, ground water is universally used for drinking, preparation of food, etc. As can be seen, the incidence of autoimmune hypothyroidism was nearly 50% higher in the area with only mild iodine deficiency compared with moderate iodine deficiency.11
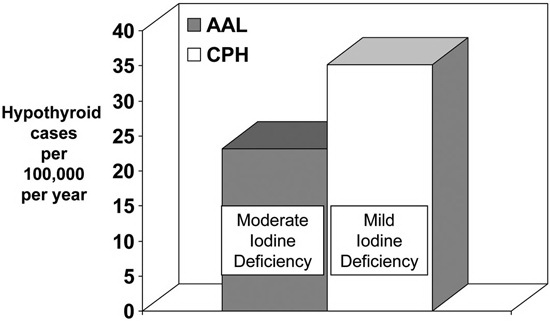
Figure 2. Standardized incidence rates of autoimmune hypothyroidism in Aalborg (AAL) with moderate iodine deficiency and in Copenhagen (CPH) with mild iodine deficiency, before the implementation of the Danish mandatory iodization of salt. The incidence rate was statistically significantly higher in Copenhagen than in Aalborg (standardized incidence rate ratio 1.46 (95% confidence interval 1.23-1.72). Data from Carlé A et al.11
Corresponding to this difference in the incidence of autoimmune hypothyroidism before iodization of salt, a general increase was observed in the incidence of hypothyroidism13 and in the prevalence of circulating thyroid antibodies14 in the Danish population after implementation of a cautious mandatory iodization of household salt and salt in bread. The program led to an increase in the median urinary iodine concentration in the population from 61 µg/L to 101 µg/L.15
Reports on several other populations have also shown that some degree of hypothyroidism is relatively rare in mild iodine deficiency and more common when iodine intake is adequate or more than adequate.16-19 Thus, viewed from the narrow standpoint of preventing autoimmune hypothyroidism, the population should be kept iodine deficient. On the other hand, this is associated with a high frequency of other diseases. What is the optimal strategy?
Current evidence clearly indicates that iodine deficiency should be avoided6,20 and that this will lower the combined burden of disease. Figure 3 illustrates the accumulated risks of different degrees of hyper- and hypothyroidism in old people who had lived their lives with moderate iodine deficiency in the city of Randers, Jutland, Denmark and comparable people who had lived with recommended iodine intake in the city of Skagen, Jutland, Denmark.21 In Skagen the ground water iodine content is high,12 caused by marine deposits near the aquifer. 22 As illustrated, the burden of hyperthyroidism was much lower in Skagen with recommended iodine intake, and this more than compensated for the higher risk of hypothyroidism.
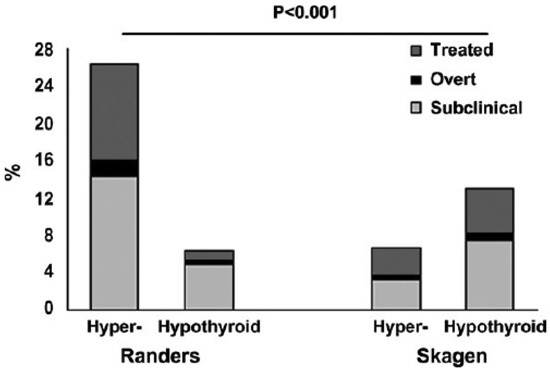
Figure 3. Comparative epidemiological study of hyper- and hypothyroidism among 75-80 year-old Danish residents of Randers (n=212) and Skagen (n=218). Median urinary iodine concentration was 55 μg/L in Randers and 160 μg/L in Skagen. Participants from Randers often (26%) had visible goitre by clinical examination. In Skagen none had goitre. Some degree of hyperthyroidism or previous therapy for hyperthyroidism was significantly more common in Randers (p<0.001), whereas hypothyroidism was more common in old residents in Skagen (p=0.019). Having no history or biochemical evidence of thyroid dysfunction it was more common in Skagen (p=0.002). Reproduced from Andersen S et al22 with permission.
The rather narrow interval of optimal iodine intake with a median urinary iodine concentration of 100-200 µg/L6 giving the lowest burden of disease makes it important to monitor and to control iodine intake from diet.23 In a population that is iodine deficient from natural diet, food enriched with iodine (typically salt) should be evenly distributed among the population. Moreover, intake of food containing excessive iodine should be limited.
Cessation of tobacco smoking and the risk of autoimmune hypothyroidism
Tobacco smoking is among the leading causes of death in most countries. Using data published by the United States Centre for Disease Control (CDC), it was calculated that in the year 2000 18.1% of deaths in the USA were attributable to smoking tobacco.24 Cessation of smoking reduces the risk of smoking associated cardiovascular disease and cancer substantially, and quitting tobacco has been vigorously promoted to prevent disease and death. However, as illustrated in Figure 4, cessation of smoking is associated with a steep, though transient, increase in the risk of developing autoimmune hypothyroidism within the two first years.25
If autoimmune hypothyroidism were the only disease to prevent in the population via cessation of smoking, it would not be advisable to encourage smokers to quit; but up to 15 years of lost life expectancy caused by smoking26 tips the balance fundamentally. Thus, it is not rational to seek to prevent autoimmune hypothyroidism by recommending people to continue smoking. On the other hand, physicians (and smokers) should be aware that nearly any complaint reported by someone who has quit smoking within the last two years should lead to a test of the thyroid function. In Denmark, 85% of the patients who were diagnosed with overt autoimmune hypothyroidism within two years after quitting smoking would theoretically not have developed hypothyroidism during this period of time if they had continued to smoke.25
The mechanism behind the surge of autoimmune hypothyroidism after cessation of smoking is currently unknown. Several studies have shown a lower prevalence of thyroid antibodies in smokers compared with non-smokers,27-29 while it is also reported that people who quit smoking may become positive for thyroid antibodies.30 Smokers have a lower prevalence of subclinical hypothyroidism than non-smokers31 but, as illustrated in Figure 4, we found no significant protective effect of current smoking on the incidence of overt autoimmune hypothyroidism, even though the odds ratio was slightly below one. This lack of protection from current smoking is in accordance with results of a previous meta-analysis of the association between smoking and overt hypothyroidism.32
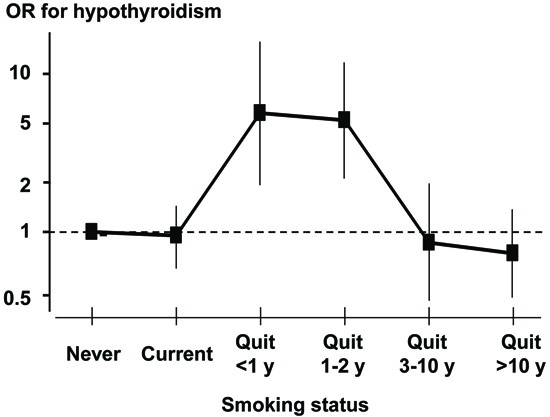
Figure 4. Risk of developing hypothyroidism according to smoking history. Odds ratios (with 95% confidence intervals) for the different groups are presented and connected with lines to illustrate a hypothetical process from non-smoker to current and then to ex-smoker with different time intervals since quitting smoking. Each group was compared with never-smokers in a multivariate logistic model including also alcohol consumption and family history of hypothyroidism. Reproduced from Carlé A et al25 with permission.
After two years had passed since quitting, the odds ratio returned to below one (Figure 4), but this was not statistically significant. Our study did not have the power to evaluate whether the long-term result of quitting smoking is a lower risk of autoimmune hypothyroidism. If all the participants who had quit smoking at some point of time were combined into one group, the risk would not be different from never-smokers,25 and it may be speculated that those who developed overt autoimmune hypothyroidism shortly after quitting smoking would have developed the disease later if they had continued smoking.
The conclusion that can be drawn from the results illustrated in Figure 4 is that optimal behaviour is never to start smoking. People would then not be exposed to the inconvenience and risks of quitting smoking. Autoimmune hypothyroidism is not the only disease that may develop in people who quit smoking. Body weight often increases after quitting33 and there is an increase in risk of developing diabetes.34,35
It is interesting to speculate on some possible mechanisms behind the rebound in autoimmunity leading to hypothyroidism in the first years after quitting smoking.
One possibility is that the increase in risk is caused by the thyroid follicular cells being exposed to extra iodide after cessation of smoking. Smokers have a high generation of thiocyanate from cyanide in tobacco smoke, and thiocyanate is a competitive inhibitor of NIS transport of iodide.36 As judged from the inhibitory effect of smoking on iodide transport into milk of breast feeding mothers (no iodine autoregulation of NIS occurs in the mammary gland and inhibition of NIS leads to a decrease in milk iodine content), smoking primarily reduces iodide transport by NIS into the thyroid by around 50%.37 In the thyroid, such inhibition of NIS will be autoregulated by enhancement of NIS gene transcription.7 It may be speculated that after many years of smoking, chronically up-regulated thyroidal NIS may stay high for a period after cessation of smoking. The excess iodide transported into the thyroid may increase the risk of autoimmune hypothyroidism, similar to the increase in risk of hypothyroidism seen after excess iodine intake with diet.5
Another possible mechanism behind the development of autoimmune hypothyroidism after cessation of smoking is oxidative damage caused by sudden exposure to oxygen in tissues. Smokers tend to have a partial oxygen deficiency in tissues,38 and it may be speculated that the sudden increase in tissue oxygen after cessation of smoking could promote autoimmunity via reactive oxygen species mediated tissue damage. This would likely act in concert with the effect of increasing iodide content in the thyroid.
A third possibility is that nicotine or other compounds in smoke dampen thyroid autoimmune activity by unknown mechanisms, somewhat similar to the protective effect of alcohol consumption discussed below. This hypothesis may be supported by the finding of a lower prevalence of circulating thyroid antibodies in smokers27-29 and also by a recent report in an animal model of autoimmunity indicating that a nicotine derivative may protect against thyroid autoimmunity.39 By contrast, current smoking did not protect against overt autoimmune hypothyroidism in our study (Figure 4), which may argue against this hypothesis.
Alcohol consumption and autoimmune hypothyroidism
In the report on causes of death in the USA in the year 2000, 3.5% of deaths were associated with excess intake of alcohol.24 On the other hand, it is known from series of studies that a moderate consumption of alcohol (compared with total abstention from alcohol) is associated with reduced risk of many diseases.40-46 Autoimmune hypothyroidism is one of the diseases that are less common in people having moderate alcohol consumption.
Figure 5 illustrates the odds ratio (OR) for developing autoimmune hypothyroidism in people split into groups according to habits of alcohol intake.47 The reference group in this analysis was people consuming 1-10 units of alcohol per week. One unit corresponds to a glass of wine or beer or spirit (~10-12 g of alcohol) and 1-10 units per week was the most common level of consumption in the Danish population under study. As shown in the figure, the OR for developing hypothyroidism was twice as high in alcohol abstainers and around half in people with the higher alcohol intake of 11-20 units/week. The small group (~6% of participants) reporting a higher than this level of alcohol consumption was not different from the reference. In a separate analysis, no difference in protective effect was observed between people who predominantly consumed beer vs. wine consumers.47

Figure 5. The dose-effect of alcohol consumption on the occurrence of overt autoimmune hypothyroidism. Data from 140 cases and 560 individually matched controls from the same population and undergoing identical investigational programs were analysed in a conditional multivariate regression model. Odds ratios (with 95% confidence limits) for developing hypothyroidism were calculated in groups having different levels of alcohol consumption. Reference in all comparisons was the group having a consumption of 1-10 units per week. Reproduced from Carlé A et al47 with permission.
Thus, moderate alcohol intake was associated with a dose-dependent protection against development of autoimmune hypothyroidism. The mechanism leading to this less aggressive reaction of the immune system against the thyroid gland when exposed to alcohol is unknown. In a recent Dutch follow-up study of relatives to patients with autoimmune thyroid disease, a similar protective effect of alcohol on the development of autoimmune hypothyroidism was observed.48 Among 38 cases of overt hypothyroidism 6.7% consumed >10 units of alcohol/week, whereas this was 23.7% of the 76 matched controls that had not developed hypothyroidism.
We previously observed a dose-dependent protective effect of alcohol intake on development of goitre and solitary thyroid nodules,49 and alcohol abusers tend to have a small fibrotic thyroid.50 It is possible that there is some overlap between the mechanisms leading to a lower rate of growth and nodule formation and those giving a lower tendency to autoimmune destruction of the thyroid gland. On the other hand, a protective effect of alcohol intake has also been described for some other autoimmune diseases,51-53 which may suggest a more direct effect of alcohol on the immune system. There is a complex interaction between alcohol and various components of the immune system, and alcohol has been shown to dampen some immune responses both in animals and in humans.54-56
An example of high vs. low risk of autoimmune hypothyroidism and the possibility of prevention
The incidence of autoimmune hypothyroidism increases by around 50% per advancing decade of life,11 it is 3.5 times higher in women than in men,11 and it seems to be three times as high in white Caucasians as in people of black African ancestry.57 Age, sex, and race are non-modifiable risk factors, but iodine intake, smoking, and alcohol intake are modifiable.
As illustrated in the present report, people with sufficient iodine intake according to the WHO/UNICEF/ICCIDD recommendations6 have a 1.5-2 times higher risk of developing autoimmune hypothyroidism compared with people having mild to moderate iodine deficiency, and people abstaining from alcohol have a four times higher risk than people who consume 11-20 units of alcohol per week. Moreover, people who stop smoking have for a two-year period a six times higher risk than those who continue smoking.
Our analyses give no indication that the differences in risk associated with these three factors are interdependent. The following comparison illustrates how strongly modifiable the risk of autoimmune hypothyroidism is by a change of environment:
The risk of developing autoimmune hypothyroidism during the year to come in a person who quit smoking, who does not consume alcohol, and who has an average recommended iodine intake of 150-200 µg/day is in the order of 30 times higher than it would have been if the same person had continued smoking, had regularly consumed two units of alcohol per day, and had been iodine deficient with a daily intake of 50-100 µg iodine.
As discussed, although prevention of autoimmune hypothyroidism via such a change in environment is incompatible with current public health programs, the example illustrates the profound effect of modifiable environment on the risk of developing autoimmune hypothyroidism. In this disorder, considered by many to be mostly dependent on non-modifiable genetic aberration, simple changes of environment are capable of modifying the risk of disease development by a factor of 30.
Conclusion
The risk of autoimmune hypothyroidism is, according to strong evidence, profoundly modifiable by a change of environment. However, modification of environment to reduce the risk of hypothyroidism may unacceptably increase the risk of other diseases. Thus, prevention of autoimmune hypothyroidism is somewhat similar to the problem facing prehistoric Greek sailors endeavouring to pass between Scylla and Charybdis: manoeuvring the ship to avoid one monster would bring them within reach of the other monster. Nonetheless, some reasonable suggestions can be put forward.
Based on the reduction in risk of developing autoimmune hypothyroidism and some other diseases, people who prefer to have daily moderate alcohol consumption can be reassured.
The fight against smoking should focus even more on convincing people never to start regular smoking in the first place. Smokers who quit should have their thyroid function tested if they present any symptoms or signs compatible with thyroid dysfunction.
Iodine intake of an iodine deficient population should be brought to the level where iodine deficiency disorders are prevented, but not higher. Iodine intake of populations should be monitored as part of public health programs.
* According to Greek mythology, the sea-monsters Scylla and Charybdis were on either side of a strait so narrow that sailors attempting to avoid one monster would come dangerously close to the other.
REFERENCES
1. Laurberg P, Andersen S 2011 Prevention in endocrinology. In: Steward & Wass (eds) Oxford Textbook of Endocrinology and Diabetes 2nd ed. Oxford University Press; pp 8-13.
2. Brand OJ, Gough SCL, 2010 Genetics of thyroid autoimmunity and the role of the TSHR. Molecular and Cellular Endocrinol 322: 135-143.
3. Brix TH, Hegedüs L, 2012 Twin studies as a model for exploring the aetiology of autoimmune thyroid disease. Clin Endocrinol 76: 457-464.
4. Rothman KH, Greenland S, Poole C, Lash TL, 2008 Proportion of disease due to specific causes. In: Rothman KH, Greenland S & Lash TL (eds) Modern Epidemiology 3rd ed. Lippincott Williams & Wilkins; pp 13-15.
5. Laurberg P, Cerqueira C, Ovesen L, et al, 2010 Iodine intake as a determinant of thyroid disorders in populations. Best Practice & Research. Clin Endocrinol Metab 24: 13-27.
6. WHO, UNICEF & ICCIDD, 2007 Assessment of iodine deficiency disorders and monitoring their elimination. A guide for programme managers 3rd ed. World Health Organization.
7. Gärtner R, 2009 Autoregulation of thyroid growth and function by iodine: independent regulation of the thyroid gland by iodocompounds. In: Preedy CR, Burrow GN & Watson TRW (eds). Comprehensive handbook of iodine: nutritional, biochemical and therapeutic aspects, Elsevier; pp 243-247.
8. Carlé A, Pedersen IB, Knudsen N, et al, 2011 Epidemiology of subtypes of hyperthyroidism in Denmark – a population based study. Eur J Endocrinol 164: 801-809.
9. Laurberg P, Jørgensen T, Perrild H, et al, 2006 The Danish Investigation on Iodine Intake and Thyroid Disease (DanThyr): status and perspectives. Eur J Endocrinol 155: 219-228.
10. Laurberg P, Pedersen IB, Carlé A et al, 2009 The U-shaped curve of iodine intake and thyroid disorders. In: Preedy VC, Burrow GN, Watson RR (eds). Comprehensive handbook on iodine: nutritional, endocrine and pathological aspects. Elsevier, pp; 449-455.
11. Carlé A, Laurberg P, Pedersen IB, et al, 2006 Epidemiology of subtypes of hypothyroidism in Denmark. Eur J Endocrinol 154: 21-28.
12. Pedersen KM, Laurberg P, Nøhr S, Jørgensen A, Andersen S, 1999 Iodine in drinking water varies more than 100 fold in Denmark. Importance for iodine content of infant formulas. Eur J Endocrinol 140: 400-403.
13. Pedersen IB, Laurberg P, Knudsen N, et al, 2007 An increased incidence of overt hypothyroidism after iodine fortification of salt in Denmark: a prospective population study. J Clin Endocrinol Metab 92: 3122-3127.
14. Pedersen IB, Knudsen N, Carlé A, et al, 2011 A cautious iodization programme bringing iodine intake to a low recommended level is associated with an increase in the prevalence of thyroid autoantibodies in the population. Clin Endocrinol 75: 120-126.
15. Rasmussen LB, Carlé A, Jørgensen T, et al, 2008 Iodine intake before and after mandatory iodization in Denmark: results from the Danish Investigation of Iodine Intake and Thyroid Diseases (DanThyr) study. Br J Nutr 100: 166-173.
16. Laurberg P, Pedersen KM, Hreidarsson A, Sigfusson N, Iversen E, Knudsen PR, 1998 Iodine intake and the pattern of thyroid disorders: a comparative epidemiological study of thyroid abnormalities in the elderly in Iceland and in Jutland, Denmark. J Clin Endocrinol Metab 83: 765-769.
17. Szabolcs I, Podoba J, Feldkamp J, et al, 1997 Comparative screening for thyroid disorders in old age in areas of iodine deficiency, long-term iodine prophylaxis and abundant iodine intake. Clin Endocrinol 47: 87-92.
18. Laurberg P, Pedersen IB, Pedersen KM, Vestergaard H, 1999 Low incidence rate of overt hypothyroidism compared with hyperthyroidism in an area with moderately low iodine intake. Thyroid 9: 33-38.
19. Teng X, Shan Z, Chen Y, et al, 2011 More than adequate iodine intake may increase subclinical hypothyroidism and autoimmune thyroiditis: a cross-sectional study based on two Chinese communities with different iodine intake levels. Eur J Endocrinol 164: 943-950.
20. Melse-Boonstra A, Gowachirapant S, Jaiswal N, Winichagoon P, Srinivasan K, Zimmermann MB, 2012 Iodine supplementation in pregnancy and its effect on child cognition. J Trace Elem Med Biol 26: 134-136.
21. Andersen S, Iversen F, Terpling S, Pedersen KM, Gustenhoff P, Laurberg P, 2009 More hypothyroidism and less hyperthyroidism with sufficient iodine nutrition compared to mild iodine deficiency – a comparative population-based study of older people. Maturitas 64: 126-131.
22. Andersen S, Pedersen KM, Iversen F, et al, 2008 Naturally occurring iodine in humic substances in drinking water in Denmark is bioavailable and determines population iodine intake. Br J Nutr 99: 319-325.
23. Laurberg P, 1994 Iodine intake – what are we aiming at? J Clin Endocrinol Metab 79: 17-19.
24. Mokdad AH, Marks JS, Stroup DF, Gerberding JL, 2004 Actual causes of death in the United States, 2000. JAMA 291: 1238-1245.
25. Carlé A, Pedersen IB, Knudsen N, et al, 2012 Smoking cessation is followed by a sharp but transient rise in the incidence of overt autoimmune hypothyroidism - A population-based case-control study. Clin Endocrinol 77: 764-772.
26. WHO, 2008 Report on the Global Tobacco Epidemic: The MPOWER package. World Health Organization.
27. Strieder TG, Prummel MF, Tijssen JG, Endert E, Wiersinga WM, 2003 Risk factors for and prevalence of thyroid disorders in a cross-sectional study among healthy female relatives of patients with autoimmune thyroid disease. Clin Endocrinol 59: 396-401.
28. Belin RM, Astor BC, Powe NR, Ladenson PW, 2004 Smoke exposure is associated with a lower prevalence of serum thyroid autoantibodies and thyrotropin concentration elevation and a higher prevalence of mild thyrotropin concentration suppression in the third National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 89: 6077-6086.
29. Pedersen IB, Laurberg P, Knudsen N, et al, 2008 Smoking is negatively associated with the presence of thyroglobulin autoantibody and to a lesser degree with thyroid peroxidase autoantibody in serum: a population study. Eur J Endocrinol 158: 367-373.
30. Effraimidis G, Tijssen JG, Wiersinga WM, 2009 Discontinuation of smoking increases the risk for developing thyroid peroxidase antibodies and/or thyroglobulin antibodies: a prospective study. J Clin Endocrinol Metab 94: 1324-1328.
31. Knudsen N, Bulow I, Laurberg P, Perrild H, Ovesen L, Jørgensen T, 2002 High occurrence of thyroid multinodularity and low occurrence of subclinical hypothyroidism among tobacco smokers in a large population study. J Endocrinol 175: 571-576.
32. Vestergaard P, 2002 Smoking and thyroid disorders – a meta-analysis. Eur J Endocrinol 146: 153-161.
33. Flegal KM, Troiano RP, Pamuk ER, Kuczmarski RJ, Campbell SM, 1995 The influence of smoking cessation on the prevalence of overweight in the United States. N Engl J Med 333: 1165-1170.
34. Yeh HC, Duncan BB, Schmidt MI, Wang NY, Brancati FL, 2010 Smoking, smoking cessation, and risk for type 2 diabetes mellitus: a cohort study. Ann Intern Med 152: 10-17.
35. Oba S, Noda M, Waki K, et al, 2012 Smoking cessation increases short-term risk of type 2 diabetes irrespective of weight gain: The Japan Public Health Center-Based Prospective Study. PLoS One 7: e17061.
36. Laurberg P, Andersen S, Knudsen N, Ovesen L, Nøhr SB, Pedersen IB, 2002 Thiocyanate in food and iodine in milk: from domestic animal feeding to improved understanding of cretinism. Thyroid 12: 897-902.
37. Laurberg P, Nøhr SB, Pedersen KM, Fuglsang E, 2004 Iodine nutrition in breast-fed infants is impaired by maternal smoking. J Clin Endocrinol Metab 89: 181-187.
38. Ditzel J, Lervang HH, 2010 Lifestyle diseases and cardiovascular risk factors are interrelated to deficiencies of major substrates in ATP synthesis. Vasc Health Risk Manag 6: 829-836.
39. Caturegli P, De Remigis A, Ferlito M, et al, 2012 Anatabine ameliorates experimental autoimmune thyroiditis. Endocrinology 153: 4580-4587.
40. Stampfer MJ, Colditz GA, Willett WC, Speizer FE, Hennekens CH, 1988 A prospective study of moderate alcohol consumption and the risk of coronary disease and stroke in women. N Engl J Med 319: 267-273.
41. Cohen S, Tyrrell DA, Russell MA, Jarvis MJ, Smith AP, 1993 Smoking, alcohol consumption, and susceptibility to the common cold. Am J Pub Health 83: 1277-1283.
42. Simon JA, Grady D, Snabes MC, Fong J, Hunninghake DB, 1998 Ascorbic acid supplement use and the prevalence of gallbladder disease. Heart & Estrogen-Progestin Replacement Study (HERS) Research Group. J Clin Epidemiol 51: 257-265.
43. Curhan GC, Willett WC, Rimm EB, Spiegelman D, Stampfer MJ, 1996 Prospective study of beverage use and the risk of kidney stones. Am J Epidemiol 143: 240-247.
44. Obisesan TO, Hirsch R, Kosoko O, Carlson L, Parrott M, 1998 Moderate wine consumption is associated with decreased odds of developing age-related macular degeneration in NHANES-1. J Am Geriatr Soc 46: 1-7.
45. Orgogozo JM, Dartigues JF, Lafont S, et al, 1997 Wine consumption and dementia in the elderly: a prospective community study in the Bordeaux area. Rev Neurol 153: 185-192.
46. Lieberoth S, Backer V, Kyvik KO, et al, 2012 Intake of alcohol and risk of adult-onset asthma. Resp Med 106: 184-188.
47. Carle A, Pedersen IB, Knudsen N, et al, 2012 Moderate alcohol consumption may protect against overt autoimmune hypothyroidism – a population-based case-control study. Eur J Endocrinol 167: 483-490.
48. Effraimidis G, Tijssen JGP, Wiersinga WM, 2012 Alcohol consumption as a risk factor for autoimmune thyroid disease: a prospective study. Eur Thyroid J 1: 99-104.
49. Knudsen N, Pedersen IB, Laurberg P, Perrild H, Ovesen L, Jorgensen T, 2001 Alcohol consumption is associated with reduced prevalence of goitre and solitary thyroid nodules. Clin Endocrinol 55: 41-46.
50. Hegedus L, Rasmussen N, Ravn V, Kastrup J, Krogsgaard K, Aldershvile J, 1988 Independent effects of liver disease and chronic alcoholism on thyroid function and size: the possibility of a toxic effect of alcohol on the thyroid gland. Metabolism 37: 229-233.
51. Wang J, Pan HF, Ye DQ, Su H, Li XP, 2008 Moderate alcohol drinking might be protective for systemic lupus erythematosus: a systematic review and meta-analysis. Clin Rheum 27: 1557-1563.
52. Di Giuseppe D, Alfredsson L, Bottai M, Askling J, Wolk A, 2012 Long term alcohol intake and risk of rheumatoid arthritis in women: a population based cohort study. BMJ 345: e4230.
53. Rasouli B, Ahlbom A, Andersson T, et al, 2012 Alcohol consumption is associated with reduced risk of type 2 diabetes and autoimmune diabetes in adults: results from the Nord-Trondelag health study. Diabet Med 30: 56-64.
54. Mandrekar P, Catalano D, Dolganiuc A, Kodys K, Szabo G, 2004 Inhibition of myeloid dendritic cell accessory cell function and induction of T cell energy by alcohol correlates with decreased IL-12 production. J Immunol 173: 3398-3407.
55. Verma S, Alexander CM, Carlson MJ, Tygrett LT, Waldschmidt TJ, 2008 B-cell studies in chronic ethanol mice. Methods Mol Biol 447: 295-323.
56. Fan J, Edsen-Moore MR, Turner LE, et al, 2011 Mechanisms by which chronic ethanol feeding limits the ability of dendritic cells to stimulate T-cell proliferation. Alcohol Clin Exp Res 35: 47-59.
57. Hollowell JG, Staehling NW, Flanders WD, et al, 2002 Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87: 489-499.
Address for correspondence:
Peter Laurberg, Professor, M.D., Department of
Endocrinology and Medicine, Aalborg Hospital, Aalborg
University, DK - 9000 Aalborg, Denmark
E-mail: peter.laurberg@rn.dk
Received 30-08-12, Accepted 29-11-12