Department of Biological Chemistry, Athens University Medical School, Athens, Greece
The classical phytoestrogens, so far known, constitute a group of plant-derived compounds which include mainly isoflavones, lignans, coumestanes, stilbenes and the flavonoids quercetin and kaempherol. The discovery of many more novel estrogen-like compounds in the plant kingdom demonstrates that the spectrum of phytoestrogens in nature is expanding. The classical as well as the novel phytoestrogens show a complex mode of action via interaction with the nuclear estrogen receptor isoforms ERα and ERβ, exhibiting either estrogen-agonist or estrogen-antagonist effects. Their final biological activity, assessed by cell culture assay systems, animal studies and clinical trials, depends on multiple factors such as the chemical structure of the phytoestrogen, the kind of tissue and cell type, the intrinsic estrogenic status, the route of administration, the metabolism as well as the time and the level of exposure. They are characterized by high tissue specificity and dose-dependent activity. However, although phytoestrogen intake as food or dietary supplements, in particular soya products and the isoflavones genistein and daidzein, has been associated with “health promoting effects”, some data indicate increased disease risk. Evidently, phytoestrogen supplementation should be viewed with caution until further studies satisfactorily delineate the effects of individual phytoestrogens on human health and disease.
Breast cancer, Estrogen receptor alpha, Estrogen receptor beta, Colon cancer, Coro-nary heart disease, Flavonoids, Osteoporosis, Phytoestrogens, Prostate cancer, SERMs
INTRODUCTION
Estrogens are steroid hormones with a complex mode of action, characterized by high tissue specificity and dose-dependent activity. They exert pleiotropic effects on a diverse range of tissues, such as ovary, testis, prostate, breast, uterus, bone, liver, immune system, cardiovascular and central nervous system.1 Estrogens promote breast and endometrial cancer in women and exacerbate autoimmune diseases, whereas the loss of estrogens during menopause has been correlated with osteoporosis, coronary heart disease, depression and neurodegeneration. Compounds which antagonize the estrogenic effects (antagonists) in some tissues, such as breast and uterus, while mimicking the estrogens effects (agonists) in other tissues, such as bone, brain and cardiovascular cells, are known as selective estrogen receptor modulators (SERMs).2
The substitution during menopause of endogenous estrogens with exogenous estrogens, widely known as hormonere placement therapy (HRT), isofenormous importance. However, the Women’s Health Initiative study raised serious concerns about the safety of HRT, resulting in low compliance of women due to the fear of breast and uterus cancer development.3 SERMs are considered as a new alternative postmenopausal therapy, since they improve bone function with minimal risk for breast and uterine cancer. A large body of evidence has demonstrated that endogenous estrogens and SERMs mediate their effects by modulating the intracellular receptor subtypes estrogen receptor alpha (ERα) and estrogen receptor beta (ERβ), both members of the nuclear receptor superfamily. The binding of estrogens or SERMs to estrogen receptors (ERα and ERβ) initiates a series of events resulting in transcriptional regulation of specific genes, RNA and protein synthesis.
Phytoestrogens are a large family of plant-derived estrogens possessing significant estrogen agonist/antagonist activity. These, naturally occurring molecules, include the isoflavonoids, the lignans, the coumestanes, the stilbens and the flavonoids quercetin and kaempherol. Their effects, mediated via the estrogen receptor subtypes ERα and ERβ, have been shown to be cell type/tissue specific and dose-dependent. The so far known phytoestrogens may act as “natural” SERMs and may possibly be considered for the prevention of postmenopausal osteoporosis and cardiovascular disease without an adverse effect on breast and uterus. However, since data are still controversial, further clinical studies are needed to delineate satisfactorily the health effects of phytoestrogens in specific population groups.4
Current data demonstrate that many more novel compounds are continuously being identified in nature exhibiting estrogenic/ antiestrogenic properties, indicating that our knowledge about phytoestrogens in nature is expanding.
This review outlines the major categories of the so far known phytoestrogens, provides the current knowledge regarding their overall biological effects and briefly describes their estrogen-like mediated mechanism of action as well as their potential in triggering other signaling pathways. The review presents the laboratory evidence for the “novel” plant-derived compounds and their modulatory activity on the ER subtypes. In order to have a better understanding of the molecular mechanisms by which phytoestrogens modulate estrogen receptor-mediated activity, it was considered essential to briefly explicate the structure of estrogen receptors and their encoding respective genes as well as some key stages in the estrogen signaling cascade.
In this review the in vitro and in vivo assays, assessing the estrogenic/antiestrogenic potential of phytochemicals, are also presented and their advantages and disadvantages are discussed.
ESTROGEN RECEPTORS
Structure Estrogens exert their effects via their respective receptors estrogen receptor alpha (ERα) and beta (ERβ), which are members of the nuclear receptor superfamily.5,6 Estrogen receptors have been identified in all types of human cells and have distinct tissue expression patterns.1 Target organs with a high estrogen receptor content are those related to reproductive functions such as the mammary gland, the ovaries and the uterus, known to be “classical” targets for estrogens. Bone, the cardiovascular system, the brain, the immune system and the liver are characterized by lower ER content.
The primary structure of the estrogen receptors has been fully characterized and their respective genes, ESR1 and ESR2 have been cloned and sequenced.7,8 The encoded protein molecules contain six functional domains (Figure 1): the transcriptional regulation (A/B) domain (AF-1) which is highly immunoreactive, the DNA binding domain (C) which is responsible for binding to specific DNA sequences known as estrogen response elements (ERE), the domain D (between C and E domains, hinge region) and the domains E and F responsible for ligand binding, dimerization and transcriptional activation (AF-2). ERα and ERβ differ in size (595 and 530 amino acids, respectively). ERα and ERβ exhibit high homology in their DNA binding domain (96%), partial homology in the ligand binding domain (53%), but very low in their A/B domain (30%) located in the amino-terminal part of the molecules. Due to high homology in the C domain, ERα and ERβ are likely to bind to various ligands with similar specificity and affinity. In ERα and ERβ the conformation of the α-helix 12 (H12) strongly depends on the nature of the ligand and evidently different ligands induce different receptor conformations, thus affecting the agonist/antagonist potency of ligands.9 Differential splicing of the ERα and ERβ genes results in various ERα and ERβ splicing variants lacking certain exons. Moore et al (1998) described five ERβ isoforms (ERβ 1-5), ΕRβ1 corresponding to the previously described ERβ-530.10 These isoforms are co-expressed with the wild type receptors in various tissues and pertinent data indicate that they are biologically active. Their role in estrogen-dependent diseases is thus under investigation.1
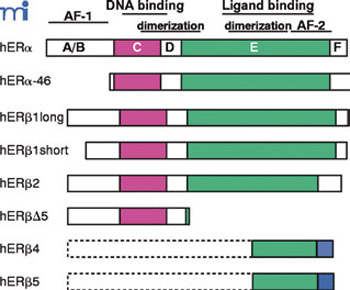
Figure 1. Schematic representation of human estrogen receptor α and β and their respective isoforms (Matthews J, Gustafsson JA, 2003 Molecular Interventions 3:281-292), used by kind permission.
Mechanisms of estrogen action
Estrogens exert their effects on estrogen responsive cells via activation of estrogen receptor (ER) and, directly or indirectly, regulation of transcription of target genes involved in the cell function. There follows a brief description of the mechanisms of estrogen action, as reviewed by Nilsson et al (2001).9 The term ER hereinafter refers to both ERα and ERβ subtypes.
Direct action of estrogen receptors on the genome
Endogenous estrogens, synthetic estrogen analogs or phytoestrogens enter the cells, where they bind to the estrogen receptor (Figure2). The conformation of the bound receptor depends strongly on the nature of the ligand. Binding results in the release of the receptor associated proteins, such as hsp90, dimerization of the receptor (ERα and ERβ may be present as homodimers or heterodimers), rapid nuclear localization of the activated ER-estrogen complex and binding of the dimer to its specific DNA sequences (EREs). The DNA bound complex interacts with other coactivator proteins such as p160 and the proteins of the CBP/P300 family, as well as other transcription factors, resulting in the remodeling of chromatin and initiation of transcription by the basal transcriptional machinery and RNA polymerase.11,12 The result is either induction or repression of genes and increase or decrease in protein synthesis, respectively. The chromatin decompaction is a key step required for the initiation of transcriptional activation and requires histone acetyltransferases (HATs).13 Many coactivator proteins required for ER activity are HATs (p160 family, p300, CBD).14 Another group of enzymes, known as histone deacetylases (HDACs), deacetylate histones to promote ER-mediated transcriptional repression.14,15

Figure 2. The genomic classical (via EREs) and non-classical (via AP-1 and viaNF-kB response elements) actions of the estrogen receptor (Nilsson et al, 2001. Physiol Rev 81: 1535-1565), used with kind permission.
An alternative pathway of ERα and ERβ action has been reported by which the receptors influence gene transcription, not through the ERE-directed classical pathway, but independently of ER binding to its consensus response elements. This is accomplished by interaction of the receptors with the transcription factor AP-1 (c-fos/c-jun dimers) by way of protein-protein interactions, thus modulating the binding of AP-1 to AP-1 sites on the DNA9,16 (Figure 2). Current research indicates that binding of SERMS to ERα transactivates the transcription through binding to EREs, whereas binding of SERMs to ERβ stimulates the AP-1 pathway, demonstrating that ERα and ERβ respond differently to certain ligands at an AP-1 element.17 Of note, some antiestrogens, such as tamoxifen, are strong agonists of AP-1 dependent transcription in certain tissues, whereas in other tissues they act as antiestrogen via the classical EREs. The estrogenic effects of tamoxifen resulting in tissue growth and differentiation, as for example in uterus and endometrium, may be attributed to the AP-1 mediated pathway, a major route via which estrogens promote growth and differentiation effects.18
Estrogen receptor interacts with another important transcription factor, nuclear factor-kB (NF-kB), resulting in suppression of genes, such as the interleukin-6 gene, or activation of genes, such as the serotonin receptor 1A gene16,19,20 (Figure 2). This suggests that estrogen receptor regulates the NF-kB mediated gene expression in a cell type-specific manner. Interleukin-6, a cytokine transcriptionally up-regulated by NF-kB, regulates bone metabolism and endothelial cell function, therefore its estrogen-mediated repression may have important implications in osteoporosis, inflammatory processes and atherosclerosis.20-22 Evidently, the interaction of ER with the transcription factor NF-kB as well as with the AP-1 is subtype and tissue specific and is considered to be involved in the inhibitory effects of estrogens on proinflammatory cytokine activity.21
In conclusion, the two ER subtypes exhibit distinct interactions with common or specific cofactors, bind in a variable manner to a variety of ligands and may act via different response elements exhibiting altered specificity and affinity. These characteristics and the differential expression of ERα and ERβ may be responsible for tissue-specific responses to estrogenic compounds.9,23
Cross talk between estrogen and other signaling pathways; Estrogen-independent activation of ER
Phosphorylation is one of the most frequent post-translational modifications of proteins on serine and threonine residues and less on tyrosines. Phosphorylation is regulated by cellular enzymes (kinases and phosphatases), which transfer a phosphate group from ATP onto target proteins or remove it correspondingly. Phosphorylation of the estrogen receptors is influenced by the cognate ligand and it affects receptor functions positively or negatively, such as transcriptional activity, stability and nucleocytoplasmic shuttling.24
Available data indicate that in several cell types extracellular signals such as insulin, IGF-I, EGF and TGF-β, via the mitogen activated protein kinases (MAPK), phosphorylate the AF-1 region of ERα and ERβ resulting in transactivation of the receptors in the absence of the ligand.25 Studies have shown that ER is phosphorylated by growth factors resulting in the initiation of ERE-mediated gene expression.26,27 For full activity of ERα AF-1, the phosphorylation οf Ser-118 is very important, where as for ERβ this effect is mediated via Ser-124. Phosphorylation may also take place at tyrosine residues of ERα, especially at Tyr-537 which regulates the ligand binding step.9
ERα and ERβ are also activated by neurotransmitters (dopamine) as well as by regulators of the general cellular phosphorylation state such as protein kinase A (PKA) and protein kinase C (PKC), which also phosphorylate ERs.28,29 The cyclins, cyclin A and D1, which are regulatory subunits of cyclin-dependent kinases (CDKs), have also been shown to be ER activators.30-32 The presence of factors other than the classical hormone, activating ERs, accompanied by an increased receptor phosphorylation, may modulate the activity of ERs in response to diverse physiological signals.33,34 The estrogen-independent activation of estrogen receptors may be important when the concentration of growth factors is locally increased or when estrogens are too low.
Membrane and mitochondrial estrogen receptors
Accumulated evidence strongly supports the presence and importance of plasma membrane estrogen receptors in a variety of cells. The membrane ER is possibly similar to the nuclear receptor, which has translocated to a particular location in the membrane, known as caveolae, allowing its cross talk with other signaling molecules.
The binding of 17β-estradiol (E2) to cell surface proteins triggers downstream signaling cascades resulting in calcium flux, adenylate cyclase activation and cAMP generation, phospholipase C activation and IP3 generation. Given that these signals are emanating from the activation of G protein related pathways and because G proteins and ER exist in caveolae, it is likely that an interaction may take place within this membrane domain. Alternatively, E2 via ER may activate another G-protein coupled receptor (GPCR) in the membrane, thus affecting G protein activation.35,36
Additional signal transduction pathways, being rapidly responsive to E2 and originating from the membrane, involve the stimulation/inhibition (being dependent on the ERα or ERβ subtype) of phosphoinositol-3hydroxy kinase (PI3K) and the familyof MAP kinases, such as extracellular-regulated kinase (ERK), p38β isoform, as well as the c-Jun N-terminal kinase (JNK).37 Of note, the signaling from the membrane can also extend to other intracellular transcription factors such as AP-1 or NF-kB, resulting in gene repression or activation. In conclusion, signals emanating from the membrane are transduced to the nuclear compartment and regulate important functions, such as cell growth and survival, migration, angiogenesis and apoptosis.1,35,36
Finally, current data documenting the presence of ER subtypes in the mitochondria of animal and human cells implicate a role of ER signaling in energy production, thus further emphasizing the complexity of ER-signaling.38,39
ERα and ERβ expression
ERa and ERβ are co-expressed in many tissues. ERα is primarily expressed in the uterus, liver, heart, kidney, whereas ERβ is primarily expressed in the ovary, prostate, lung, gastrointestinal tract, bladder, central nervous system and hematopoietic system.1,9
Characterization of mice lacking either ERα οr ERβ οr both (αERKO, βERKO and αβERKO) has yielded valuable information about the unique role of each subtype in estrogen action in vivo.40 Briefly, results showed that ERα is required for normal mammary gland development and mediates estrogen-stimulated increase in bone formation, while ERβ’s role in bone is less clear. ERα is required for protection of vascular and endothelial cells against injury as well as for cardioprotection in a myocardial ischemia model. ERα appears to play a role in adipocyte growth and proliferation, the ERβ also possibly involved in adipogenesis.1 ERα and ERβ are expressed in various brain areas and ER-mediated effects are considered to provide neuroprotection. In a mouse model of brain injury, estradiol treatment protects wild-type and ERβ-null mice from brain injury, while this protection is abolished in ERα-null mice, thus suggesting an important role for ERα.41 Ηοwever, recent data indicate that ERβ-mediates decreased microglia activation, suggesting a role of ERβ in neuroprotection.42 On the other hand, ERα or ERβ selective agonists have been shown to protect hippocampal neurons against glutamate-induced cell death and enhanced bcl-2 expression in hippocampal neurons, suggesting that activation of either ERα or ERβ can promote neuroprotection.43 Taken together, as ERβ is highly expressed in the brain compared to the reproductive organs in females, ERβ-selective agonists may be of benefit and protect, at least in part, from some neurodegeneration leading pathways.44
Accumulating evidence supports the keyrole ofER subtypes in some cancers, the ERβ expression being significantly reduced in breast, prostate and colon cancer.1In ovarian cancer, ERα is expressed in tumors of both epithelial and stromal origin, whereas ERβ is abundantly expressed predominantly in granulosa cell-derived tumors and to a lesser extent in epithelial origin tumors.45 Malignant ovarian tumors originating from epithelial surface constitute about 90% of ovarian cancers and express low levels of ERβ compared to normal tissues. Recent data point out that restoration of ERβ in ovarian cancer cells leads to inhibition of proliferation and enhancement of apoptosis, thus suggesting that ERβ may play a possible tumor-suppressor role in ovarian carcinogenesis.46 Taken together, several lines of evidence indicate that ERβ exerts an inhibitory action on ERα-mediated gene expression and in many instances opposes the actions of ERα.23
Since many phytoestrogens bind to ERβ with higher affinity than ERα and because their potential to transactivate ERβ is higher than ERα, phytoestrogens may be of benefit in tissues in which ERβ-mediating signaling plays a significant role.44
PHYTOESTROGENS
Structure/Classification
The main classes of phytoestrogens are the isoflavones (genistein, daidzein, glycitein, equol and biochanin A), the lignans (enterolactone, enterodiol), the coumestanes (coumestrol), the flavonoids (quercetin, kaempferol), the stilbenes (resveratrol) and the mycotoxins (zearalenol)47 (Figure 3). They are either of dietary plant origin or ingested as precursors and then metabolized in vivo by the microflora of the human gut. Indeed, equol is produced from daidzein by the intestinal microflora, while the mammalian lignans, enterodiol and enterolactone, are formed from plantlignan glycoside precursors by the activity of the gut microflora in the proximal colon.48,49 Of note, in some studies myco-estrogens are not included in the group of phytoestrogens because they are not fungal-derived but plant devired estrogens.49 The aforementioned compounds are all polyphenols and structurally resemble the natural estrogens.
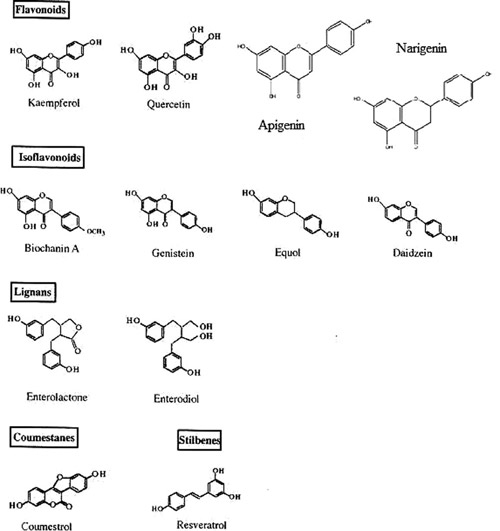
Figure 3. The chemical structure of the so far known phytoestrogens.
Polyphenols are present in all plants, while there are >8000 phenolic structures identified in nature which may be categorized in more than 10 classes. These may be simple molecules (such as phenolic acids) or high lypolymerized (i. e. tannins). Flavonoids, the most common polyphenols in the plant kingdom, contain more than 5000 compounds subdivided into six major classes such as flavones (e.g. apigenin, luteolin), flavonols (e.g. quercetin, kaempherol), flavanones (e.g. naringenin, hesperidin), flavanols (e.g. catechins, epicatechin, gallocatechin), anthocyanidins (e.g. cyanidin) and isoflavones (e.g. genistein, daidzein).50
Flavonoids do not exhibit an estrogenic effect; however, the isoflavones (genistein, daidzein) and some flavones, flavanones and flavonols (apigenin, kaempferol and naringenin) activate estrogen-receptor-mediated signaling.
Absorption and Bioavailability
Exploration of the bioavailability of isoflavones (the most common form of phytoestrogens), particularly genistein and daidzein, revealed that it depends on the gut microflora activity. Data show that, despite the considerable degradation of isoflavones in the gut, their plasma concentrations are significant. Isoflavones are mainly present as inactive glycosides and, after removal of the sugar residue in the gut by bacteria (beta-glycosidases), they become active compounds, taken up by enterocytes and entering the peripheral circulation.51 Following absorption, they are reconjugated in the liver, mainly to glucuronic acid and to a lesser degree to sulphuric acid.52,53 There is a large variability in genistein and daidzein metabolism in humans, which has been attributed to variable individual intestinal flora and transit time. In the Japanese, genistein has been found in micromolar concentrations, but in western populations, levels are much lower.49 Of note, Setchel and Cassidy (1999) have shown that in an adult, consumption of 50 mg/day of isoflavones may give rise to plasma concentrations ranging from 200-3000 nM.54 Several other studies assessing plasma concentrations of phytoestrogens have demonstrated that the plasma levels in humans range from 10nm-10μM.55 With regard to lignans, they are metabolized to enterolactone and enterodiol in the gut. In the Finnish population, consumption of wholegrain bread (200g/day) has resulted in variable plasma concentrations of enterolactone (15-41.4 nmol/l).49
A recent study assessing the effect of an 8-week dietary soy intervention on urinary isoflavone excretion among young girls (8-14 years old) revealed a very high urinary isoflavone excretion as compared to adult women. Such data suggest less intestinal degradation and/or greater absorption of isoflavones in young people, emphasizing that further investigations into the pharmacokinetics and age at consumption of isoflavones are needed.56
Soy infant formulas are widely used and may result in a daily exposure of infants to isoflavones (genistein, daidzein and equol) that is 6- to 11-fold higher on a body weight basis from the dose that has hormonal effects in adults consuming soy foods. The mean plasma concentration of total genistein in infants fed soy-based formula has been reported to be 2,500 pmol/ml (684 ng/ml) as compared to 276 pmol total genistein/ml blood concentration found in Asians on a traditional diet that is high in soy content.57,58 It has been reported that the circulating concentration of isoflavones in infants fed soy-based formulas were 13,000-22,000 times higher than plasma estradiol concentrations in early life and may exert biological effects, whereas the contribution of isoflavones from breast milk and cow milk was negligible.57 In addition, it has been shown that infants fed soy-based formula maintain a high steady state plasma concentration of isoflavones, which may possibly be explained by reduced intestinal biotransformation, as evidenced by low or undetectable concentrations of equol and other metabolites.59
In a recent report, isoflavones in breastfed infants, whose mothers consumed soy for 2 to 4 days, were significantly increased in the infants urine (from 29.8 to 111.6nmol/mg creatinine) and in breast milk increased from 5.1 to 70.7nmol/l.60 Of note, in some countries such as Israel, the soy-based formula is widespread, with rates far beyond clinical indications (in case of suspicion of cow’s milk allergy).61 Since there is a broad spectrum of infant foods becoming available, there may be a high exposure to dietary isoflavones during the first year of life. Due to the lack of definitive evidence of hormonal activities of isoflavones at doses relevant to the soy-fed infants, further studies in experimental animals and human populations exposed to isoflavone-containing products and in particular soy-based infant formulas are necessary.62
Flavonoids are mainly found conjugated with sugars (glycosides) in plants and occasionally as aglycones. Evidence supports that the aglycone form of flavonoids as well as their glycosides show a considerable degree of intestinal absorption, ranging from 10-60%, depending on the type and the dietary source of the flavonoid. For example, the absorption of querquetin from tea has been shown to be half that of onions. Given that querquetin in onions is found mainly as a glycoside moiety, it has been suggested that the glycosidic form of flavonoids may enhance absorption.63 Current evidence indicates that many of the metabolites of phytoestrogens and flavonoids formed, either in the gut or in liver, may be also biologically active and may mediate estrogen-signaling.64,65 Taken together, the bioavailability of dietary substances determines their activity in vivo. However, the relevance of in vitro studies to the in vivo situation should be considered with caution. Although metabolism in peripheral tissues also takes place, most of the conversion seems to occur at the level of the gut and liver, therefore the target tissue levels may be analogous to those in plasma.
Intake
Several studies have investigated the dietary intake of polyphenols and phytoestrogens; results, however, are contradictory. Numerous factors, such as light, environmental conditions, plant genetics, ripeness and species variety, affect the formation and the content of flavonoids and phytoestrogens in plants. Moreover, different methods assessing the content of polyphenols in plants have given variable results.63 The most widely studied phytoestrogens are the isoflavones genistein and daidzein from soy food (mainly consumed in the Orient), whereas lignans are mainly consumed by Europeans, since foods such as cereal brans, legumes and vegetables (known to be rich in lignans) play important role in the European diet. Soya beans contain 560-3810mg/kg isoflavones, depending on variety and growing conditions. Soya milk, beans prouts etc., contain 13-2030 mg isoflavones/ kg, depending on the starting raw material and final water content. Soy foods, however, are now widely available to European consumers and soya-based European foods contain 38-3000mg isoflavones/kg, depending on the source of soya and dilution with other ingredients.66 In the literature, various reports point out that the estimated daily intake of flavonoids differs among countries, ranging from 20-100mg/day.63 Dietary assessment of phytoestrogens gives estimates varying between an intake ranging from 50mg to 200 mg of phytoestrogen/day in a traditional Japanese diet.67,68 In Asian countries, an average daily dietary intake of soy and isoflavones is estimated to be 50g and 20-150mg, respectively, while in western countries the intake is lower, i.e. 1g daily and 2mg daily, respectively.69 It must be underlined that women consuming the commercially available phytoestrogen preparations are taking doses ranging from 50-150mg aglycones/day.70
In view of the above, it becomes evident that when screening phytochemicals to assess their possible estrogenic activity, the levels of the compounds tested must be at a range that is thought to be achievable in humans in vivo. In this regard, many factors should be taken into account, such as the daily intake of the phytochemical, the rates of metabolism and clearance as well as the plasma levels, factors which are known to vary considerably between individuals.
Phytoestrogens are modulators of ERα and ERβ-mediated activity
A large body of evidence demonstrates various features of phytoestrogens: estrogen-like action, estrogen receptor binding, ER-transactivation, estrogen-dependent target genet expression or cellular growth effects. Different studies, however, provide variable results regarding the estrogen-like potencies of the phytoestrogens. This may be due to the fact that the potencies of several phytoestrogens have been tested in various cell systems using different techniques (i.e. radioligand binding assays, transactivation assays, target gene expression) under different conditions (such as different dosages, in presence or in absence of estradiol), so that results are not comparable.
Given the conflicting results in the literature, Mueller et al (2004) employed a human endometrial Ishikawa cell line that stably expresses human ERα or ERβ to determine the potencies of the best known phytoestrogens in one comparable system for both ERα and ERβ activity.64 They found that the soy-derived genistein, coumestrol and equol displayed a preference for transactivation of ERβ-ERE responses compared to ERα-ERE responses and were 10-to 100-fold less potent than diethylstilbestrol. Resveratrol, enterolactone and its human metabolite 6-OH enterolactone and human metabolites of daidzein were weak agonists to both ERα and ERβ. Interestingly, they showed that phytoestrogens affect the transcriptional activity of ERα and ERβ in an ERE sequence-dependent manner,64,71 thus implying a degree of promoter dependency. Their ligand binding measurements revealed that genistein, coumestrol and equol had high binding affinity to ERα and ERβ and with a distinct preference for ERβ.64 Quercetin, a common phytoestrogen, has been shown to induce ERE-dependent transactivation in MCF-7 cells through both ERα and ERβ but with a higher capacity, like genistein, to stimulate ERβ responses as compared to the stimulation ERα responses.72 It must be stressed that Kostelac et al (2003) revealed that the isoflavones genistein and daidzein, its metabolite equol, and coumestrol were able to modulate the binding of both ERα and ERβ to estrogen-response elements (EREs).73 Of note, genistein and daidzein preferentially activated the binding of ERβ to ERE, whereas coumestrol and equol showed only a slight preference in the binding of ERβ to ERE compared to the binding of ERα to ERE. Such data lead to the conclusion that phytoestrogens differ not only in their ERE-transactivation potencies and their ER-binding affinities, but also in their ability to increase the ER binding onto DNA-response elements (EREs).
On the other hand, Harris et al (2005), in MCF-7 cells transfected with ERα or ERβ, evaluated the dose-dependent responses of various phytoestrogens in the absence and presence of 17β-estradiol.55 Their results revealed that genistein, daidzein, apigenin and coumestrol had a differential and robust transactivation of ERα- and ERβ-induced transcription, with up to 100-fold stronger activation of ERβ. Equol, naringenin and kaempferol were weaker agonists. In the presence of 0.5 nM 17β-estradiol, the addition of genistein, daidzein and resveratrol superstimulated the system, while kaempferol and quercetin acted as antagonists at the highest doses. In the same line, Muller et al (2004) have shown that in the presence of diethylstilbestrol, resveratrol displayed a biphasic activity: at low doses it increased the DES-induced activity of ERα and ERβ, but at high doses it inhibited the activity of ERα and ERβ.64
It is of interest that the ability of phytoestrogens to modulate cellular proliferation is also dose-dependent and estrogenic status-dependent. Resveratrol, genisteina and quercetin have shown biphasic modulation with regard to proliferation of ER positive breast cancer cell lines. They stimulate proliferation at low concentration (physiologically relevant), the ER possibly involved in adverse cell proliferative effects.74-76 However, they inhibit proliferation at concentrations higher than 50-60μΜ, the inhibitory cellular growth effects considered to be exerted by way of ER-independent pathways. Of note, Shao et al (2000) showed that genistein’s anti-proliferative effects are more pronounced in the presence of estradiol, implying that the “good estrogen” action of genistein is relevant to chemoprotection.77
Animal studies have also shown that phytoestrogens exert variable effects depending on the absence or presence of endogenous estradiol. For example, a group of immature, ovariectomized Sprague-Dawley rats in gesting a commercially available, closed-formula diet containing “high amounts” of genistein and daidzein (21mg genistein and 14mg daidzein per 100g of rat feed) failed to exhibit a uterotrophic response to administered estradiol. This finding suggests that the response was already stimulated to nearly maximal levels by phytoestrogens before the animals were treated with estradiol.78 However, in another study involving immature, intact Sprague-Dawley females exposed to higher doses of genistein (37.5 or 75mg per 100g body weight of a modified AIN-76 diet), no detectable increase in uterine weight was observed when compared to controls fed the same diet.79 This result implies that, at least with respect to this assay, intact females are much less responsive than ovariectomized females to dietary phytoestrogens. Such findings emphasize the importance of variable dosages and the impact of estrogenic status in the overall estrogenic effect of phytoestrogens.
Phytoestrogens, similar to estrogens, have been shown to act via several membrane-initiated signaling mechanisms in cell lines expressing the membrane version of ERα (mERα), resulting in the activation of ERK kinases.80-82 Phytoestrogens have been shown not only to exert direct effects on ER activity but also to affect the formation of endogenous 17β-estradiol, thus regulating indirectly estrogen-signaling. Indeed, some inhibit aromatase, an enzyme that catalyzes the conversion of testosterone to estradiol, thus protecting against breast cancer via ER-independent mechanisms.83,84 Metabolites of genistein and daidzein isolated from human urine (i.e. glucuronides, sulfates) have also demonstrated interactions with estrogen receptor α and β, indicating that metabolism of isoflavones not only by enteric bacteria but also by hepatic enzymes may affect estrogenic activity.65
In conclusion, the so far known phytoestrogens and some of their human metabolites, produced either by enteric bacteria or hepatic enzymes, are selective estrogen receptor modulators, exhibiting variable potencies towards ERα-or ERβ-mediated responses, the ERβ-mediated activity preferentially induced. Finally, most phytoestrogens modulate many other signalling cascades in various cell types involving MAP kinase and NF-kapa B signalling pathways, AP-1-mediated signalling, cell cycle regulation, apoptosis and other nuclear receptor-mediated signalling.85-89
In view of the above data, care should be taken when defining the estrogenic/antiestrogenic of a compound. The intrinsic estrogenic status and the dose should be considered, especially in the context of using a compound to prevent symptoms associated with estrogen deficiency during menopause or to prevent hyperestrogenic effects in breast and endometrial cancer. Evidently, it is essential to assess the phytoestrogens’ effects at multiple levels, in vitro and in vivo, in order to obtain a full picture which may be relevant to different physiological or pathological states in humans (i.e. menopause, premenopause, breast cancer).
NOVEL ESTROGEN-LIKE COMPOUNDS OF PLANT ORIGIN
Apart from the known phytoestrogens, accumulating evidence attests to the fact that there are many more phytoestrogens in nature. A detailed presentation of the recently identified novel phytoestrogens in nature is given below. Their chemical structures are shown in Figure 4.
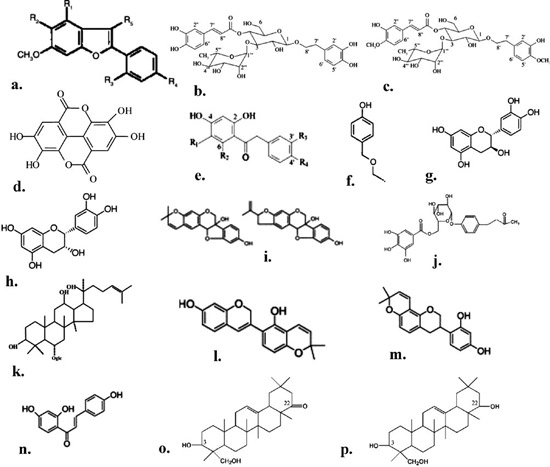
Figure 4. The chemical structure of some novel phytoestrogens. a. arylbenzofurans, b. acteoside, c. martynoside, d. ellagic acid, e. deoxybenzoines, f. ethoxymethylphenol, g. catechin, h. epicatechin, i. glyecollins, j. lindleyin, k. gRhi, l. glabrene, m. glabridin, n. isoliquitireginin chalone, o. soyasapogenol A, p. soyasapogenol B.
Halabalaki et al (2000) isolated three novel arylo-benzofurans, namely ebenfuran I, ebenfuran II and ebenfuran III, from the plant Onobrychis ebenoides. All three compounds showed high binding affinity for the native ER.90 Further investigation by Papoutsi et al (2004) revealed that ebenfuran II, at low concentrations (10-6-10-9M), demonstrated a selective estrogen receptor modulator profile, being antiestrogenic in MCF-7breast cancer cells and estrogenic inosteoblasts (KS483 cell line), without any stimulatory activity in uterine cells, implying that it may be considered for the prevention and treatment of breast cancer and osteoporosis.91 Acteoside and martynoside, which are plant phenylpropanoid glycosides, have been shown to antagonize both ERα and ERβ-mediated signaling.92 Papoutsi et al (2006), by using the appropriate endpoint assays, demonstrated that martynoside was anti-estrogenic in MCF-7 cells, induced mineralization in osteoblasts and inhibited endometrial cell proliferation, thus implying its potential as natural SERM.92
Ellagic acid, a dietary polyplenol, present in abundance in strawberries, has shown significant antiestrogenic activity in MCF-7 breast cancer cells at low concentration (10-9M-10-7M) and estrogenic activity in osteoblasts (induced mineralization), without any effects on endometrium-derived epithelial cells. In this regard, ellagic acid may be considered as natural SERM.93
Deoxybenzoins, intermediates in the biosynthesis of isoflavones, are found in several plants and marine sources, such as Glycyrrhiza sp., Trifolium subterraneum and Ononis spinosa and have structural similarities to isoflavones. Fokialakis et al (2004) showed that deoxybenzoins constitute a new class of phytoestrogens exhibiting affinities for ERα and ERβ, grossly comparable to those of daidzein, while some demonstrated considerable selectivity and transcriptional bias towards ERβ. Their activity, in stimulating the proliferation of ER-positive breast cancer cells and regulating the expression of endogenous and stably transfected reporter genes, differed considerably, with some inhibiting cell proliferation, while effectively inducing gene expression at the same time.94
4-Ethoxymethylphenol (4ΕΜ) is another novel phytoestrogen, found in Maclura pomifera that acts as an agonist of ERα and ERβ in MCF-7 breast cancer cells and in Hela cells. The presence of coactivators SRC-1, CBP and E6-AP, enhanced, like estrogen, the 4EM-mediated transcription of ERα. The activity of 4EM was specific for ER and did not activate the transcription of the progesterone receptor.95
Tea catechins, which are flavonoids, include catechin, epicatechin, epicatechin gallate, epigallocatechin and the epigallocatechin gallate. Experimental evidence indicates that in HeLa cells, transiently transfected with an ERE-regulated luciferase reporter and an ERα or ERβ expression vector, tea catechins alone did not induce luc activity in either of ERs. However, high concentrations of ECG or EGCG (5X10-6M) reduced the E2-induced estrogen effects, whereas lower concentrations increased significantly the E2-induced luc activity via ERα and ERβ. In view of the lower doses found in human plasma after tea drinkingalongwiththeirestrogeniceffects,theroleof usual tea drinking might be helpful for the prevention of estrogen deficient diseases, such as menopausal disorders and osteoporosis.96
Phytoalexins constitute a chemically heterogeneous group of antimicrobial substances. They are synthesized de novo, accumulate in plants as a stress response and are products of a plant’s secondary metabolism. The glyceollins represent a group of phytoalexins whose biosynthesis is increased in response to stress signals. Glyceollin accumulates in high concentrations in soybeans under stress conditions. The glyceollin isomers I-III are derived from the precursor daidzein in the glyceollin pathway. The glyceollin can represent as much as 56% of the total isoflavone composition in soybean. In vitro data show that glyceollins display only slight estrogenic activity, but they cause a dose-dependent suppression of E2-induced transactivation and MCF-7 proliferation. Glyceollins suppressed estrogen activity through both ERα and ERβ in transiently transfected HEK 293 cells. Such data imply that glyceollins, being unique antagonists on ER in both HEK 293 and MCF-7 cells, may represent an important component of the health effect of soybased foods.97
The phytochemical lindleyin isolated from Rhei rhizoma (rhubarb), a herbal medicine extract traditionally used in Chinese medicine, acts as an antagonist (10-5M) equally to both ERα and ERβ, being a novel phytoestrogen. However, it had no effect on TRα or PPARγ, suggesting that the ER interactions are specific. lindleyin has a phenolic backbone and is a glycoside obtained from Aeonium lindrleyi, a crassulacea endemic to the Canary Islands.98
Ginseng is a popular herbal medicine that has been used for over 2000 years in oriental countries. A component of ginseng, saponin, known as gingenoside (gingenoside-Rh1), has been shown to activate the transcription of the ERE-responsive lucreporter gene in MCF-7 breast cancer cells and CV-1 cells at high concentration (50 μΜ), implying that this compound is a weak phytoestrogen, presumably by binding and activating the estrogen receptor.99
Consumption of Glycyrrhiza glabra, the licorice plant,canbetracedback6000years.There is evidence that compounds isolated from the licorice root exhibit varying degrees of ER action in different estrogen responsive tissues. Briefly, results indicated that glabrene, isoliquiritigenin (IlC) and glabridin bind ER. The stimulatory effects of glabrene in vivo were tissue specific and similar to those of estradiol. The effects of concomitantly increasing concentrations of glabrene and IlC on the growth of breast tumor cells were variable, promoting ER-dependent growth at low concentrations (10nM-10μΜ) and exerting ER independent antiproliferative activity at concentrations >15 μΜ.100,101
Saponins are, inter alia, important components of soy. Recent evidence has shown that a soy sapogenol has the potential to induce estrogen receptor mediated signaling.102
BIOLOGICAL EFFECTS
Epidemiological, clinical and animal studies as well as studies in vitro have provided data on the effect of phytoestrogens on human health and disease. Accumulating evidence, based on epidemiological and animal studies, has indicated that phytoestrogens intake may have protective effects against various cancers. However, controversial results have also been reported.49,103 Phytoestrogens, originating from various plant sources, have shown variable effects on different types of cancer. It is noteworthy that many of the epidemiological and experimental studies have focused on isoflavones and soy products. It is only recently that interest is increasing in the protective effects of other classes of phytoestrogens, such as lignans.49
It has been shown that in adult women the consumption of phytoestrogen-rich foods such as soy, a source of isoflavones, is not or only slightly protective against breast cancer. No negative effects of soy on breast cancer have been reported, while it may be beneficial if consumed before puberty or during adolescence or at very high doses.49,104,105 Such findings are in agreement with conclusions derived from studies of immigrants and other epidemiological studies.49,106 On the other hand, a diet based on wholegrain products, low in lignans, resulting in low plasma enterolactone levels, has been associated with increased breast cancer risk. A high plasma enterolactone (30-70 nmol/l) is probably protective against breast cancer, at least in Nordic countries with high wholegrain intake.49,107 In a recent prospective study, high genistein circulating levels were associated with reduced breast cancer risk in the Dutch population, whereas no effects of lignans on breast cancer risk were observed.108 It is important to mention that animal and cell culture studies have demonstrated that certain phytoestrogens like genistein and resveratrol not only reduce but also stimulate estrogen-dependent growth such as uterine growth and breast cancer growth, depending on dose and timing of exposure.49,76,109-113 Several other potential adverse health effects have also been reported.114,115 However, several other lines of evidence indicate that genistein and resveratrol can protect against breast cancer in rat by regulating important mammary growth and differentiation pathways, the timing of exposure being critical for the mammary protective effects of genistein.116 Taken together, the role of phytoestrogens in breast cancer is controversial and their overall clinical efficacy is questionable, while negative effects on the breast cannot be completely excluded.
Prostate cancer, one of the main causes of death among men in western societies, has been studied intensively in relation to phytoestrogen intake. Epidemiological, animal and cell culture studies have demonstrated that dietary phytoestrogens, mainly soy and soy-derived isoflavones, may play a protective role against prostate cancer.49 In a recent prospective study in 43,509 Japanese men (age 45 to 74 years), who generally have a high intake of isoflavones, intakes of genistein, daidzein and soy food decreased risk of localized prostate cancer, whereas positive associations were seen between isoflavones and advanced prostate cancer.117 Well-designed trials in human subjects, however, assessing the role of lignans in prostate cancer are not available, thus any definite conclusions for this class of phytoestrogens can not be drawn.49,118
Data on the effect of soy and isoflavones on colon cancer, based on experimental rat models, are conflicting, showing either no effect or even a stimulatory one. Ryebran or isolated lignans seem to protect against colon cancer and the formation of polyps.49 The positive findings from animals, however, can-not be extrapolated to humans, thus, well-designed experimental studies are needed to define the impact of phytoestrogens on human colon cancer.
Several animal and in vitro studies indicate that phytoestrogens, mainly daidzein and genistein, prevent bone loss. Human studies, however, which are of short duration (6 months) and include a small number of subjects, haved emonstrated that phytoestrogens exert only moderately beneficial effects on bone.67,68 Based on data from the EU-funded project phytohealth, there is a suggestion, but no conclusive evidence, that phytoestrogens, primarily genistein and daidzein, given as soyabean-protein isolates, whole-soyabean foods or extracts, supplements or pure compounds, have a beneficial effect on bone health.119 However, it is considered that long-lasting, well-designed clinical trials are needed to prove a specific role of phytoestrogens in the prevention of osteoporosis.
The phytoestrogen intake and the incidence of hot flushes and vaginal dryness in postmenopausal women has been studied extensively, yielding either favorable effects or no effect at all.112,119,120 Animal studies have led to the conclusion that isoflavones (in particular their metabolic product equol) lead to infertility (mainly in females) and endometrial hyperplasia in cattle, sheep and the guineapig. Despite the fact that phytoestrogens are implicated in adverse effects upon fertility in various animals, there are no similar reports on humans consuming large amounts of these compounds (such as Asian women). In humans, intake of phytoestrogens has been reported to interfere with the regulation of the menstrual cycle in premenopausal women (increased length of the menstrual cycle and reduced lH, FSH and progesterone levels).47
There is growing evidence linking the intake of soy-based foods and genistein with a reduction of coronary heart disease. Many of the studies have shown that whole-soyabean foods exert favorable effects on cardiovascular disease, especially on serum lipoprotein profile, while isolated isoflavones seem to improve only the endothelial function.67,70,119 The cardioprotective effect of the individual phytoestrogens, however, has yet to be substantiated by long-term controlled trials.121 Finally, in cell cultures it has been shown that genistein may exert neuroprotective functions or induce neurotoxicity.122-124 Recent in vivo findings strengthen the idea that soy induces neurodegeneration.125 Epidemiological data demonstrate a positive correlation between tofu consumption and brain atrophy.126 It is concluded that further studies are needed to delineate the effects of phytoestrogen on cognitive functions.
The variation between studies and the conflicting results obtained may be due to multiple factors, such as the soy product used or the type of the individual phytoestrogen used, their plant origin, the dose delivered, the time and duration of exposure, the metabolism, the route of administration, the bioavailability, as well as the intrinsic estrogenic status. Given that differences in the above parameters may result in variable and in many instances divergent effects, it becomes evident that, at present, definite conclusions on the health effects of individual phytoestrogens cannot be drawn. The studies conducted so far are limited to soya isoflavones and soya products. Further systematic research is needed to evaluate the clinical effects of each individual phytoestrogen at the physiological and pharmacological levels, taking also into account whether these substances would be delivered in the context of food or as dietary supplement or even as pharmaceutical agents.127,128
At present, although there are no approved health claims for phytoestrogens, many preparations in the form of plant extracts or mixtures, containing varying amounts of isolated phytoestrogens, are commercially available on the market in numerous forms as dietary supplements and as health food products.70,129 Given that these products are not under any regulatory controls, their composition is not consistent, their components vary greatly and, since there have not been systematic dose-ranging studies, no dietary recommendations for each of these products can be made; therefore, their safety and clinical effectiveness are questionable. Evidently, the use of the so-called health-promoting phytoestrogen-enriched products should be viewed with caution.
TEST SYSTEMS ASSESSING PHYTOESTROGENS’ ESTROGENIC ACTIVITY
In vitro test systems
The estrogenic/antiestrogenic potential of phytoestrogens is usually assessed by an array of in vitro test systems as described below:
1) Radiometric competitive receptor binding assay.
Given that the first step in the ER transduction cascadeisbindingofaligandtoER,themeasurement of ligand binding is an important assay characterizing the potential of a test compound to interact with the ER. The ‘classic’ ligand binding assay uses radioactively labeled estradiol, which competes with the test compound for ER binding sites. This competitive binding assay provides relative binding affinities of test compounds to ERα or ERβ compared to estrogens, which is usually unlabeled estradiol or diethylstilbestrol (DES). The use of a fluorescein labeled estradiol instead of radioactive label made it possible to use fluorescence polarization to measure binding affinities. Data indicate that relative binding affinity rankings of various compounds are identical in both assays. The advantage of the radioactive binding assay is that both quantification of ER binding sites of any tissue or cell sample expressing ER can be obtained, and binding affinities of compounds may also be determined. In contrast, fluorescence polarization requires the use of purified ER due to the high background fluorescence in crude extracts. The advantage of fluorescence polarization is that it is a high-throughput assay which can screen many compounds for ER binding affinity within a day or less.
2) The luciferase reporter gene assay using cells lacking endogenous ER.
The binding of the liganded ER dimer to the promoter region of its target gene on DNA specific sites, i.e. the estrogen response element (ERE) is a crucial step in the ER signaling pathway. Transient transactivationassaysorrecombinantcellassays,inwhichcells are cotransfected with the cDNA for ERα, or ΕRβ and a reporter gene containing an ERE, are widely used to measure ligand-induced ER-mediated gene activation. In this assay, a mammalian cell line lacking endogenous ER is transfected with an expression plasmid carrying the cDNA of ERα or ERβ together with ERE linked to a luciferase reporter DNA. Addition of test compounds, candidates for estrogen-like activity, induces dose-dependent transcription of the reporter luciferase that can easily be monitored and quantified. It is a very sensitive assay whereby very weak to highly potent estrogens can be analyzed for their estrogenicity and antiestrogenicity.
3) The luciferase reporter gene assay using cells containing endogenous ER.
In the transient transactivation assays, exogenous receptor (ERα or ERβ) is forced into a cell accustomed to the lack of ER; therefore, some effects measured may not reflect the physiological response of the analyzed cell type. These assays measure the potential of a “candidate” estrogen to trigger ER signaling at the level of the ERα/ERE or ERβ/ERE pathway. To account for these limitations, cell lines with endogenous ER expression (like MCF-7) can be used to measure the potential of a “candidate” estrogen to transactivate the native ER/ERE pathway.
4) Cell lines with endogenous ER expression measuring the expression of endogenous ER-target genes (end point assays).
Cell lines with endogenous ER expression can also be used to measure the expression of endogenous ER target genes, i.e. the analysis of end-points markers, such as mRNA or protein expression. The measurement of endogenous ER target gene (or protein) expression represents a valuable physiological assay for tissue specific estrogenicity or antiestrogenicity. In this type of assay, all steps of the ER signaling transduction pathway are considered, i.e. ER ligand binding, ER expression, ER dimerization, nuclear translocation, nuclear binding, available co-activators, etc. It is important to mention that the right protein (or gene) marker has to be monitored dependent on the chosen cell line.
5) Proliferation assay using an established cell line that is known to respond to estrogens.
The proliferation assay assesses the growth promoting effects of a “candidate” estrogen, since estrogens are known stimulants of cellular growth, whereas antiestrogens arrest the growth of cells (dependingon the cell line). This assay systemme asures the potential of a “candidate” estrogen to trigger generalized effects on growth, which are the reflection of complex transcriptional factors and may differ from the effects of the “candidate” estrogen on ligand-mediated regulation of a single target gene or protein.130,131
Available data regarding the estrogenic/antiestrogenic potency of compounds are predominantly obtained with ligand-binding, transactivation assays and proliferation assays. Evidence shows that 1) it is difficult to compare data about the estrogenic potential of compounds evaluated by different methods using different end points; 2) the relative estrogenic potency of compounds determined with different assays might vary greatly; and 3) it cannot be expected that the correlation of the results obtained is always good. It is important to mention that the estrogenic potencies of compounds determined by different assays are not comparable, whereas the sensitivity of the different assays represented by the EC50 values for the standard estrogen 17β-estradiol differ greatly (by two or three orders of magnitude), the cell free binding assays being the less sensitive.132
The aforementioned broad spectrum of in vitro test systems may be useful to elucidate the mechanism of action of the test phytoestrogens and to set priorities for further assessment. It is important that the combination of several in vitro test systems be required in order to predict validly the effects in vivo. However, the in vitro assays do not include either metabolites of compounds or aspects of the absorption of compounds, thus they may generate false-negative or false-positive effects. In vivo studies are only able to predict the action of a substance in the organism, since under in vivo conditions the substance is exposed to absorption processes and multiple metabolic transformation.
In vivo test systems
Various in vivo assays (animal models) are used to characterize the estrogenic potency of phytoestrogens and their mechanism of action. The animals used are usually immature, hypophysectomized or ovariectomized rats, mice or rabbits. The test phytoestrogen is given orally or subcutaneously. Among the most common animal models are several tumor models, the uterotrophic assay and the analysis of estrogen sensitive endpoints (such as morphological, histological, biochemical and molecular endpoints) in the uterus and other estrogen sensitive target tissues, such as the vagina, the mammary gland, the liver, the bone, the cardiovascular system and the brain.
The uterotrophic assay assesses the ability of phytoestrogens to stimulate uterine growth. However, this assay may not be exactly suitable for assessing estrogenicity, since there are compounds, like raloxifene, that exert tissue-specific estrogen-like activity without effects on the uterus.133 Tumor models are mainly used to assess the potential chemopreventive properties of phytoestrogens. There are several approaches, such as spontaneous carcinogenesis, chemical carcinogen-induced tumor models and tumor models by xenotransplantation of tumor cells. Spontaneous carcinogenesis may be applied for prostate and the endometrium. Chemical carcinogen-induced tumor models involve the exposure of rats to DMBA or NMU for the development of mammary carcinomas. Finally, some tumor cell lines, if xenotransplanted to immunodeficient nude mice or rats, grow tumors at the ectopic site which eventually metastasize through blood or lymphatics. This model is usually used for breast, prostate and endometrial tumors.134
Additionally, there are more specific animal models to evaluate the possible beneficial effects of phytoestrogens in osteoporosis, atherosclerosis and neurodegeneration. These include the ovariectomized adult rat model for osteoporosis, the rabbit model for high-cholesterol induced atheromatosis and several animal models for brain injury.135-145
In conclusion, the use of a suitable panel of different in vitro test systems combined with a final assessment in animal models will predict the real estrogenic potential of phytoestrogens.
CONCLUSION
A high-phytoestrogen diet may result in various health effects; however, some of the results obtained are still controversial. It must be stressed that present data are limited in characterizing the biological effects of soy products and the isoflavones genistein, daidzein, while only a few data exist on lignans. laboratory evidence indicates that apart from the so far known phytoestrogens, e.g. the isoflavones, lignans, coumestanes, the stilbenes and the flavonoids quercetin and kaempferol, there are many more phytoestrogens identified in nature; thus the spectrum of phytoestrogens is expanding and their biological role awaits determination. The experimental methodology followed is of high importance in defining the estrogenic effect of phytochemicals, the in vitro studies providing the first clues about the estrogenic potency, whereas the use of animal models indicates the real estrogenic potential of phytoestrogens. Many data have shown a wide range of hormonal activitiesof phytoestrogens (estrogenic/antiestrogenic via the ER subtypes). Overall, however, phytoestrogen biological activity depends on multiple factors, e.g. the chemical form of thephyto estrogen, the route of administration, the metabolism, the intrinsic estrogenic status and the time and the level of exposure. On the market there are several preparations containing varying concentrations of individual phytoestrogens of different origin and their use as dietary supplements may result in high levels of phytoestrogens, relative to those provided in whole foods. In addition, the idea that what is “natural” is also safe may not prove valid.
In this respect, the consequences of phytoestrogen intake may not necessarily be beneficial and in some cases may actually increase disease risk. Because of the lack of well-designed, prospective studies in humans for individual phytoestrogens and due to the fact that the commercial preparations of phytoestrogens are not subjected to any regulatory controls, dietary recommendations for individual phytoestrogens cannot be given. Caution is rather suggested for their consumption and each preparation should be thoroughly evaluated individually before it is used as a dietary supplement or as a therapeutic agent.
REFERENCES
1. Deroo BJ, Korach KS, 2006 Estrogen receptors and human disease. The J Clin Invest 116: 561-570.
2. McDonnell DP, Connor CE, Wijayaratne A, Chang C-Y, Norris JD, 2002 Definition of the molecular and cellular mechanisms underlying the tissue-selective agonist/ antagonist activities of selective estrogen receptor modulators. Recent Prog Horm Res 57: 295-316.
3. Writing Group for the Women’s Health Initiative Investigators, 2002 Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 288: 321-333.
4. Cassidy A, 2003 Potential risks and benefits of phytoestrogen-rich diets. Int J Vitam Nutr Res 73: 120-126.
5. Evans RM, 1988 The steroid and thyroid hormone superfamily. Science 240: 889-895.
6. Beato M, Herrlich P, Schutz G, 1995 Steroid hormone receptors. Many actors in search of a plot. Cell 83: 851 2. 858.
7. Greene Gl, Gilna P, Waterfield M, et al, 1986 Sequence and expression of human estrogen receptor complementary DNA. Science 231: 1150-1154.
8. Mosselman S, Polman J, Dijkema R, 1996 ERβ: identification and characterization of a novel human estrogen receptor. FEBS lett 392: 49-53.
9. Nilsson S, Makela S, Treuter E, et al, 2001 Mechanisms of estrogen action. Physiol Rev 81: 1535-1565.
10. Moore JT, McKee DD, Slentz-Kesler K, et al, 1998 Cloning and characterization of human estrogen receptor beta isoforms. Biochem Biophys Res Commun 247: 75-78.
11. Horwitz KB, Jackson TA, Bain Dl, Richer JK, Takimoto GS, Tung l, 1996 Nuclear receptor coactivators and corepressors. Mol Endocrinol 10: 1167-1177.
12. Freedman I, 1999 Increasing the complexity of coactivation in nuclear receptor signaling. Cell 97: 5-8.
13. Wolffe AP, Hayes JJ, 1999 Chromatin disruption and modification. Nucleic Acids Res 27: 711-720.
14. Moggs JG, Orphanides G, 2001 Estrogen receptors: orchestrators of pleiotropic cellular responses. EMBO 2. 21: 775-781.
15. Wolffe AP, 1997 Chromatin remodeling regulated by steroid and nuclear receptors. Cell Res 7: 127-142.
16. Cerillo G, Rees A, Manchanda N, et al, 1998 The oestrogen receptor regulates NF-kB and AP-1 activity in a cell-specific manner. J Steroid Biochem Molec Biol 67: 79-88.
17. Paech K, Webb P, Kuiper GG, et al, 1997 Differential ligand activation of estrogen receptors ERα and ERβ at AP1 sites. Science 277: 1508-1510.
18. Webb P, Lopez GN, Uht RM, Kushner PJ, 1995 Tamoxi-fen activation of the estrogen receptor/AP1 pathway: potential origin for the cell-specific estrogen-like effects of antiestrogens. Mol Endocrinol 9: 443-456.
19. McKay LI, Cidlowski JA, 1999 Molecular control of immune/inflammatory responses: interactions between nuclear factor-kappa B and steroid receptor-signaling pathways. Endocr Rev 20: 435-459.
20. Wissink S, Van Der Burg B, Katzenellenbogen BS, Van Der Saag PT, 2001 Synergistic activation of the serotonin 1α receptor by nuclear factor-kappa b and estrogen. Mol Endocrinol 15: 543-552.
21. Pfeilschifter J, Köditz R, Pfohl M, Schatz H,2002 Changes in proinflammatory cytokine activity after menopause. Endocr Rev 23: 90-119.
22. Quaedackers ME, Van Den Brink CE, Wissink S, et al, 2001 4-hydroxytamoxifen transrepresses nuclear factor-kappa B activity in human osteoblastic U2-OS cells through estrogen receptor (ER) alpha and not through ER beta. Endocrinology 142: 1156-1166.
23. Matthews J, Gustafsson JA, 2003 Estrogen signaling: a subtle balance between ERα and ERβ. Μol Interv 3: 281-292.
24. Castano E, Chen LW, Vorojeikina DP, Motides AC, 1998 The role of phosphorylation in human estrogen receptor function. J Steroid Biochem Molecul Biol 65: 101-110.
25. Kato S, Endoh H, Masuhiro Y, et al, 1995 Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science 270: 1491-1494.
26. Lu Q, Ebling H, Mittler J, Bahr WE, Karas RH, 2002 MAP kinase mediates growth factor-induced nuclear translocation of oestrogen receptor alpha. FEBS lett 2. 516: 1-8.
27. Ring A, Dowsett M, 2004 Mechanisms of tamoxifen resistance. Endocr Relat Cancer 11: 643-658.
28. Gangolli EA, Conneely OM, O’Malley BW, 1997 Neurotransmitters activate the human estrogen receptor in a neuroblastoma cell line. J Steroid Biochem Mol Biol 2. 61: 1-9.
29. Cenni B, Picard D, 1999 Ligand-independent activation of steroid receptors: new roles for old players 10: 41-46.
30. Zwijsen RMl, Wientjens E, Klompmaker R, Van der Sman J, Bernards R, Michalides RJAM, 1997 CDK-independent activation of estrogen receptor by Cyclin D1. Cell 88: 405-415.
31. Weigel NL, Zhang Y, 1998 Ligand-independent activation of steroid hormone receptors. J Mol Med 76: 469-479.
32. Trowbridge JM, Rogatsky I, Garabedian MJ, 1997 Regulation of estrogen receptor transcriptional enhancement by the cyclin A/Cdk2 complex. Proc Natl Acad Sci USA 2. 94: 10132-10137.
33. Hall JM, Couse JF, Korach KS, 2001 The multifaceted mechanisms of estradiol and estrogen receptor signal. J Biol Chem 276: 36869-36872.
34. Whilte R, Parker MG, 1998 Molecular mechanisms of steroid hormone action. Endocr Relat Cancer 5: 1-14.
35. Kelly MJ, Levin ER, 2001 Rapid actions of plasma membrane estrogen receptors. Trends Endocrinol Metab 12: 152-156.
36. Levin ER, 2002 Celular functions of plasma membrane estrogen receptors. Steroids 67: 471-475.
37. Simoncini T, Hafezi-Moghadam A, Brazil DP, ley K, Chin W, Liao JK, 2000 Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3-OH kinase. Nature 407: 538-541.
38. Solakidi S, Psarra AM, Sekeris C, 2005 Differential subcellular distribution of estrogenr eceptor isoforms: localization of ERα in the nucleoli and ERβ in the mitochondria of human osteosarcoma SaOS-2 and hepatocarcinoma HepG2 cell lines. Biochimica et Biophysica Acta 1745: 382-392.
39. Chen J-Q, Yager J, Russo J, 2005 Regulation of mitochondrial respiratory chain structure and function by estrogens/estrogen receptors and potential physiological/ pathophysiological implications. Biochimican et Biophysica Acta 1746: 1-17.
40. Couse JF, Korach KS, 1999 Estrogen receptor null mice: What have we learned and where will they lead us? Endocrine Reviews 20: 358-417.
41. Dubal DB, Zhu H, Yu J, et al, 2001 Estrogen receptor alpha, not beta, is a critical link in estradiol-mediated protection against brain injury. Proc Natl Acad Sci USA 2. 98: 1952-1957.
42. Baker AE, Brautinam VM, Watters JJ, 2004 Estrogen modulates microglial inflammatory mediator production via interactions with estrogen receptor beta. Endocrinology 145: 5021-5032.
43. Zhao L, Wu TW, Brinton RD, 2004 Estrogen receptor subtypes alpha and beta contribute to neuroprotection and increased Bcl-2 expression in primary hippocampal neurons. Brain Res 1010: 22-34.
44. Koehler KF, Helguero LA, Maldosen LA, Warner M, Gustafsson JA, 2005 Reflections on the discovery and significance of estrogen receptor beta. Endocr Rev 26: 465-478.
45. Chu S, Mamers P, Burger HG, Fuller PJ, 2000 Estrogen receptor isoform gene expression in ovarian stromal and epithelial tumors. J Clin Endocrinol Metab 85: 12001205.
46. Lazennec G, 2006 Estrogen receptor beta, a possible tumor suppressor involved in ovarian carcinogenesis. Cancer lett 231: 151-157.
47. Benassayag C, Perrot-Applanat M, Ferre F, 2002 Phytoestrogens as modulators of steroid action in target cells. J Chromatogr B 777: 233-248.
48. Atkinson C, Frankenfeld CL, Lampe JW, 2005 Gut bacterial metabolism of the soy isoflavone daidzein: exploring the relevance to human health. Exp Biol Med (Maywood) 230: 155-170.
49. Adlercreutz H, 2002 Phyto-estrogens and cancer. The lancet Oncol 3: 364-373.
50. Kris-Etherton PM, Hecker KD, Bonanome A, et al, 2002 Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am J Med 113(9B): 71S-88S.
51. Branca F, Lorenzetti S, 2005 Health effects of phytoestrogens. Forum Nutr 100-11.
52. Shelnutt SR, Cimino CO, Wiggins PA, Ronis MJ, Badger TM, 2002 Pharmacokinetics of the glucuronide and sulfate conjugates of genistein and daidzein in men and women after consumption of a soy beverage. Am J Clin Nutr 76: 588-594.
53. Zhang Y, Hendrich S, Murphy PA, 2003 Glucuronides are the main isoflavone metabolites in women. J Nutr 2. 133: 399-404.
54. Setchell KD, Cassidy A, 1999 Dietary isoflavones: biological effects and relevance to human health. J Nutr 129: 758S-767S.
55. Harris DM, Besselink E, Henning SM, Go VLW, Heber D, 2005 Phytoestrogens induce differential estrogen receptor alpha- or beta-mediated responses in transfected breast cancer cells. Exp Biol Med 230: 558-568.
56. Maskarinec G, Oshiro C, Morimoto Y, Hebshi S, Novotny R, Franke AA, 2005 Urinary isoflavone excretion as a compliance measure in a soy intervention among young girls: a pilot study. Eur J Clin Nutr 59: 369-375.
57. Setchell KD, Zimmer-Nechemias L, Cai J, Heubi JE, 1997 Exposure of infants to phytoestrogens from soy-based infant formula. Lancet 350: 23-27.
58. Adlercreutz H, Markkanen H, Watanabe S, 1993 Plasma concentrations of phyto-estrogens in Japanese men. Lancet 342: 1290-1210.
59. Setchell KD, Zimmer-Nechemias L, Cai J, Heubi JE, 1998 Isoflavone content of infant formulas and the metabolic fate of these phytoestrogens in early life. Am J Clin Nutr 2. 68: 1453S-1461S.
60. Franke AA, Haim BM, Custer LJ, Tatsumura Y, Hebshi S, 2006 Isoflavones in breastfed infants after mothers consume soy. Am J Clin Nutr 84: 406-413.
61. Berger-Achituv S, Shohat T, Romano-Zelekha O, et al, 2005Wide spread use of soy-based formula with outclinical indications. J Pediatr Gastroenterol Nutr 41: 660-666.
62. Chen A, Rogan WJ, 2004 Isoflavones in soy infant formula: a review of evidence for endocrine and other activity in infants. Annu Rev Nutr 24: 33-54.
63. Ross JA, Kasum CM, 2002 Dietary flavonoids: Bioavailability, metabolic effects and safety. Annu Rev Nutr 22: 19-34.
64. Mueller SO, Simon S, Chae K, Metzler M, Korach KS, 2004 Phytoestrogens and their human metabolites show distinct agonistic and antagonistic properties on estrogen receptor α (ERα) and ERβ in human cells. Toxicological Sciences 80: 14-25.
65. Kinjo J, Tsuchihashi R, Morito K, et al, 2004 Interactions of phytoestrogens with estrogen receptors α and β (III). Estrogenic activities of soy is of lavoneaglycones and their metabolites isolated from human urine. Biol Pharm Bull 27: 185-188.
66. Fletcher RJ, 2003 Food sources of phyto-oestrogens and their precursors in Europe. Br J Nutr 89 : Suppl 1: 39-43.
67. Albertazzi P, Purdie DW, 2002 The nature and utility of the phytoestrogens: a review of the evidence. Maturitas 2. 42: 173-185.
68. Brzezinski A, Debi A, 1999 Phytoestrogens: the “natural” selective estrogen receptor modulators? Eur J Obstetr Gynecol Reprod Biol 85: 47-51.
69. Cos P, Bruyne T, Apers S, et al, 2003 Phytoestrogens: recent developments. Planta Med 69: 589-599.
70. Branca F, Lorenzetti S, 2005 Health effects of phytoestrogens. Forum Nutr 57: 100-111.
71. Bowers J, Tyulmenkov V, Jernigan S, Klinge C, 2000 Resveratrol acts as a mixed agonist/antagonist for estrogen receptors α and β. Endocrinology 141: 3657-3667.
72. van der Woude H, ter Veld M, Jacobs N, van der Saag P, Murk A, Rietjens I, 2005 The stimulation of cell proliferation by quercetin is mediated by the estrogen receptor. Mol Nutr Food Res 49: 763-771.
73. Kostelac D, Rechkemmer G, Briviba K, 2003 Phytoestrogens modulate binding response of estrogen receptors α and β to the estrogen response element. J Agric Food Chem 51: 7632-7635.
74. Zava DT, Duwe G, 1997 Estrogenic and antiproliferative properties of genistein and other flavonoids in human breast cancer cells in vitro. Nutr Cancer 27: 31-40.
75. Cappelletti V, Fioravanti L, Miodini P, Di Fronzo G, 2000 Genistein blocks breast cancer cells in the G(2)M phase of the cell cycle. J Cell Biochem 79: 594-600.
76. Levenson AS, Gehm BD, Pearce ST, et al, 2003 Resveratrol acts as an estrogen receptor (ER) agonist in breast cancer cells stably transfected with ERα. Int J Cancer 2. 104: 587-596.
77. Shao ZM, Shen ZZ, Fontana JA, Barsky SH, 2000 Genistein’s “ER-dependent and independent” actions are mediated through ER pathways in ER-positivebreast carcinoma cell lines. Anticancer Res 20: 2409-2416.
78. Boettger-Tong H, Murthy L, Chiappetta C, et al, 1998 A case of a laboratory animal feed with high estrogenic activity and its impact on in vivo responses to exogenously administered estrogens. Environ Health Perspect 106: 369-373.
79. Santell RC, Chang YC, Nair MG, Helferich WG, 1997 Dietary genistein exerts estrogenic effects upon the uterus, mammary gland, and the hypothalamic/pituitary axis in rats. J Nutr 127: 263-269.
80. Virgili F, Acconcia F, Ambra R, Rinna A, Totta P, Marino M, 2004 Nutritional flavonoids modulate estrogen receptor α signaling. IUBMB life 56: 145-151.
81. Somjen D, Kohen E, Lieberherr M, et al, 2005 Mem-branal effects of phytoestrogens and carboxy derivatives of phytoestrogens on human vascular and bone cells: new insights based on studies with carboxy-biochanin A. J Steroid Biochem Mol Biol 93: 293-303.
82. Watson CS, Bulayeva NN, Wozniak Al, Finnerty CC, 2005 Signaling from the membrane via membrane es-trogen receptor-alpha: estrogens, xenoestrogens and phytoestrogens. Steroids 70: 364-371.
83. Le Bail J-C, Champavier Y, Chulia A-J, Habrioux G, 2000 Effects of phytoestrogens on aromatase, 3β and 17β-hydroxysteroid dehydrogenase activities and human breast cancer cells. life Sciences 66: 1281-1291.
84. Basly JP, Lavier MC, 2005 Dietary phytoestrogens: potential selective estrogen enzyme modulators? Planta Med 71: 287-294.
85. Wattel A, Kamel S, Prouillet C, et al, 2004 Flavonoid quercetin decreases osteoclastic differentiation induced by RANKL via a mechanism involving NF kappa B and AP-1. J Cell Biochem 92: 285-295.
86. Ise R, Han D, Takahashi Y, et al, 2005 Expression profiling of the estrogen responsive genes in response to phytoestrogens using a customized DNA microarray. FEBS Lett 579: 1732-1740.
87. Schreihofer DA, 2005 Transcriptional regulation by phytoestrogens in neuronal cell lines. Mol Cell Endicrinol 2. 231: 13-22.
88. Xiao CW, Wood C, Gilani GS, 2006 Nuclear receptors: potential biomarkers for assessing physiological functions of soy proteins and phytoestrogens. J AOAC Int 2. 89: 1207-1214.
89. Vina J, Borras C, Gambini J, Sastre J, Pallardo FV, 2005 Why females live longer than males? Importance of the upregulation of longevity-associated genes by oestrogenic compounds. FEBS lett 579: 2541-2545.
90. Halabalaki M, Aligiannis N, Papoutsi Z, et al, 2000 Three new arylobenzofurans from onobrychis ebenoides and evaluation of their binding affinity for the estrogen receptor. J Nat Prod 63: 1672-1674.
91. Papoutsi Z, Kassi E, Papaevangeliou D, et al, 2004 Plant 2-arylobenzofurans demonstrate a selective estrogen receptor modulator profile. Steroids 69: 727-734.
92. Papoutsi Z, Kassi E, Mitakou S, et al, 2006 Acteoside and martynoside exhibit estrogenic/antiestrogenic properties. J Steroid Biochem Mol Biol 98: 63-71.
93. Papoutsi Z, Kassi E, Tsiapara A, Fokialakis N, Chrousos GP, Moutsatsou P, 2005 Evaluation of estrogenic/antiestrogenic activity of ellagic acid via the estrogen receptor subtypes Eralpha and Erbeta. J Agric Food Chem 53: 7715-7720.
94. Fokialakis N, Lambrinidis G, Mitsiou D, et al, 2004 A new class of phytoestrogens; evaluation of the estrogenic activity of deoxybenzoins. Chem Biol 11: 397-406.
95. Pearce V, Nawaz Z, Xiao W, Wiedenfeld D, Boyle N, SmithD,2003 4- Ethoxymethylphenol: a novel phytoestrogen that acts as an agonist for human estrogen receptors. J Steroid Biochem Mol Biol 84: 431-439.
96.Kuruto-Niwa R, Inoue S, Ogawa S, Muramatsu M, Nozawa R, 2000 Effects of tea catechins on the ERE-regulated estrogenic activity. J Agric Food Chem 48: 6355-6361.
97. Burow M, Boue S, Collins-Burow B, et al, 2001 Phytochemical glyceollins, isolates from soy, mediate antihor-monal effects through estrogen receptor α and β. J Clin Endocrinol Metab 86: 1750-1758.
98.Usui T, Ikeda Y, Tagammi T, et al, 2002 The phytochemical lindleyin, isolated from Rhei rhizoma, mediates hormonal effects through estrogen receptors. J Endocrinol 175: 289-296.
99. Lee Y, Jin Y, Lim W, et al, 2003 A ginsenoside-Rh1, a component of ginseng saponin, activates estrogen recep-tor in human breast carcinoma MCF-7 cells. J Steroid Biochem Mol Biol 84: 463-468.
100.Tamir S, Eizenberg M, Somjen D, Izrael S, Vaya J, 2001 Estrogen-like activity of glabrene and other constituents isolated from licorice root. J Steroid Biochem Mol Biol 78: 291-298.
101.Maggiolini M, Statti G, Vivacqua A, et al, 2002 Estrogenic and antiproliferative activities of isoliquiritigenin in MCF7 breast cancer cells. J Steroid Biochem Mol Biol 82: 315-322.
102.Rowlands JC, Berhow MA, Badger TM, 2002 Estrogenic and antiproliferative properties of soy sapogenols in hu-man breast cancer cells in vitro. Food and Chem Toxicol 40: 1767-1774.
103.Adlercreutz H, Heinonen SM, Penalvo-Garcia J, 2004 Phytoestrogens, cancer and coronary heart disease. Biofactors 22: 229-236.
104.Limer JL, Speirs V, 2004 Phytoestrogens and breast cancer chemoprevention. Breast Cancer Res 6: 119-127.
105.Peeters PH, Keinan-Boker L, vander Schouw YT, Grobbee DE, 2003 Phytoestrogens and breast cancer risk. Review of the epidemiological evidence. Breast Cancer Res Treat 77: 171-183.
106.Adlercreutz H, 2002 Phytoestrogens and breast cancer. J Steroid Biochem Mol Biol 83: 113-118.
107.Boccardo F, Puntoni M, Guglielmini P, Rubagotti A, 2006 Enterolactone as a risk factor for breast cancer: a review of the published evidence. Clin Chim Acta 365: 58-67.
108.Verheus M, van Gils CH, Keinan-Boker L, Grace PB, Bingham SA, Peeters PH, 2007 Plasma phytoestrogens and subsequent breast cancer risk. J Clin Oncol 25: 648 655.
109.Messina M, McCaskill-Stevens W, lampe JW, 2006 Ad-dressing the soy and breast cancer relationship: review, commentary and workshop proceedings. J Natl Cancer Inst 98: 1275-1284.
110.Hilakivi-Clarkel, ChoE, Onojafe I, Raygada M,Clarke R, 1999 Maternal exposure to genistein during pregnancy increases carcinogen-induced mammary tumorigenesis in female rat offspring. Oncol Rep 6: 1089-1095.
111.Newbodl RR, Banks EP, Bullock B, Jefferson WN, 2001 Uterine adenocarcinoma in mice treated neonatally with genistein. Cancer Res 61: 4325-4328.
112.Kurzer MS, 2003 Phytoestrogen supplement use by women. J Nutr 133: 1983S-1986S.
113. Day JK, Besch Williford C, McMann TR, et al, 2001 Dietary genistein increased DMBA-induced mammary adenocarcinoma in wild-type, but not ER alpha KO, mice. Nutr Cancer 39: 226-232.
114. Whitten Pl, lewis C, Russell E, Naftolin F, 1995 Potential adverse effects of phytoestrogens. J Nutr 125: 771S-776S.
115. Hilakivi-Clarke l, Cho E, deAssisS,et al, 2001 Maternal and prepubertal diet, mammary development and breast cancer risk. J Nutr 131: 154S-157S.
116. Whitsett TG Jr, lamartinlere CA, 2006 Genistein and resveratrol: mammary cancer chemoprevention and mechanisms of action in the rat. Expert Rev Anticancer Ther 6: 1699-1706.
117. Kurahashi N, Iwasaki M, Sasazuki S, Otani T, Inoue M, Tsunane S, Japan Public Health Center-Based Prospective Study Group, 2007 Soy product and isoflavone consumption in relation to prostate cancer in Japanese men. Cancer Epidemiol Biomarkers Prev 16: 538-545.
118.Morrissey C, Watson RW, 2003 Phytoestrogens and prostate cancer. Curr Drug Targets 4: 231-241.
119.Cassidy A, Albertazzi P,Lise Nielsen I, e tal, 2006 Critical review of health effects of soyabean phytoestrogens in post-menopausal women. Proc Nutr Soc 65: 76-92.
120.Sirtori CR, Arnoldi A, Johnson SK, 2005 Phytoestrogens: end of a tale? Ann Med 37: 423-438.
121. Clarkson TB, 2002 Soy, soy phytoestrogens and cardiovascular disease. J Nutr 132: 566S-569S.
122.Jin Y, Wu H, Cohen EM, et al, 2007 Genistein and daidzein induce neurotoxicity at high concentrations in primary rat neuronal cultures. J Biomed Sci 14: 275-284.
123. Kajta M, Domin H, Grynkiewicz G, lason W, 2007 Genistein inhibits glutamate-induced apoptotic processes in primary neuronal cell cultures: An involvement of aryl hydrocarbon receptor and estrogen receptor/glycogen synthase kinase-3beta intracellular signaling pathway. Neuroscience 145: 592-604.
124. Sonee M, Sum T, Wang C, Mukherjee SK, 2004 The soy isoflavone, genistein, protects human cortical neuronal cells from oxidative stress. Neurotoxicology 25: 885 2. 891.
125. Choi EJ, Lee BH, 2004 Evidence for genistein mediated cytotoxicity and apoptosis in rat brain. Life Sci 75: 499 2. 509.
126. Goddard IW, 2003 Soy-induced brain atrophy? J Anti Aging Med 6: 335-336.
127. Cassidy A, 2003 Potential risks and benefits of phytoestrogen-rich diets. Int J Vitam Nutr Res 73: 120-126.
128.Usui T, 2006 Pharmaceutical prospects of phytoestrogens. Endocr J 53: 7-20.
129. Piersen CE, 2003 Phytoestrogens in botanical dietary supplements: implications forc ancer. Integr Cancer Ther 2: 120-138.
130. Mueller SO, 2002 Overview of in vitro tools to assess the estrogenic and antiestrogenic activity of phytoestrogens. J Chromatography B 777: 155-165.
131.Diel P, Smolnikar K, Michna H, 1999 In vitro testsystems for the evaluation of the estrogenic activity of natural products. Planta Medica 65: 197-203.
132.Gutendorf B, Westendorf J, 2001 Comparison of an array of in vitro assays for the assessment of the estrogenic potential of natural and synthetic estrogens, phytoestrogens and xenoestrogens. Toxicology 166: 79-89.
133.Jefferson WN, Padilla-Banks E, Clark G, Newbold RR, 2002 Assessing estrogenic activity of phytochemicals using transcriptional activation and immature mouse uterotrophic responses. J Chromatogr B Analyt Technol Biomed Life Sci 777: 179-189.
134.Diel P, Schmidt S, Vollmer G, 2002 In vivo test systems forthequantitativeandqualitativeanalysisofthebiologi-cal activity of phytoestrogens. J Chromatogr B Analyt Technol Biomed life Sci 777: 191-202.
135.Jee WS, Yao W, 2001 Overview: animalmodels of osteopenia and osteoporosis. J Musculoskel Neuron Interact 1: 193-207.
136.Food and Drug Administration, 1994 Guidelines for preclinical and clinical evaluation of agents used in the prevention of treatment of postmenopausal osteporosis.
137.Kalu DN, 1991 The ovariectomized rat model of postmenopausal bone loss. Bone Miner 15: 175-191.
138.Picazo O, Azcoitia I, Garcia-Segura lM, 2003 Neuroprotective and neurotoxic effects of estrogens. Brain Res 990: 20-27.
139. Wise PM, 2000 Estradiol: a protective factor in the adult brain. J Pediatr Endocrinol Metab 13: Suppl 6: 14251429.
140. Hu X, Qiu J, Grafer MR, Rea HC, Rassin DK, Perez-Polo JR, 2003 Bcl-2 family members make different contributions to cell death in hypoxia and/or hyperoxia in rat cerebral cortex. Int J Dev Neurosci 21: 371-377.
141. Raghupathi R, Strauss KI, Zhang C, Krajewski S, Reed JC, McIntosh, 2003 Temporal alterations in cellular Bax:Bcl-2 ratio following trauma brain injury in the rat. J Neurotrauma 20: 421-435.
142. Garcia-Estrada J, Del Rio JA, luquin S, Soriano E, Garefa-Segura lM, 1993 Gonadal hormones down-regulate reactive gliosis and astrocyte proliferation after a penetrating brain injury. Bran Res 628: 271-278.
143. Garcia-Estrada J, luquin S, Fernandez AM, Garcia-Se-gura lM, 1999 Dehydroepiandrosterone, pregnenolone and sex steroids down-regulate reactive astroglia in the male rat brain after a penetrating brain injury. Int J Dev Neurosci 17: 145-151.
144. Azeoitia I, Sierra A, Garcia-Segura lM, 1998 Estradiol prevents kainic acid-induced neuronal loss in the rat dentate gyrus. Neuroreport 9: 3075-3079.
145. Azcoitia I, Sierra A, Garcia-Segura lM, 1999 Neuroprotective effects of estradiol in the adult rat hippocampus: interaction with insulin-like growth factor-I signalling. J Neurosci Res 58: 815-822.
Address for correspondence:
P. Moutsatsou, Department of Biological Chemistry,
Medical School, University of Athens,
75 Mikras Asias Str., 115 27 Goudi, Athens, Greece, Tel.-Fax: +30 210 7462682,
e-mail: pmoutsatsou@med.uoa.gr
Received 03-03-07, Revised 05-05-07, Accepted 06-06-07