Departments of 1Laboratory Medicine, Division of Pathology and 2Neurosurgery, St. Michael’s Hospital, University of Toronto, Toronto, ON, Canada, 3Department of Pathology, Mayo Clinic, Rochester, MN, USA and 4Department of Pathology, Trillium Health Centre, Mississauga, ON, Canada
We report the case of a 49-year-old woman presenting with Cushing disease and visual disturbance. An atypical, aggressive, invasive pituitary tumor regrew despite several surgeries. Detailed morphologic investigation by histology, immunohistochemistry and electron microscopy documented a Crooke cell adenoma, a rare form of ACTH-producing pituitary tumor. Recognition of such adenomas is of importance given their aggressive behavior and tendency to recur. More studies are needed to explain the pathobiology of this not invariably functional pituitary adenoma.
Crooke cell adenoma, Histology, Immunohistochemistry, Neoplasm, Ultrastructure
INTRODUCTION
Pituitary tumors producing ACTH are a biologically disparate group of adenomas. Several subtypes can be distinguished on the basis of clinical, histologic, immunohistochemical and ultrastructural features.1-5 Their correct classification is of importance given differences in biologic behavior, therapy and prognosis.6-13 Endocrinologically functional ACTH adenomas are associated with Cushing disease or Nelson syndrome.14,15 Clinically nonfunctioning tumors are classified as “silent corticotroph adenomas” of subtypes 1 and 2.3,4 Both pituitary carcinoma and the recently described pituitary blastoma often produce excess ACTH with resultant Cushing disease.16-19 Crooke cell adenoma is relatively rare but not invariably functional.20
Herein, we report the case of a 49-year-old woman presenting with Cushing disease and visual disturbance. The tumor invaded the cavernous sinus and was only partly removed by transsphenoidal surgery. The tumor regrew and several surgeries were performed. No metastases were documented. Detailed morphologic investigation including histology, immunohistochemistry and electron microscopy revealed the characteristic features of Crooke cell adenoma.
CASE REPORT
The patient, a 49-year-old woman, presented at the age of 43 with Cushing disease and visual field deficit. Magnetic resonance imaging (MRI) showed a large, multi-lobed sellar tumor, an unusual finding since only 8% of patients with Cushing disease present with mass effects including visual symptoms.3,20
In 2004, despite three separate transsphenoidal resections for recurrent tumor, the patient’s initial complaint of visual deficit persisted. After much of the tumor was removed, mild improvement of Cushing disease was noted. Follow-up consisted of radiotherapy, subsequent treatment of temozolomide (85 mg p.o. daily) and serial neuroimaging. The tumor responded to temozolomide. Treatment was discontinued for a short interval after which symptoms resurfaced, prompting cabergoline (1 mg weekly) treatment, however with no effect. Five years after initial surgery, the patient presented with rapidly progressing left temporal visual field loss, ataxia and proximal muscle weakness. MRI revealed a sellar, right parasellar and suprasellar tumor measuring 1.5cm TR x 1.5 cm AP x 1.3 cm SI (Figure 1). The parasellar tumor was stable. The tumor encased and constricted the left carotid artery. A stereotactically guided, endoscopic, subtotal resection of the tumor was performed. Adjuvant therapy consisted of fractionated stereotactic radiotherapy (50 Gy in 25 fractions) and subsequent temozolomide chemotherapy (85 mg p.o. daily). No metastases were apparent. Positron emission tomography (PET) scan and cytology on the cerebrospinal fluid were not performed.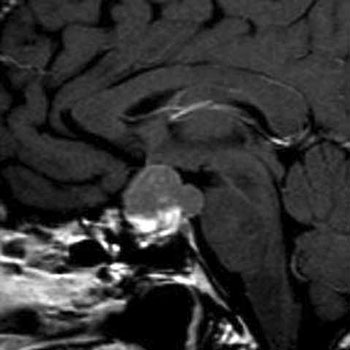
Figure 1. Magnetic resonance imaging (MRI) demonstrates a large suprasellar tumor. Sagittal view.
MORPHOLOGIC METHODS
The majority of the specimen was fixed in 10% buffered formalin, routinely processed for light microscopy and paraffin-embedded. Hematoxylin-eosin, periodic acid-Schiff and Gordon-Sweet silver stains were applied. Five-micron sections were subjected to immunohistochemical (streptavidin-biotin-peroxidase complex method) staining for the entire spectrum of adenohypophysial hormones including: growth hormone (GH) (Ventana, Tucson, AZ; polyclonal; 1:50); prolactin (PRL) (Ventana: SPM108; 1:1000); adrenocorticotropic hormone (ACTH) (Ventana, Tucson, AZ; polyclonal; 1:200); luteinizing hormone (LH) (Ventana, Tucson, AZ; ZAL11; 1:3000); follicle-stimulating hormone (FSH) (Ventana, Tucson, AZ; INN-hFSH-60; 1:3000); thyroid-stimulating hormone (TSH) (Ventana, Tucson, AZ; QB2/6; 1:400); and α-subunit (Biogenex, Sanraman, CA; monoclonal; 1:50). Antibody sources, clonality and dilution have been previously described.21,22 Additional antibodies applied included: MIB-1 (Dako, Carpinteria, CA; MIB-1; 1:20); p53 (Dako, Carpinteria, CA; DO7; 1:2000); p27 (Dako, Carpinteria, CA; SX53G8; 1:50); topoisomerase-2 alpha (Dako, Carpinteria, CA; Ki-S1;1:100); CAM 5.2 (Ventana, Tucson, AZ;); O(6)-Methylguanine-DNA methyltransferase (MGMT) (Thermofisher, Fremont, CA; MG3.1; 1:50); and vascular endothelial growth factor (VEGF) (Santa Cruz Biotechnology, Santa Cruz, CA; A-20; 1:50). For electron microscopy, tissue was fixed in 2.5% glutaraldehyde, osmicated, dehydrated in graded ethanol, processed through propylene oxide, embedded in epoxy resin and studied on a Hitachi H7650 digital electron microscope.21,22
PATHOLOGIC FINDINGS
Based upon the morphologic features, a diagnosis of Crooke cell adenoma was made. The tumor resected in 2009 showed features similar to those of the tumor initially resected in 2004. Histologically, it consisted of amphophilic, polygonal cells exhibiting a diffuse growth pattern and featuring perinuclear ring-like hyaline material. The cells of the tumor were PAS-positive and showed no significant cellular or nuclear pleomorphism (Figure 2). There was a loss of the acinar structure and the reticulin fiber network, a characteristic feature of pituitary tumors. No mitotic figures, necrosis, inflammatory cells or calcification were observed in the 2004 specimen. The 2009 specimen featured large zones of necrosis. The cells of all specimens showed extensive Crooke hyalinization. Cytoplasmic immunoreactivity for ACTH and perinuclear low molecular weight keratin staining were evident (Figures 3, 4). The tumor cells were negative for other adenohypophysial hormones. The MIB-1 nuclear labeling index ranged from 5% to 8%. Almost all tumor cell nuclei were immunopositive for topoisomerase-2 alpha. The tumor cells showed 80% immunoreactivity for p27, few cells showing p53 nuclear immunostaining (Figure 5). Nearly all the tumor cell nuclei were immunonegative for MGMT. Cytoplasmic reactivity for low molecular weight keratin and VEGF was noted in nearly all tumor cells (Figure 6).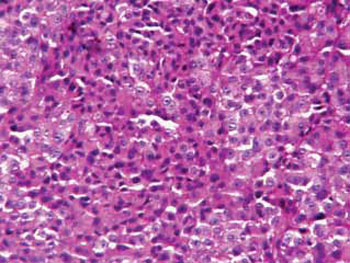
Figure 2. Light microscopy demonstrates a cellular pleomorphic, PAS positive tumor showing Crooke hyaline change. PAS immunostain. Original magnification: X250.
Figure 3. The tumor cells are immunoreactive for ACTH. Many tumor cells show marked Crooke hyalinization. Immunostaining for ACTH. Original magnification: X250.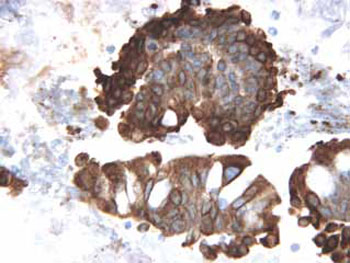
Figure 4. The tumor cells are immunopositive for low molecular weight keratin. Immunostaining for low molecular weight keratin. Original magnification: X400.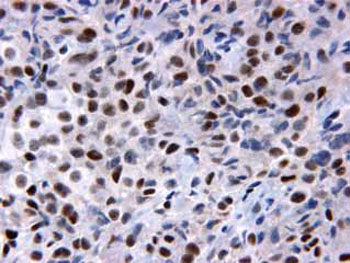
Figure 5. Many tumor cell nuclei are immunopositive for p27. Immunostaining for p27. Original magnification: X400.
Figure 6. Almost all tumor cells show cytoplasmic immunopositivity for VEGF. Crooke hyalinization is prominent. Immunostaining for VEGF. Original magnification: x400.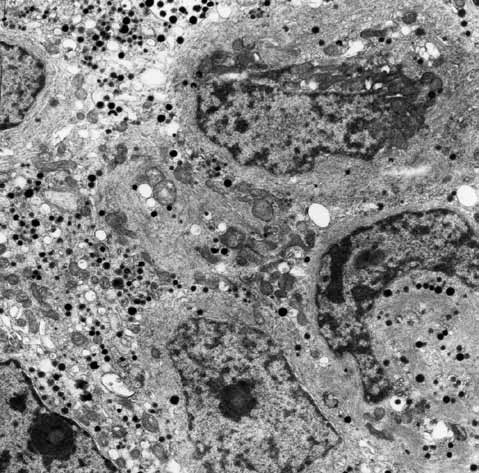
Figure 7. Electron microscopy shows the presence of many microfilaments in the cytoplasm of tumor cells. Transmission electron micrograph. Original magnification: X3000.
Ultrastructural studies showed the adenoma to consist of large, ovoid cells with spherical nuclei containing macronucleioli and delicate heterochromatin. Their cytoplasm contained numerous intermediate cytokeratin filaments corresponding to Crooke hyaline, which obscured both rough endoplasmic reticulum and Golgi complexes (Figure 7). Secretory granules were both localized to the Golgi region and peripherally displaced within the cells. Most measured 100-200 nm and were smaller than those usually seen in corticotroph cells (200-350 nm). The mitochondria, although displaced by filaments, exhibited normal morphologic features.
DISCUSSION
Crooke hyaline change was first described in 1935 by the endocrinologist A. C. Crooke who, while studying with Dorothy Russell at the London Hospital, reported his findings in 12 pituitaries of patients with Cushing disease.23 It affects non-neoplastic corticotrophs in any setting of glucocorticoid excess, including that due to the administration of glucocorticoids.
Corticotrophs of the anterior pituitary normally contain small numbers of perinuclear cytokeratin filament bundles. Persistently elevated blood cortisol levels result in accumulation of these cytokeratin filaments with reduction, displacement of cytoplasmic organelles and secretory granules. Cytoplasmic accumulation of filaments results in a distinctive cellular alteration known as Crooke hyaline change in which a circumferential ring of keratin gives the cell cytoplasm a distinctly hyalinized appearance on hematoxylin-eosin and PAS stains.23-28
On rare occasions, Crooke change also occurs in ACTH-producing pituitary adenomas.24-26 Such adenomas display variable but often extensive Crooke hyalinization, as demonstrated in our case. Patients with Crooke cell adenoma may have previously shown evidence of hypercortisolism and Cushing disease.29,30 In other instances, the tumor is functionally silent and unassociated with Cushing disease.26,31,32 In clinicopathologic terms, Crooke cell tumors are unique and should not be confused with typical endocrinologically active corticotroph adenomas.
Although first described by Felix et al,24 it was the large series of George et al26 that clearly defined the endocrine, radiologic and behavioral profile of Crooke cell adenoma. Innately aggressive, nearly all are atypical macroadenomas, being invasive and prone to recurrence, and are associated with significant morbidity.6,8-10,26,33-37 Rare examples of Crooke cell carcinomas have also been reported.17,18,34-36 By definition, such tumors undergo cerebrospinal and/or systemic metastases.38-40
Cell proliferation markers are routinely used to assess tumor aggressiveness. In the present case, increased MIB-1 nuclear labeling was accompanied by surprisingly low p53 expression. This is unusual in that p53 staining is associated with large tumor size, invasiveness and aggressive behavior.9,33,41 p53 is a tumor suppressor protein playing an important role in cell response to DNA injury. p53 activation may result in cell cycle arrest, DNA repair and apoptosis.42 In our case, strong and diffuse immunopositivity for topoisomerase 2 alpha and VEGF was also apparent. Both of these biomarkers have been found to be good predictors of tumor aggressiveness as well as indicators of poor or unfavorable prognosis.43,44 An unexpected finding in this study was the high percentage of cell nuclei expressing p27, a protein that plays a regulatory role in the cell cycle by inhibiting the activity of cyclin dependent kinases (CDKs), resulting in suppression of cell cycle progression and causing G1 arrest. Lloyd et al, showed that there is a marked decrease in the expression of p27 in malignant endocrine neoplasms compared to normal tissues. This suggests that rapid cell proliferation should be accompanied by a decrease in the number of p27 immunoreactive tumor cell nuclei.45 We cannot provide a conclusive explanation as to why p27 expression was high in the aggressive Crooke cell tumor investigated by us. It was recently reported that p27 may have an opposing role as well: it may function as a tumor suppressor but may also have an oncogenic effect.46 Aside from low p53 and high p27 stainings, our findings are consistent with the view that biomarkers are useful in predicting tumor behavior.9,33,47-50
On balance, studies of Crooke cell adenomas pose more questions than answers. No explanation can be provided as to why Crooke hyalinization is rare in corticotroph tumors and why Crooke cell adenomas are sensitive to glucocorticoid excess, are aggressive and in some cases secrete large amounts of ACTH despite massive cytoplasmic accumulation of microfilaments, a feature of suppressed activity in normal corticotrophs.
ACKNOWLEDGMENT
The authors acknowledge the generous support of the Jarislowsky and Lloyd Carr-Harris Foundations. Also appreciated is the excellent secretarial assistance of Mrs. Denise Chase of the Mayo Clinic Transcription Service.
REFERENCES
1.Kovacs K, Scheithauer BW, Horvath E, Lloyd RV, 1996 The World Health organization classification of adenohypophysial neoplasms. A proposed five-tier scheme. Cancer 78: 502-510.
2.Kovacs K, Horvath E 1986 Tumors of the pituitary gland. In: Hartmann WH, Sobin LH (eds) Atlas of tumor pathology, 2nd series, Fascicle 21. Armed Forces Institute of Pathology, Washington, DC.
3.Horvath E, Kovacs K, Killinger DW, Smyth HS, Platts ME, Singer W, 1980 Silent corticotropic adenomas of the human pituitary gland: a histologic, immunologic, and ultrastructural study. Am J Pathol 98: 617-638.
4.Horvath E, Kovacs K, Smyth HS, et al, 1988 A novel type of pituitary adenoma: morphological features and clinical correlations. J Clin Endocrinol Metab 66: 1111-1118.
5.Hassoun J, Charpin C, Jaquet P, Lissitzky JC, Grisoli F, Toga M, 1982 Corticolipotropin immunoreactivity in silent chromophobe adenomas: A light and electron microscopic study. Arch Pathol Lab Med 106: 25-30.
6.Ezzat S, Asa SL, Couldwell WT, et al, 2004 The prevalence of pituitary adenomas: a systematic review. Cancer 101: 613-619.
7.Asa SL, Ezzat S, 1998 The cytogenesis and pathogenesis of pituitary adenomas. Endocr Rev 19: 798-827.
8.Terada T, Kovacs K, Stefaneanu L, Horvath E, 1995 Incidence, pathology, and recurrence of pituitary adenomas: study of 647 unselected surgical cases. Endocr Pathol 6: 301-310.
9.Thapar K, Kovacs K, Scheithauer BW, et al, 1996 Proliferative activity and invasiveness among pituitary adenomas and carcinomas: an analysis using the MIB-1 antibody. Neurosurgery 38: 99-106.
10.Hsu DW, Hakim F, Biller BM, et al, 1993 Significance of proliferating cell nuclear antigen index in predicting pituitary adenoma recurrence. J Neurosurg 78: 753-761
11.Minematsu T, Miyai S, Kajiya H, et al, 2005 Recent progress in studies of pituitary tumor pathogenesis. Endocrine 28: 37-41.
12.Levy A, 2004 Pituitary disease: presentation, diagnosis, and management. J Neurol Neurosurg Psychiatry 75: iii47-52.
13.Arafah BM, Nasrallah MP, 2001 Pituitary tumors: pathophysiology, clinical manifestations and management. Endocr Relat Cancer 8: 287-305.
14.Kovacs K, Hovath E, Kerenyi NA, Sheppard RH, 1976 Light and electron microscopic features of a pituitary adenoma in Nelson’s syndrome. Am J Clin Pathol 65: 337-343.
15.Kovacs K, Horvath E, Coire C, et al, 2006 Pituitary corticotroph hyperplasia preceding adenoma in a patient with Nelson’s syndrome. Clin Neuropathol 25: 74-80.
16.Scheithauer BW, Kovacs K, Horvath E, et al, 2008 Pituitary blastoma. Acta Neuropathol 116: 657-666.
17.Pernicone PJ, Scheithauer BW, Sebo TJ, et al, 1997 Pituitary carcinoma: a clinicopathologic study of 15 cases. Cancer 79: 804-812.
18.Zafar MS, Mellinger RC, Chason JL, 1984 Cushing’s disease due to pituitary carcinoma. Henry Ford Hosp Med J 32: 61-65.
19.Holthouse DJ, Robbins PD, Kahler R, Knuckey N, Pullan P, 2001 Corticotroph pituitary carcinoma: case report and literature review. Endocr Pathol 12: 329-341.
20.Scheithauer BW, Jaap AJ, Horvath E, et al, 2002 Clinically silent corticotroph tumors of the pituitary gland. Neurosurgery 47: 723-729.
21.Kovacs K, Stefaneanu L, Ezzat S, Smyth HS, 1994 Prolactin-producing pituitary adenoma in a male-to-female transsexual patient with protracted estrogen administration: a morphologic study. Arch Pathol Lab Med 118: 562-566.
22.Rotondo F, Kovacs K, Horvath E, Bell CD, Lloyd RV, Scheithauer BW, 2006 Immunohistochemical expression of nestin in the non-tumorous hypophysis and in pituitary neoplasms. Acta Neuropathol 111: 272-277.
23.Crooke AC, 1935 A change in the basophil cells of the pituitary gland common to conditions which exhibit the syndrome attributed to basophil adenoma. J Pathol Bacteriol 41: 339-349.
24.Felix UA, Horvath E, Kovacs K, 1981 Massive Crooke’s hyalinization in corticotroph cell adenomas of the human pituitary: a histological, immunocytological and electron microscopic study of three cases. Acta Neurochir (Wien) 58: 235-243.
25.Neumann PE, Horoupian DS, Goldman JE, et al, 1984 Cytoplasmic filaments of Crooke’s hyaline change belong to the cytokeratin class: an immunocytochemical and ultrastructural study. Am J Pathol 116: 214-222.
26.George DH, Scheithauer BW, Kovacs K, et al, 2003 Crooke’s cell adenoma of the pituitary: an aggressive variant of corticotroph adenoma. Am J Surg Pathol 27: 1330-1336.
27.Horvath E, Kovacs K, Josse R, 1983 Pituitary corticotroph cell adenoma with marked abundance of microfilaments. Ultrastruct Pathol 5: 249-255.
28.Eschbacher JM, Coons SW, 2006 Cytokeratin CK20 is a sensitive marker for Crooke’s cells and the early cytoskeletal changes associated with hypercortisolism within pituitary corticotrophs. Endocr Pathol 17: 365-376.
29.Horvath E, Scheithauer BW, Kovacs K, Lloyd RV 2002 Hypothalamus and pituitary. In: Graham DI, Lantos PL (Eds). Greenfield’s Neuropathology Vol 1, New York, Arnold Publishers, NY; pp, 983-1062.
30.Ikeda H, Yoshimoto T, Ogawa Y, Mizoi K, Murakami O, 1997 Clinico-pathological study of Cushing’s disease with large pituitary adenoma. Clin Endocrinol 46: 669-679.
31.Saeger W, Ludecke DK, Geisler F 1990 The anterior lobe in Cushing’s disease/syndrome. In Cushing’s Syndrome, and Other Hypercortisolemic States. Progress on Endocrine Research and Surgery, vol 5, pp, 147–156. DK Ludecke, G Chrousos, G Tolis (eds), New York: Raven Press.
32.Ludecke DK, Flitsch J, Knappe UJ, Saeger W, 2001 Cushing’s disease: a surgical view. J Neuro-Oncol 54: 151-166.
33.Thapar K, Scheithauer BE, Kovacs K, Pernicone PJ, Laws ER Jr, 1996 P-53 expression in pituitary adenomas and carcinomas. Correlation with invasiveness and tumor growth fractions. Neurosurgery 38: 765-770.
34.Scheithauer BW, Kovacs KT, Laws ER, Randall RV, 1986 Pathology of invasive pituitary tumors with special reference to functional classification. Neurosurgery 65: 733-744.
35.Gaffey TA, Scheithauer BW, Lloyd RV, et al, 2002 Corticotroph carcinoma of the pituitary: a clinicopathological study. J Neurosurg 96: 352–360.
36.Scheithauer BW, Gaffey TA, Lloyd RV, et al 2006 Pathobiology of pituitary adenomas and carcinomas. Neurosurgery 59: 341-53.
37.Kovacs K, Diep CC, Horvath E, et al, 2005 Prognostic indicators in an aggressive pituitary Crooke’s cell adenoma. Can J Neurol Sci 32: 540-545.
38.Takeshita A, Inoshita N, Taguchi M, et al, 2009 High incidence of low O6-methylguanine DNA methyltransferase expression in invasive macroadenomas of Cushing’s disease. Europ J Endocrinol 161: 553-559.
39.Grossman AB, 2005 Clinical review: diagnosis and management of pituitary carcinomas. J Clin Endocrinol Metab 90: 3089-3099.
40.Scheithauer BW, Kurtkaya-Yapicier O, Kovacs KT, Young WF Jr, Lloyd RV, 2005 Pituitary carcinoma: a clinicopathological review. Neurosurgery 56: 1066-1074.
41.Pernicone PJ, Scheithauer BW 1993 Invasive pituitary adenomas and pituitary carcinomas. In Lloyd RV (eds), Surgical pathology of the pituitary gland. Philadelphia, WB Sanders, Co, pp, 121-136.
42.Levine AJ, 1997 the cellular gatekeeper for growth and division. Cell 88: 323-331.
43.Lidhar R, Korbonits M, Jordan S, et al, 1999 Low expression of the cell cycle inhibitor p27Kip1 in normal corticotroph cells, corticotroph tumors, and malignant pituitary tumors. J Clin Endocrinol Metab 84: 3823-3830.
44.Vidal S, Kovacs K, Horvath E, et al, 2002 Topoisomerase II alpha expression in pituitary adenomas and carcinomas: relationship to tumor behavior. Mod Pathol 15: 1205-1212.
45.Lloyd RV, Jin L, Qian X, Kulig E, 1997 Aberrqant p27kip1 expression in endocrine and other tumors. Amer J Pathol 150: 401-407.
46.Serres MP, Zlotek-Klotkiewicz E, Concha C, et al, 2011 Cytoplasmic p27 is oncogenic and cooperates with Ras both in vivo and in vitro. Oncogene 30: 2846-2858.
47.Sumi T, Stefaneanu L, Kovacs K, Asa S, Rindi, 1993 Immunohistochemical study of p53 protein in human and animal pituitary tumors. Endocr Pathol 4: 95-99.
48.Kontogeorgos G, 2006 Predictive markers of pituitary adenoma behavior. Neuroendocrinology 83: 179-188.
49.Saeger W, 2004 Proliferation markers and cell cycle inhibitors in pituitary adenomas. Front Horm Res 32: 110-126.
50. Wolfsberger S, Wunderer J, Zachenhofer I, et al, 2004 Expression of cell proliferation markers in pituitary adenomas- correlation and clinical relevance of MIB-1 and anti-topoisomerase-II alpha. Acta Neurochir (Wien) 146: 831-839.
Address for correspondence:
Fabio Rotondo, BSc, Department of Laboratory Medicine,
Division of Pathology, St. Michael’s Hospital, 30 Bond Str.,
Toronto, Ontario, Canada, M5B1W8, Tel.: 416-864-5851,
Fax: 416-864-5648, E-mail: rotondof@smh.ca
Received 25-10-11, Revised 02-12-11, Accepted 06-01-12